Abstract
The Burseraceae is an important family of resin-producing trees and shrubs, which have yielded biologically active essential oils. Boswellia dalzielii and Canarium schweinfurthii are members of the family that are used in West African traditional medicine for a variety of ailments. The leaf essential oils of B. dalzielii have been obtained from 2 different locations in north-central Nigeria, while the leaf and stem bark essential oils of C. schweinfurthii have been obtained from 3 locations. The chemical compositions of the essential oils have been determined by gas chromatography-mass spectrometry and show wide variation, especially for the leaf essential oils. The leaf essential oils of B. dalzielii and C. schweinfurthii have been screened for antibacterial and antifungal activity; C. schweinfurthii leaf essential oil showed remarkable activity against Aspergillus niger with a minimum inhibitory concentration of 78.1 μg/mL.
Keywords
The Burseraceae is composed of 18 genera and around 640 species of mainly tropical trees and shrubs. 1 Historically and commercially important members of the family include frankincense (Boswellia spp.) and myrrh (Commiphora myrrha). 2 Several genera, especially Boswellia, Bursera, Canarium, Commiphora, Dacryodes, and Protium, have yielded biologically active essential oils. 3
The genus Boswellia ranges across Africa, the Arabian Peninsula, and India, and is the source of the aromatic oleo-gum resin frankincense. The West African species, Boswellia dalzielii Hutch, ranges from western Chad, through Nigeria and Burkina Faso, to Mali. 4 Bark decoctions of B. dalzielii have been used in traditional medicine to treat various maladies, including malaria, toothaches, sores, abscesses, mental disorders, yellow fever, asthma, wounds, and dysentery, 5 -9 while the leaves have been used to protect stored food from insect pests. 10,11
The leaf essential oil from Adamawa State, Nigeria, yielded an essential oil rich in α-pinene (45.7%) and γ-terpinene (11.5%), 12 while the leaf essential oil from Ségbana region, Benin, had α-pinene (15.2%), myrcene (5.7%), δ-3-carene (27.7%), p-cymene (9.5%), β-phellandrene (8.5%), and isolongifolene (6.2%), as major constituents. 13 Oleo-gum resin essential oils from Nigeria were found to contain α-pinene (21.7%-76.6%), α-thujene (2.0%-17.6%), myrcene (up to 35.2%), p-cymene (0.3%-15.6%), and limonene (1.1%-32.9%), 4 while oleo-gum resin essential oils from Burkina Faso were dominated by α-pinene (21.0%-56.0%), followed by carvone (2.1%-5.4%) and α-copaene (1.8%-5.0%). 14 Two chemotypes of B. dalzielii, based on oleo-gum resin essential oil compositions, have been described, 1 dominated by α-pinene and the other with myrcene as the major component. 4,14
Most of the species of Canarium are found in tropical Asia and the Pacific, 1 but 2 species are found in tropical Africa, Canarium madagascariense Engl., found along the eastern coast of Africa and the western and central regions of Madagascar and Canarium schweinfurthii Engl., found in the rainforest belt of central and West Africa. 15 A decoction of the bark of C. schweinfurthii is used traditionally to treat dysentery, gonorrhea, cough, chest pains, pulmonary affections, stomach complaints, and food poisoning; the pounded bark is used to treat leprosy and ulcers; and the oleo-gum resin is taken internally for roundworm infections and other intestinal parasites, and externally for skin infections and eczema. 16 Oleo-gum resin essential oils from Cameroon 17 and Gabon 18 have been dominated by limonene (42.7%-52.1%), while a resin essential oil from Uganda showed γ-terpinene (34.5%), α-phellandrene (17.9%), α-thujene (14.0%), β-phellandrene (12.9%), and p-cymene (8.5%) as the major components. 19
As part of our ongoing exploration of the essential oils of the Burseraceae, we have examined the essential oils of B. dalzielii and C. schweinfurthii growing in the wild in north-central, Nigeria.
