Abstract
The cause of the immunogenicity of Acanthopanax senticosus injection has been studied. The method used for the preparation of the injection includes boiling the plant material in water, precipitation with ethanol, and filtration to produce the extract. In the current study, this extract was treated with saturated ammonium sulfate to collect the protein from the extract. Indicators of systemic allergy, including total immunoglobulin (IgE), interleukin -4 (IL-4), and interferon-gamma (IFN-γ) levels in the antiserum of Guinea pigs sensitized with this protein extract were used to determine the immunogenicity of the protein extract. Testing was performed using Western blotting to identify IgE in antiserum binding in Guinea pigs sensitized by injection. The target protein band was further identified by matrix-assisted laser desorption/ionization-time of flight mass spectrometry. The protein extract from A. senticosus caused positive reactions and significantly increased the levels of total IgE, IL-4, and IFN-γ in the serum of sensitized Guinea pigs. Western blotting showed that a group of proteins with a molecular weight of 60 000 in the A. senticosus protein extract acted as the major immunogenic protein by binding to serum IgE of Guinea pigs sensitized with A. senticosus injection. The protein was a pyruvate kinase homologous protein with high sequence similarity to the pyruvate kinase protein of Vitis vinifera. It was concluded that the allergic reaction caused by the injection of A. senticosus may be related to the interaction of these macromolecular proteins within the body.
Acanthopanax senticosus (Rupr. et Maxim.) Harms (Family: Araliaceae) injection is a traditional Chinese medicine, utilizing extracts from the roots, rhizomes, and stems of the species. 1 It has the functions of strengthening the liver and kidneys, strengthening bone, and can improve and regulate the body’s immune function. 2 -5 Since the publication of the first case report in 1988, reports of its adverse reactions have gradually increased. 6 Of the 33 types of traditional Chinese medicine injections (TCMI) listed in the 2004 National Essential Drugs List, the number of adverse drug reaction (ADR) reports for A. senticosus ranked fourth. 7 The recorded ADR of A. senticosus injection includes rash, dizziness, headache, and fever. According to the World Health Organization (WHO), ADR severity grading standard, A. senticosus injection can be life-threatening grade I and can lead to pathophysiological changes in various organs. 8 As a result, serious complications from the use of the injection cannot be ignored and the allergic compounds of A. senticosus injection urgently require identification and quality improvement.
The production of A. senticosus injection is by water extraction and alcohol precipitation. The main disadvantage of this method is that it is impossible to completely remove macromolecular substances such as plant proteins. Indirect enzyme-linked immunosorbent assay (ELISA) and ultrafiltration electrophoresis experiments have been used to detect the presence of certain macromolecular proteins in A. senticosus injection, and the results suggest that these proteins have strong immunogenicity and the ability to trigger type I allergic reactions. 9 Different methods, including chemical reaction, molecular weight distribution, and ELISA, have been used to identify the immunogenicity of A. senticosus injection. However, due to the different sensitivities of these methods, each has its own advantages and disadvantages. 10 -12 It has, therefore, been difficult to obtain comprehensive and reliable information on macromolecular substances in trace residual impurities in TCMI by only 1 method.
Drawing on the concept of proteomics, this study used Western blotting coupled with mass spectrometry to identify immunologic proteins in A. senticosus injection, to provide a methodological reference for the basic screening of allergens in TCMI.
Materials and Methods
Animals and Materials
Specific pathogen-free (SPF) male and female white Guinea pigs ranging from 250 to 350 g body weight and certified SCXK (Hei) 2 008 004 were provided by the Safety Evaluation Center of Heilongjiang University of Chinese Medicine and maintained under SPF conditions for 5 days prior to experimentation.
Acanthopanax senticosus was bought from Harbin Sankeshu Medicine Market (Hulin, Heilongjiang, China); A. senticosus extract was prepared by boiling the plant material in water and precipitation with ethanol, according to the Chinese Pharmacopeia Commission Prescription of Chinese Medicine (Volume 18) of Drug Standards. 1
Other materials used in this study included protein molecular weight standard reagent markers (Beijing Kangrun Chengye Biotechnology Co., Ltd.), Guinea pig total immunoglobulin E (IgE) kit, horseradish peroxidase (HRP)-anti-Guinea IgE, Guinea pig interferon (IFN)-r kit (RD company, USA), A. senticosus injection (batch number 20160702), and HRP-labeled IgE secondary antibody (batch number 160024, Shanghai Yiji Industrial Co., Ltd., China) and an Autoflex speed matrix-assisted laser desorption (MALDI)/ionization time of flight (TOF)/TOF-mass spectrometer (Bruker Dalton, Germany).
