Abstract
Quality problems with Schisandra chinensis (Turcz.) Baill (SCB) and Schisandra sphenanthera Rehd.et Wils. (SSR) have been increasing owing to their obvious difference in supply and demand. In this study, qualitative and quantitative studies were carried out on the contents of various components of SCB and SSR, which provided the reference for the quality control of the raw materials of SCB and SSR. A ZORBAX Eclipse Plus C18 column (1.8 μm, 100 mm × 2.1 mm) was used with a column temperature of 40 °C, a mobile phase consisting of a gradient of acetonitrile and water, a flow rate of 0.3 mL/min, and an injection sample of 1 µL. Ultraviolet detection was operated at 220 nm throughout the analyzing time. Almost all the compounds achieved baseline separation. In addition, this method showed a favorable recovery (98.3%-102.5% for SCB and 99.1%-100.5% for SSR) and good precision and stability, confirming the reliability of the experimental data.
Schisandra chinensis (Turcz.) Baill (SCB) was promoted in European therapy to a new pharmacopoeial plant species in 2009, and a monograph for it appeared for the first time in Supplement 2009 to the Polish Pharmacopeia, 8th Ed. (equivalent to Supplements 6.3-6.5 in the European Pharmacopeia, 6th Ed.). Since then, SCB has been widely used. According to the Pharmacopeia of the People’s Republic of China (PPRC), 1 prescriptions of 101 Chinese patent medicines contained SCB, while only 11 contained Schisandra sphenanthera Rehd.et Wils. (SSR). The obvious difference between supply and demand has led to the price of SCB being much higher than that of SSR. In addition, some enterprises have also been driven by their own interests by replacing SCB with SSR. On the other hand, the components and pharmacological actions of SCB and SSR have their own advantages. Therefore, it was necessary to conduct an in-depth study of them to give full play to their respective advantages, their rational use, and the sustainable development of resources.
It is generally believed that lignans are the main active components of SCB and SSR, which are used in the treatment of asthma, 2 acute myocardial infarction, 3 neurodegenerative diseases, 4 -6 for antiaging, 7 and hepatoprotection. 8,9 Schisantherin A (Sth A), Schisantherin B (Sth B), and Schisandrin A (Sin A) are the main lignans present in large quantities in SSR, whereas Schisandrol A (Sol A), Schisandrol B (Sol B), Sth A, Sth B, Sin A, Schisandrin B (Sin B), and Schisandrin C (Sin C) are the main lignans present in large quantities in SCB. Sth A has been used for the quality evaluation of SSR owing to its bioactive effect and high content in the herb, but Sin A was used for SCB following the procedures stated in PPRC. 1 Sth A is common to both SSR and SCB, and a single component such as Sin A alone could not be responsible for the pharmacological activities of SCB because different compounds, which might work synergistically, have different pharmacological activities. Thus, it was not enough that 1 particular lignan constituent was determined alone.
Of course, using as many as possible components simultaneously to evaluate the quality of traditional Chinese medicine (TCM) also seemed unfeasible because most chemical reference substances were not commercially available. In addition, most TCMs are so complex that the procedures of separating, identifying, and determining several components at the same time are rather tedious for pharmaceutical quality evaluation. A large number of papers have reported a variety of methods, using high-performance liquid chromatography (HPLC) with ultraviolet (UV) detection, 10,11 capillary electrochromatography (CEC), 12 and liquid chromatographic-mass spectrometry (LC-MS). 13,14 At present, HPLC is widely used for the determination of lignans in SCB and SSR. However, this method takes a long time and consumes a lot of organic solvents. 15,16 Rapid resolution liquid chromatography (RRLC), which has the advantages of short analysis time and less solvent consumption, might be a good way to solve this problem. In this experiment, a rapid method for the determination of 7 lignans (Sol A, Sol B, Sth A, Sth B, Sin A, Sin B, and Sin C) was established by RRLC, and the contents of these in SCB and SSR from different regions were determined, which provides the reference for the quality control of SCB and SSR.