Results and Discussion
Essential Oil Compositions
Boswellia dalzielii
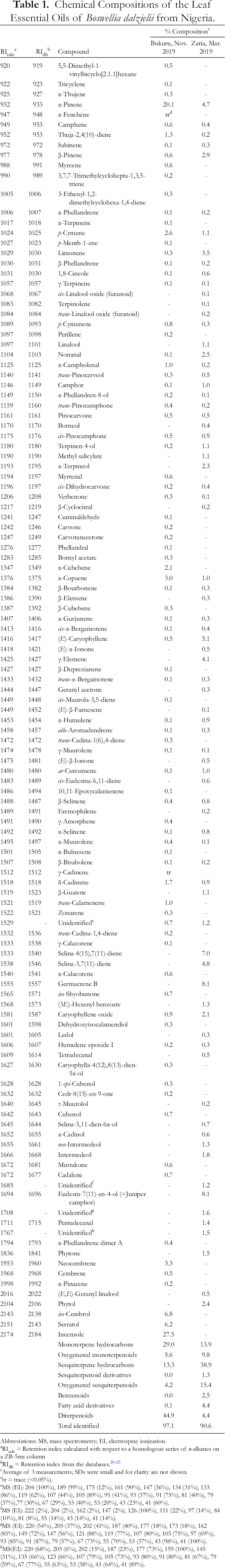
Hydrodistillation of leaves of B. dalzielii collected from Zaria, Kaduna State, and Bukuru, Plateau State, Nigeria (Figure 1(A)), yielded essential oils with yields of 1.76% and 1.65%, respectively. The essential oils were analyzed by gas chromatography-mass spectrometry (GC-MS) (Table 1).

Collection sites of Boswellia dalzielii (A) and Canarium schweinfurthii (B).
Chemical Compositions of the Leaf Essential Oils of Boswellia dalzielii from Nigeria.
MS, mass spectrometry; EI, electrospray ionization.
aRIcalc = Retention index calculated with respect to a homologous series of n-alkanes on a ZB-5ms column
cAverage of 3 measurements; SDs were small and for clarity are not shown.
dtr = trace (<0.05%).
eMS (EI): 204 (100%), 189 (99%), 175 (12%), 161 (90%), 147 (36%), 134 (31%), 133 (86%), 119 (62%), 107 (44%), 105 (89%), 95 (41%), 93 (37%), 91 (75%), 81 (40%), 79 (37%),77 (30%), 67 (29%), 55 (40%), 53 (20%), 43 (23%), 41 (60%).
fMS (EI): 222 (2%), 204 (2%), 162 (2%), 147 (2%), 126 (100%), 111 (22%), 97 (14%), 84 (10%), 81 (8%), 55 (14%), 43 (14%), 41 (14%).
gMS (EI): 220 (54%), 205 (37%), 202 (41%), 187 (40%), 177 (18%), 173 (18%), 162 (85%), 149 (72%), 147 (56%), 121 (88%), 119 (77%), 107 (80%), 105 (75%), 97 (69%), 93 (85%), 91 (87%), 79 (57%), 67 (73%), 55 (70%), 53 (37%), 43 (98%), 41 (100%).
hMS(EI): 220 (68%), 205 (6%), 202 (15%), 187 (23%), 177 (73%), 159 (100%), 145 (31%), 135 (66%), 123 (66%), 107 (79%), 105 (73%), 93 (80%), 91 (80%), 81 (67%), 79 (59%), 67 (77%), 55 (63%), 53 (38%), 43 (64%), 41 (89%).
The 2 leaf essential oils of Nigerian B. dalzielii show significant quantitative differences. The sample from Zaria, Kaduna State, which was collected during the dry season, had low concentrations of monoterpene hydrocarbons (13.9%), but relatively large concentrations of sesquiterpene hydrocarbons (38.9%) and oxygenated sesquiterpenoids (15.4%). The major components in the Zaria sample were germacrene B (8.1%), juniper camphor (8.1%), selina-4(15),7(11)-diene (7.0%), and (E)-caryophyllene (5.1%). The concentration of α-pinene was low, only 4.7%. In contrast, the leaf essential oil from Bukuru, Plateau State, which was collected during the rainy season, was rich in monoterpene hydrocarbons (20.1% α-pinene) and the diterpenoids incensole (27.5%), iso-cembrol (6.8%), and serratol (6.2%). The leaf essential oil of B. dalzielii from Gombi, Adamawa State, Nigeria, showed α-pinene (45.7%) and γ-terpinene (11.5%) as the major components, with only low concentrations of sesquiterpene hydrocarbons (2.5%) or oxygenated sesquiterpenoids (2.6%); diterpenoids were absent. 12 The major components in the leaf essential oil of B. dalzielii from Ségbana, North Benin, were δ-3-carene (27.7%), α-pinene (15.2%), p-cymene (9.5%), β-phellandrene (8.5%), isolongifolene (6.2%), and myrcene (5.7%). 13 No diterpenoids were reported in the Benin sample. Thus, the essential oils from the leaves of B. dalzielii show a wide variation in chemical composition. It is difficult, however, to account for these variations at this time. The 4 samples were collected from different geographical locations and at different times of the year, both factors of which may profoundly affect the chemical compositions.