The animal experimental protocol was reviewed by the Animal Ethics Committee of Heilongjiang University of Traditional Chinese Medicine, license number: SN14073-D.
Extraction of Protein From A. senticosus Extract by the Saturated Ammonium Sulfate Method
According to the production process of A. senticosus injection, 1 A. senticosus powder was decocted twice, for 2 hours each time. The combined filtrates were concentrated, filtered, and alcohol added with stirring to reduce the volume to 50%. After standing for 12 hours and being centrifuged at 5000×g for 20 minutes, the supernatant was collected to obtain A. senticosus extract. One hundred milliliters of the extract was placed on a magnetic stirrer, and 80% saturated ammonium sulfate was slowly added to the ice bath as it was stirred. The solution was allowed to completely precipitate and was then centrifuged at 2100×g for 30 minutes to obtain a precipitate. The protein precipitate was dissolved in a small amount of pure water and then placed in a dialysis bag (molecular weight 3500) and continuously dialyzed in distilled water. The distilled water was continuously changed until the ammonium sulfate was completely removed. After dialysis, the membrane liquid was freeze-dried and the lyophilized product was subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis detection and content determination.
PAGE of Protein Products
For SDS-PAGE electrophoresis, a 15% polyacrylamide separation gel and 5% concentrated gel SDS-PAGE were used. Electrophoresis was carried out using a BG-verMINI vertical electrophoresis apparatus (Thermo Fisher, USA). Fifteen milligrams of protein extract was dissolved in 10 µL of loading buffer consisting of Tris-hydrochloric acid, pH 6.8 (60 mM), SDS (2%), bromophenol blue (0.1%), glycerin (25%), and β-mercaptoethanol (14.4 mM). This was heated in boiling water for 3 minutes before the protein solution was loaded onto the gel. The voltage used was set in a range of 60-120 V to assess the electrophoretic performance. After the electrophoresis was finished, the gel was dyed for 30-60 minutes and then rinsed with distilled water several times. The gel was then decolorized for 24 hours until the protein band was clear.
Determination of Protein Content in A. senticosus Extracts
The A. senticosus protein precipitate was dissolved in 2 mL of distilled water, and a 0.05 mL sample was placed in a test tube and diluted to 1 mL with distilled water. The solution (40 μL) was taken from each tube and placed in a colorimetric well, and 180 µL of Coomassie Brilliant Blue was added. The colored solution was mixed, allowed to stand for 2 minutes, and the absorbance measured using a colorimetric instrument at 595 nm.
Passive Cutaneous Anaphylaxis Test
Serum samples (100 µL) from Guinea pigs sensitized with A. senticosus protein precipitate were diluted 2-fold in physiological saline and then injected intradermally into the shaved sides of a Guinea pig. After 48 hours, sera transferred Guinea pigs were stimulated by injection of 1% Evan’s Blue containing A. senticosus protein precipitate. The animals were euthanized, and the diameter of the blue spot under the skin was recorded 30 minutes later.
Determination of IgE, IL-4, and IFN-γ in Guinea Pig Serum After Sensitization with Protein
Extract from A. senticosus
Eighteen Guinea pigs were randomly divided into 3 treatment groups: an A. senticosus protein extract group treated with 13.5 mg lyophilized protein extract powder dissolved in 1 mL water for injection; an ovalbumin (OVA) positive group treated with 1 mg ovalbumin dissolved in 1 mL water for injection; and a negative control group injected with saline. The experimental drugs were injected into the skin on the back of a Guinea pig at 4 points, once every other day for 3 treatments. Fourteen days after the last sensitization, the Guinea pigs were anesthetized with ether and blood was taken from the heart, allowed to coagulate at room temperature, centrifuged at 2100×g for 20 minutes to separate the serum, and placed in a refrigerator at −20°C for later use. Guinea pig , total IgE, interleukin (IL)-4, and IFN-γ assay kits were used to measure the total IgE, IL-4, and IFN-γ levels in Guinea pig antiserum by the double-antibody sandwich method, and the optical density (OD) values of each group were recorded.