Results and Discussion
Comparison of the RRLC chromatograms using a ZORBAX Eclipse Plus C18 column Rapid Resolution HD 2.1 × 100 mm I.D., 1.8 µm (Agilent, USA) with a ZORBAX Eclipse Plus C18 column Rapid Resolution HD 2.1 × 50 mm I.D., 1.8 µm (Agilent, USA) led to the former being accepted with regard to factors such as degree of separation and the peak number. Taking into consideration factors such as the number of peaks, separation effect, and analysis time factors, we chose acetonitrile-water as the mobile phase in gradient elution comparing the chromatograms of different mobile phase conditions (acetonitrile-water, methanol-water). 15 Using diode-array detector (DAD) ultraviolet detection, all reference compounds had significant UV absorption at 220 nm. With the same determined analysis conditions except for various flow rates (0.3, 0.35, 0.4 mL/min), the results showed that an increased flow rate was accompanied by a shortened analysis time, an increased pressure rise, and a decline in the degree of separation. Considering the analysis time, degree of separation and pressure, a flow rate of 0.3 mL/min was chosen. With the other conditions maintained the same, column temperatures of 30 °C, 35 °C, and 40 °C were used. The degree of separation was raised with the boosted temperature. In consideration of the applicable temperature of the chromatographic column and separation effect, a column temperature of 40 °C was adopted.
Three-parallel injections of blank solvent (90% ethanol solution), mixed calibration standards, SCB sample solution, and SSR sample solution were carried out respectively. As shown in Figure 1, the results showed that all target compounds were well separated and the sample solvent for determination of composition caused no interference or overlapping. Typical RRLC profiles of SSR and SCB are shown in Figure 1(B),(C). Seven calibration curves were formed by plotting the peak area ratios (y) versus the corresponding concentrations (x) with 7 different linearity ranges requiring correlation coefficients (r) of 0.99 or better (Table 1). Good linear relationships (r = 0.9997 for all analytes, except 0.9996 for Sin B) are listed in Table 1. Both the limit of detection (LOD) and limit of quantification (LOQ) for the analytes were determined by signal-to-noise ratios of 3/1 and 10/1, respectively. All compounds showed a commendable sensitivity, and the LOD and LOQ for the 7 lignans were demonstrated around a range of 0.174-0.254 µg/mL and 0.381-0.636 µg/mL, respectively.

Typical chromatograms for determination of (A) mixed calibration standards, (B) Schisandra chinensis, and (C) Schisandra sphenanthera (peak 1 = Sol A, 2 = Sol B, 3 = Sth A, 4 = Sth B, 5 = Sin A, 6 = Sin B, 7 = Sin C, using the proposed separation conditions: mobile phases: B—acetonitrile and D-water in the gradients as follows: 0-0.2 minutes, 45%-53% B; 0.2-8 minutes, 53%-55% B; 8-11 minutes, 55%-95% B; 11-14 minutes, 95% B; flow rate: 0.3 mL/min; Column: ZORBAX Eclipse Plus C18 (1.8 µm, 100 mm × 2.1 mm); protected column: a ZORBAX SB-C18 guard column (1.8 µm, 5.0 mm × 2.1 mm); column temperature: 40 °C; injection volume: 1 µL; ultraviolet detection: 220 nm. (Peak 1 = SolA, 2 = Sol B, 3 = SthA, 4 = Sth B, 5 = Sin A, 6 = Sin B, 7 = Sin C, respectively.)
Linearity, Regression Equation, LOD, LOQ, and Linear Range for the Reference Compounds.

LOD, limit of detection; LOQ, limit of quantification.
Mixed calibration standards were injected 6 times, continuously. The results showed that the relative SD (RSD) of the corresponding peak areas for Sol A, Sol B, Sth A, Sth B, Sin A, Sin B, and Sin C were 0.99%, 1.0%, 1.7%, 1.6%, 0.74%, 0.78%, and 0.87%, respectively, which demonstrated that this novel method was reliable and repeatable. Stability was verified by RRLC analyses performed at time intervals of 0, 1, 2, 4, 8, 12, 16, and 24 hours after sample solution preparation, with samples kept at room temperature. The stability of the test solution was evaluated through the rate of change by comparing the peak area after various time intervals with that of the original prepared solutions (0 hours). The RSD of Sol A, Sol B, Sth A, Sth B, Sin A, Sin B, and Sin C for SCB were 0.97%, 1.00%, 1.90%, 1.10%, 1.90%, 0.97%, and 1.10%, respectively, and 0.47%, 0.70%, and 0.12% for SSR. All the analytes showed great stability; the rate of change of the analytes was below 1.90% at time intervals of 24 hours after preparation. Accuracy was assessed through the recovery test using the standard addition method. Three known concentration levels (about equivalent to 0.8, 1.0, and 1.2 times of the concentration of the sample) of the 7 lignan standards were joined into corresponding 0.075 g samples in SCB and SSR in triplicates. The results of the recovery tests were obtained by comparing the results of the original samples with the fortified samples. The mean recoveries of the 7 lignans were 98.3%-102.5% with an RSD less than 1.90% for SCB, and 99.1%-100.5% with an RSD less than 1.73% for SSR. The same batches of SCB and SSR were used to prepare 6 sample solutions. The RSD of the relative retention time and relative peak area was less than 1.9%, indicating that this RRLC method was precise for the quantitative analysis of the major active compounds in SCB and SSR.