Canarium schweinfurthii
Leaves and stem bark samples were obtained from C. schweinfurthii from Azare, Bauchi State; Bukuru, Plateau State; and Donga, Taraba State, Nigeria (Figure 1B). The leaf essential oils were obtained in 1.35%, 1.42%, and 1.82% yield, respectively, while the bark essential oils were obtained in 1.83%, 1.76%, and 1.85% yield, respectively. The chemical compositions of C. schweinfurthii leaf and bark essential oils are compiled in Tables 2 and 3, respectively.
Chemical Compositions of the Leaf Essential Oils of Canarium schwienfurthii from Nigeria.

MS, mass spectrometry; EI, electrospray ionization.
aRIcalc = Retention index calculated with respect to a homologous series of n-alkanes on a ZB-5ms column.
cAverage of 3 measurements; SDs were small and for clarity are not shown.
dtr = trace (<0.05%).
eMS (EI): 140 (12%), 126 (100%), 111 (8%), 98 (37%), 95 (20%), 81 (20%), 70 (24%), 67 (26%), 55 (28%), 53 (17%), 43 (51%), 41 (42%).
fMS (EI): 220 (44%), 205 (25%), 202 (31%), 187 (30%), 162 (77%), 149 (56%), 147 (50%), 135 (29%), 121 (71%), 119 (75%), 107 (68%), 105 (66%), 97 (53%), 93 (67%), 91 (71%), 79 (49%), 67 (63%), 55 (59%), 53 (28%), 41 (100%), 41 (83%).
gMS (EI): 220 (71%), 205 (7%), 202 (10%), 187 (18%), 177 (79%), 159 (100%), 145 (27%), 135 (62%), 123 (66%), 107 (78%), 105 (66%), 93 (76%), 91 (76%), 81 (63%), 67 (73%), 55 (68%), 43 (70%), 41 (95%).
Chemical Compositions of the Stem Bark Essential Oils of Canarium schwienfurthii from Nigeria.

MS, mass spectrometry; EI, electrospray ionization.
aRIcalc = Retention index calculated with respect to a homologous series of n-alkanes on a ZB-5ms column.
cAverage of 3 measurements; SDs were small and for clarity are not shown.
dMS (EI): 152 (3%), 134 (28%), 119 (17%), 109 (24%), 95 (21%), 92 (100%), 91 (86%), 81 (51%), 79 (40%), 77 (19%), 70 (14%), 67 (14%), 65 (14%), 55 (14%), 53 (18%), 43 (26%), 41 (34%).
eMS (EI): 168 (5%), 153 (5%), 140 (13%), 139 (10%), 126 (100%), 98 (32%), 95 (16%), 81 (11%), 70 (16%), 67 (18%), 55 (15%), 53 (14%), 43 (46%), 41 (38%).
There was considerable variation in the leaf essential oil compositions of C. schweinfurthii. Monoterpene hydrocarbons (54.9%) were the principal class of compounds in the sample from Bukuru, Plateau State, with limonene (19.3%), α-pinene (17.4%), β-pinene (10.9% account for the observe), and p-cymene (5.2%), as the major monoterpene components. The Bukuru sample was also rich in juniper camphor (eudesm-7(11)-en-4α-ol, 16.1%). The leaf essential oil sample collected from Donga, Taraba State, on the other hand, showed a predominance of sesquiterpene hydrocarbons (56.1%), dominated by (E)-caryophyllene (14.6%), germacrene B (14.6%), and γ-elemene (7.1%). The essential oil from leaves collected from Azare, Bauchi State, had (E)-caryophyllene (18.7%), caryophyllene oxide (9.1%), limonene (7.2%), and phytol (6.3%) as the major components. As was observed for B. dalzielii leaf essential oils, the compositions of C. schweinfurthii leaf essential oils also show wide variation. The stem bark essential oils of C. schweinfurthii were dominated by monoterpene hydrocarbons (14.5%-22.7%) and oxygenated monoterpenoids (62.2%-77.0%). The major monoterpene was limonene (3.4%-18.3%), while α-terpineol (24.1%-38.1%) and terpinen-4-ol (3.6%-8.6%) were the main oxygenated monoterpenoids. To our knowledge, there have been no previous reports on the leaf or stem bark essential oils of C. schweinfurthii. Essential oils from the oleo-gum resins have been previously reported, however, and these materials are generally dominated by monoterpenes. 17 -19
Antibacterial and Antifungal Activity
The leaf essential oils of B. dalzielii (Zaria sample) and C. schweinfurthii (Bukuru sample) have been screened for antibacterial and antifungal activity (Table 4). The best activity was observed for C. schweinfurthii leaf essential oil against Aspergillus niger (minimum inhibitory concentration, MIC = 39.1 µg/mL). The major components in the Bukuru sample of C. schweinfurthii essential oil were limonene (19.3%), α-pinene (17.4%), juniper camphor (16.1%), β-pinene (10.9%), and p-cymene (5.2%). Several of the major essential oil components have been screened for antifungal and antibacterial activity (Table 5). The antifungal activities of α-pinene, β-pinene, as well as α-terpineol (MIC = 78.1 µg/mL) against A. niger (Table 5), may account for the observed activity of C. schweinfurthii leaf essential oil. The high concentration of juniper camphor may also be a factor but that compound was not available to us for screening. Note that juniper camphor was also relatively abundant in B. dalzielii leaf essential oil from Zaria (8.1%), but the essential oil did not exhibit a pronounced effect on A. niger (MIC = 156.3 µg/mL).