Systemic Allergy
Fourteen days after the last sensitization, the same doses of drugs in each group were injected into the ear vein to challenge the sensitized Guinea pigs. Symptoms of systemic allergy were observed 30 minutes after injection and assessed as follows: 0 = no symptoms, 1 = scratching and rubbing around the nose, 2 = puffiness around the mouth and eyes, 3 = difficulty breathing, mouth and tail bun, 4 = convulsions, and 5 = death. 13
Immunological Characteristics and Protein Identification of A. senticosus Injection as Identified by Western Blotting
Guinea pigs were sensitized with A. senticosus injection (batch number 20160702, 5.6 mL/kg) to obtain antiserum, with the sensitization protocol being the same as for the protein extract.
The immunological characteristics of A. senticosus proteins were analyzed using the Western blotting method. The protein bands of the extract in the gel were transferred to the nitrocellulose membrane by using a Bio-Rad (Bio-Rad, USA) transfer tank, and then the nitrocellulose membrane was washed for 5 minutes twice with Tris-buffered saline (TBS). The membrane was blocked with 1% bovine serum albumin (BSA) blocking solution overnight at 4°C, and the membrane was washed 3 times for 5 minutes each time. Sensitized Guinea pig serum was added at a 1:2 dilution with 1% BSA-TBS, and the solution was incubated at 37°C. After incubation for 2 hours, the membrane was washed 3 times, and a 1:100-fold diluted secondary HRP-anti-Guinea IgE was added. The membrane was washed 5 times at room temperature for 2 hours and the color was revealed by exposure. The relative molecular mass was analyzed after imaging.
Mass Spectrometric Detection of Target Bands
Target strips in the SDS-PAGE gel underwent enzyme digestion and were then analyzed by MALDI-TOF/TOF-mass spectrometry. The ultraviolet (UV) wavelength was set to 355 nm, the repetition rate was 200 Hz, the acceleration voltage was 20 000 V, and the optimal mass resolution was 1500 Da. The scan quality range was 700-3200 Da. The mass spectra of all experimental samples were obtained using the default mode. Flex Analysis (Germany, Bruker Dalton Co., Ltd.) software was used to filter peaks and identify signal peaks. The NCBI database was searched using BioTools (Germany, Bruker Dalton Co., Ltd.) software to find related proteins with matching peptide masses ranging from 800 to 4000 Da.
Results
SDS-PAGE Results
The gel protein electrophoresis after SDS-PAGE is shown in Figure 1, where the molecular weights of the proteins in A. senticosus were mainly between 20 kDa and 70 kDa, with 4 distinct bands clearly visible.

Results of the sodium dodecyl sulfate-polyacrylamide gel electrophoresis of proteins extracted from Acanthopanax senticosus.
Determination of Protein Content by the Saturated Ammonium Sulfate Method
The total protein was quantitatively determined by the Coomassie Brilliant Blue method as 360.8 mg/kg.
Passive Cutaneous Anaphylaxis Test Results After Immunization
The serum of A. senticosus protein precipitate sensitized Guinea pigs caused positive reactions, with the mean diameter of the blue spot being 4.18 ± 0.19 mm and the positive reaction rate 67%. After passive injection of A. senticosus protein precipitate antiserum and stimulation by injection of 1% Evan’s Blue containing A. senticosus protein precipitate, the dye exudated into the skin to form a clear blue spot, as shown in Figure 2.