Different chromatographic parameters were used for the separation of the sample solution to assess the influence of change caused by the following conditions: flow rate 0.28, 0.30, and 0.32 mL/min and column temperature of 38 °C, 40 °C, and 42 °C. Only 1 factor was changed at a time, while the others remained unchanged. The influence of the parameters (flow rate and column temperature) on the resolution of the sample solution are detailed in Table 2, from which it could be deduced that this method in these test ranges was robust because of the high-quality peak separation and symmetry under the above conditions.
Summary of Resolution Values for Method Stability Testing for the 7 Lignan Compounds.
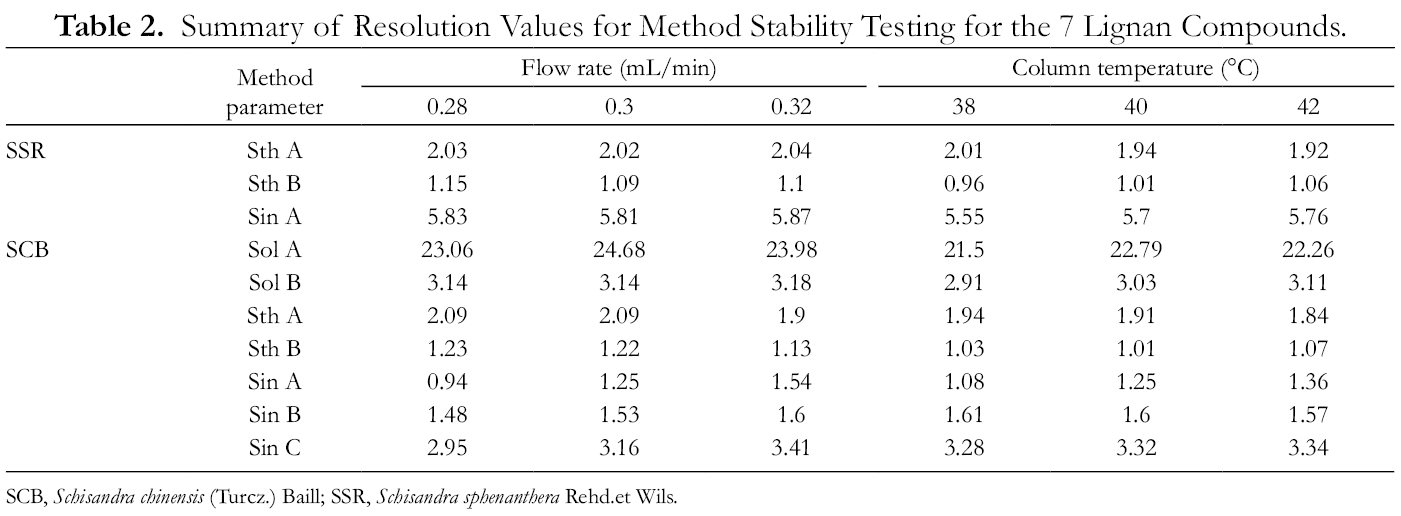
SCB, Schisandra chinensis (Turcz.) Baill; SSR, Schisandra sphenanthera Rehd.et Wils.
Nowadays, RRLC is widely used for the multicomponent analysis of TCM because it involves less organic solvent consumption, a shorter testing time, and higher resolution in contrast with HPLC. The novel RRLC method used in this study allowed a rapid, reliable, and efficient test for simultaneously measuring 7 lignans (Sol A, Sol B, Sth A, Sth B, Sin A, Sin B, and Sin C) within 14 minutes. Meanwhile, this method was successfully verified in view of good repeatability, linearity, precision, and accuracy.