Antibacterial and Antifungal Activities (MIC, μg/mL) of Burseraceae EOs from Nigeria.

MIC, minimum inhibitory concentration; EO, essential oil.
aGentamicin for bacteria, amphotericin B for fungi.
Antibacterial and Antifungal Activities (MIC, μg/mL) of Individual Chemical Components.

MIC, minimum inhibitory concentration.
Conclusions
The leaf and stem bark essential oils of C. schweinfurthii are reported for the first time. The leaf essential oil of C. schweinfurthii showed pronounced antifungal activity against A. niger. The leaf essential oils of B. dalzielii have also been analyzed. The leaf essential oils of both B. dalzielii and C. schweinfurthii show a wide variation in chemical compositions, which may be attributed to geographical location and climatic conditions. The variations in essential oil compositions may have important consequences in the traditional uses of these members of the Burseraceae.
Materials and Methods
Plant Material
Leaves of B. dalzielii were collected directly from source trees from Kabanni Rock, Zaria (11°06′ 40.61″ N, 7°43′ 21.72″ E, 644 m elev., Zaria local government, Kaduna State, northern Nigeria, located some 82.3 km north-east of Kaduna town) during the dry season in the month of March 2019 (Figure 1A). Leaves of B. dalzielii were also collected from Bukuru (9°47′ 38.36″ N, 8°51′ 50.29″ E, 1277 m elev., Jos South Local Government, Plateau State) in the month of November 2019. The plants were taxonomically identified and authenticated by Muh’d Sule and Namadi Sunusi. A voucher specimen (Voucher number 0900121) has been deposited in the Department of Biological Sciences, Ahmadu Bello University, Nigeria. The leaves were manually removed, air-dried in the laboratory for 7 days, and then pulverized using an electric blender. Samples (450 g) of leaves of B. dalzielii from Kaduna and Plateau States (Jos) were subjected to hydrodistillation in an all-glass Clevenger-type apparatus. The B. dalzielii samples and water were mixed in a ratio of 2:6, and the mixtures were stirred constantly during hydrodistillation for 3-4 hours. The yields of the oils were 1.76% and 1.65% on a dry weight basis, respectively. The oils were dried over anhydrous sodium sulfate and stored in sealed vials under refrigeration (4 °C) prior to analysis.
Canarium schweinfurthii leaf and stem bark samples were taken directly from source trees in 3 states in northern Nigeria: Azare (11°40′ 29.21″N, 10°11′ 26.48″E, 644 m elev., Katagum Local Government, Bauchi State), Bukuru (9°47′ 38.36″N, 8°51′ 50.29″E, 1277 m elev., Jos South Local Government, Plateau State), and Donga (7°43′ 00″N, 10°03′ 00″E, 135 m elev., situated on Donga River in Donga Local Government, Taraba State) (Figure 1B). Both leaves and stem bark of C. schweinfurthii were obtained during the dry season in the month of March 2019. The plants were taxonomically identified and authenticated by Namadi Sunusi. A voucher specimen (voucher number 02639) has been deposited in the Department of Biological Sciences, Ahmadu Bello University, Nigeria. The leaves and stem bark were manually removed, air-dried in the laboratory for 7-10 days, and then pulverized using an electric blender. Samples (450 g each) of both leaves and stem bark of C. schweinfurthii, respectively, were subjected to hydrodistillation in an all-glass Clevenger-type apparatus. Each of the C. schweinfurthii samples and water were mixed in a ratio of 2:6, and the mixtures were stirred constantly during hydrodistillation for 3 hours until no more essential oil was apparent in the distillate. The yields of the leaf essential oils were 1.35%, 1.42%, and 1.82% yield, respectively, while the bark essential oils were obtained in 1.83%, 1.76%, and 1.85% yield, respectively, on a dry weight basis. The oils were dried over anhydrous sodium sulfate and stored in sealed vials under refrigeration (4 °C) prior to analysis.