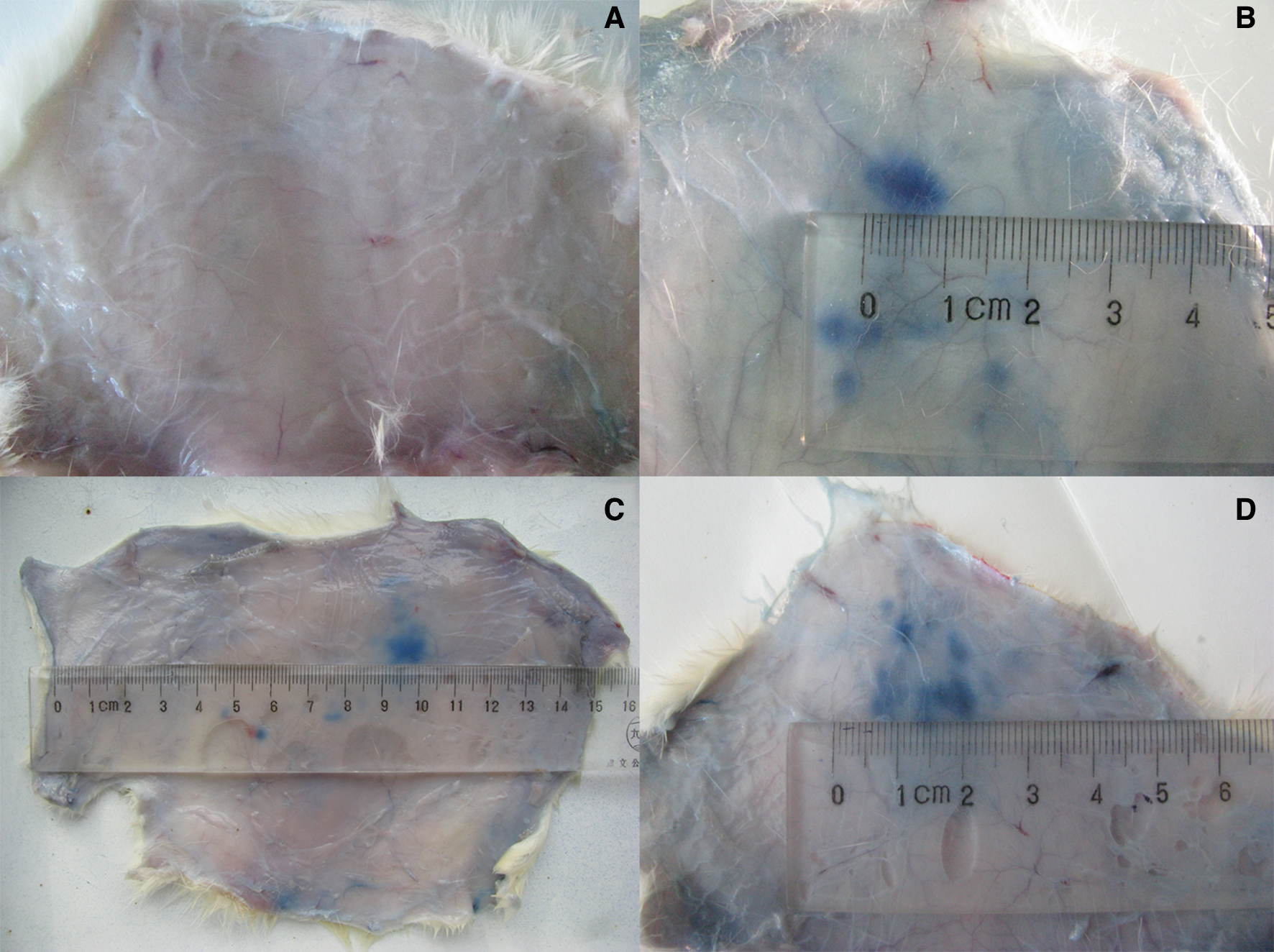
Passive cutaneous anaphylaxis results of (a) control group, (b) ovalbumin group (1 mg:1 mL) and (c, d) Acanthopanax senticosus protein precipitate group (13.5 mg:1 mL)
Total IgE, IL-4, and IFN-γ Levels of Protein-Sensitized Guinea Pigs
The total IgE, IL-4, and IFN-γ contents of the protein extracts in sensitized Guinea pigs are shown in Figure 3. The levels of total IgE, IL-4, and IFN-γ of the positive drug albumin group and the protein extract group were significantly different from those of the blank group (P < 0.05).