Based on this method, we could establish quality standards for SCB and SSR through multicomponent qualitative and quantitative determination, and further evaluate the quality of the corresponding proprietary Chinese medicine on the market. The contents of 10 batches of SCB and 9 batches of SSR were determined by this method. As shown in Table 3, almost all SSR did not contain Sol A, Sol B, Sin A, and Sin B, except for d. (SSR). The lignan contents of SCB and SSR were significantly different between batches and from different origins. In general, the lignan contents of SCB were higher than those of SSR. Based on this, we could distinguish SCB from SSR. Therefore, this method could provide a reference for the quality control of SCB and SSR.
Lignan Contents of Schisandra chinensis and Schisandra sphenanthera From Different Habitats (n = 19) mg/g.

LOD, limit of detection.
Note: “—” indicates that it was below LOD.
Experimental
Materials and Chemicals
HPLC-grade acetonitrile (CH3CN) was purchased from Merck (USA). A Milli-Q Integral 5 system (Millipore, USA) was used to produce ultrapure water. Sol B (purity: >98%, lot number: 15072404), Sth A (purity: >98%, lot number: 15082707), Sth B (purity: >98%, lot number: 15012609), Sin B (purity: >98%, lot number: 15042206), and Sin C (purity: >98%, lot number: 15042207) were provided by Chengdu Biopurify Phytochemicals Ltd. (Chengdu, China), while Sol A (purity: 99.9%, lot number: JBSG-FAKJ) and Sin A (purity: 99.5%, lot number: 1A11-1B9N) were purchased from National Institutes for Food and Drug Control (Beijing, China). Ten batches of SCB and 9 batches of SSR were purchased from Shandong, Heilongjiang, Henan, Shanxi, Sichuan, Gansu, Jilin, Guangxi, and Liaoning provinces (Table 3).
Apparatus and Chromatographic Conditions
The liquid chromatography was performed on an Agilent 1200 series RRLC system (Agilent, USA) consisting of a quad-pump, a micro-vacuum degasser, a DAD, an auto-sampler, and a thermostatted column compartment. The analytes were separated using gradient elution on a ZORBAX Eclipse Plus C18 column (1.8 µm, 100 mm × 2.1 mm, Agilent, USA) protected by a ZORBAX SB-C18 guard column (1.8 µm, 5.0 × 2.1 mm, Agilent, USA) with a mobile phase B (CH3CN) and mobile phase D (water) at a flow rate of 0.3 mL/min. The percentage of organic modifier (phase B) was changed linearly as follows: 0-0.2 minutes, 45%-53% B; 0.2-8 minutes, 53%-55% B; 8-11 minutes, 55%-95% B; 11-14 minutes, 95% B, with a total liquid chromatography analysis time per injection of 14 minutes and a post-run of 6 minutes. The sample injection volume was set at 1 µL, and wavelength monitoring was performed at 220 nm; the column temperature was maintained at 40 °C.
Sample and Standard Solutions Preparation
The standard stock solutions were prepared by dissolving Sol A (2.46 mg), Sol B (1.59 mg), Sth A (1.38 mg), Sth B (1.18 mg), Sin A (2.38 mg), Sin B (2.81 mg), and Sin C (1.09 mg) in 10 mL methanol. All the stock solutions were stored at 4 °C in a fridge. The standard stock solution with diluted in methanol into a series of standard solutions (dilution factor: 1, 2, 5, 10, 25, 50, 125, 250). These solutions were analyzed by the RRLC method, and the calibration curves of the analytes were constructed by plotting the peak area (y-axis) versus concentration (x-axis). The chromatogram of the mixture of 7 standard lignans is shown in Figure 1(A).
About 20 g dried raw herbs of SCB/SSR kept in a desiccator were ground into a fine powder. Approximately 0.15 g SCB/SSR were accurately weighed and mixed with 90% ethanol aqueous solution in 25 mL volumetric flasks, then ultrasonicated for 10 minutes, and cooled to room temperature. The volumes were adjusted to 25 mL with 90% ethanol aqueous solution. All prepared samples were filtered through 0.20 µm syringe filters before injecting into the RRLC system.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