Gas Chromatography-Mass Spectrometry
The essential oils from B. dalzielii and C. schweinfurthii were subjected to GC-MS analysis as previously reported 4 : Shimadzu GCMS-QP2010 Ultra, electron impact (EI) mode with electron energy = 70 eV, scan range = 40-400 atomic mass units, scan rate = 3.0 scans/seconds, and Shimadzu GC-MS solution software v. 4.45 (Shimadzu Scientific Instruments, Columbia, MD, USA); ZB-5ms fused silica capillary GC column (Phenomenex, Torrance, CA, USA); (5% phenyl)-polymethylsiloxane stationary phase, 0.25 µm film thickness; helium carrier gas, column head pressure = 552 kPa, flow rate = 1.37 mL/min; injector temperature = 260 °C, ion source temperature = 260 °C; GC oven temperature program: initial temperature = 50 °C, temperature increased 2 °C/minute to 260 °C. For each sample, a 5% w/v solution in dichloromethane was prepared, 0.1 µL was injected using a split ratio of 30:1. Each sample was injected 3 times. Identification of the individual components of the essential oils was determined by comparison of the retention indices, determined using a series of n-alkanes, in addition to the comparison of the mass spectral fragmentation patterns with those found in the MS databases 20 -23 using the LabSolutions GC-MS solution software v. 4.45 (Shimadzu Scientific Instruments, Columbia, MD, USA) and with matching factors >90%.
Antibacterial and Antifungal Screening
The essential oils were screened for antibacterial activity against bacteria (Bacillus cereus [ATCC No. 14579], Staphylococcus aureus [ATCC No. 29213], and Staphylococcus epidermidis [ATCC No. 12228]), and for antifungal activity against the molds (Aspergillus fumigatus [ATCC No. 96918], Aspergillus niger [ATCC No. 16888], Cryptococcus neoformans [ATCC No. 32045], Microsporum canis [ATCC No. 11621], Microsporum gypseum [ATCC No. 24102], Trichophyton mentagrophytes [ATCC No. 18748], and Trichophyton rubrum [ATCC No. 28188]), and 1 yeast (Candida albicans [ATCC No. 18804]) using the microbroth dilution technique 24,25 as previously reported. 26,27 Individual essential oil components ((E)-caryophyllene, (+)-α-pinene, (−)-α-pinene, (–)-β-pinene, p-cymene, (+)-limonene, (–)-limonene, α-terpineol, (E)-caryophyllene, caryophyllene oxide, and phytol were obtained from Sigma-Aldrich (St. Louis, MO, USA) and were used as received without additional purification.
All bacteria were cultured on tryptic soy agar (Sigma-Aldrich, St. Louis, MO, USA). All fungi were cultured on yeast malt agar (Sigma-Aldrich, St. Louis, MO, USA). For the bacteria and fungi, 50 µL of 1% (w/v) solution of the samples in dimethyl sulfoxide (DMSO) was diluted in 50 µL of cation-adjusted Mueller Hinton broth (CAMHB) (Sigma-Aldrich, St. Louis, MO, USA). The sample solutions were then serially diluted (1:1) in fresh CAMHB to obtain concentrations of 2500, 1250, 625, 313, 156, 78, 39, and 20 µg/mL. The microbes were harvested from a fresh culture and added to each well at a concentration of approximately 1.5 × 108 colony-forming unit (CFU)/mL for bacteria and 7.5 × 107 CFU/mL for fungi, and the 96-well microdilution plates for bacteria were incubated at 37 °C and the fungi were incubated at 35 °C for 24 hours. The minimum inhibitory concentration (MIC) was determined as the lowest concentration with no turbidity. Gentamicin (Sigma-Aldrich, St. Louis, MO, USA) was used as a positive antibiotic control and DMSO was used as the negative control (50 µL DMSO diluted in 50 µL broth medium, and then serially diluted as above). For fungi, the above-mentioned method was implemented using yeast-nitrogen base growth medium (Sigma-Aldrich, St. Louis, MO, USA) and Amphotericin B (Sigma-Aldrich, St. Louis, MO, USA) as a positive antifungal control.
Footnotes
Acknowledgments
This work was carried out as part of the activities of the Aromatic Plant Research Center (APRC, https://aromaticplant.org/).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