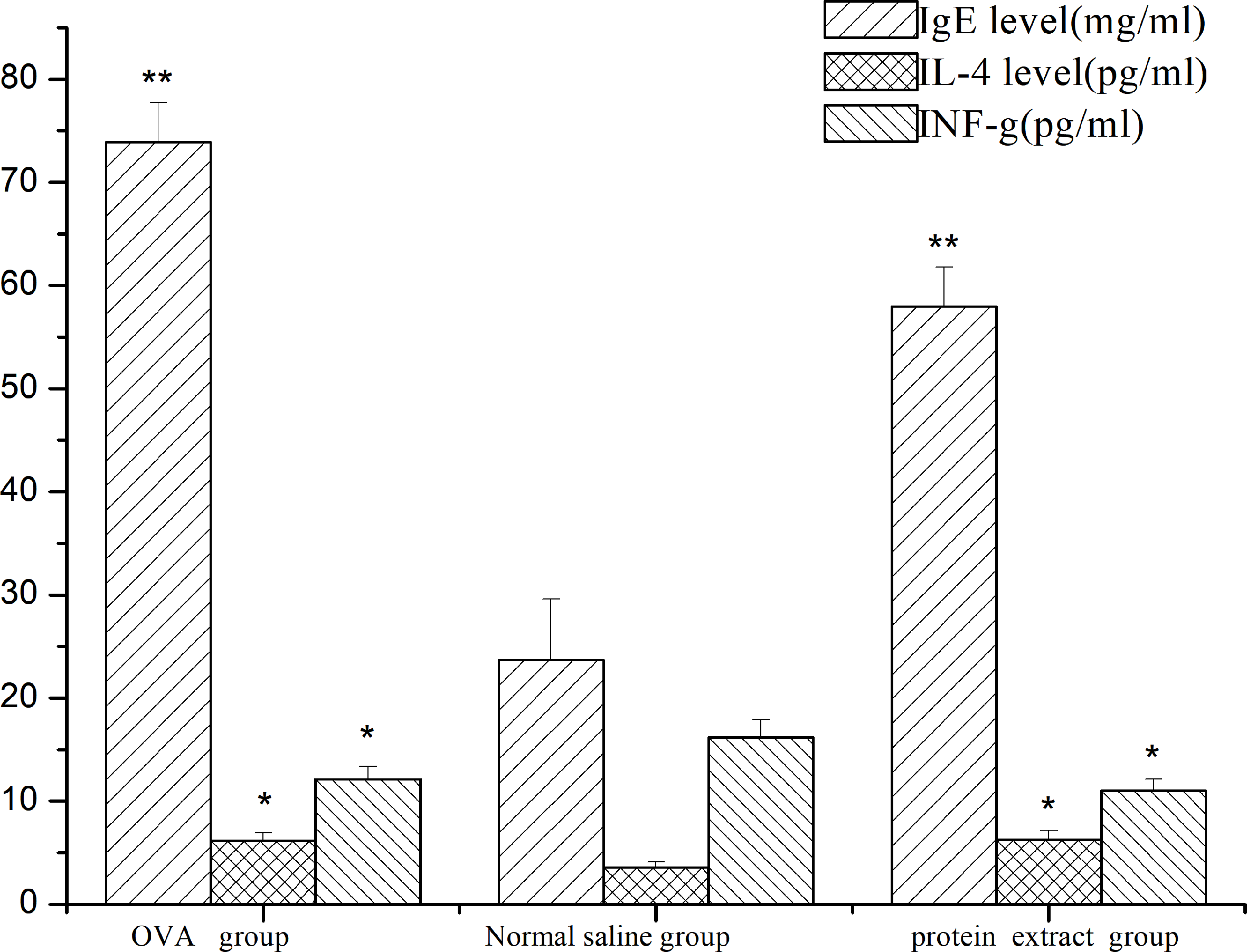
Levels of immunoglobulin E (IgE), interleukin-4 (IL-4), and interferon-gamma (IFN-γ) in guinea pig serum after sensitization of protein extract from Acanthopanax senticosus (±SD, n = 6, *P ≤ 0.05, **P ≤ 0.01).
Systemic Allergy Results
Obvious systemic reactions in the protein extract sensitized Guinea pigs were observed within 30 minutes after stimulation, while the saline negative control group Guinea pigs showed no reactions. In the animals sensitized with 13.5 mg protein extract, a number had puffiness around the mouth and eyes, while a number had convulsions (Figure 4).

System allergy symptom scores in guinea pigs. OVA, ovalbumin.
Identification of Immunogenic Protein in A. senticosus Injection
The immunogenic protein from A. senticosus protein extract was identified from the immunological characteristics of the Guinea pig mixed serum sensitized with A. senticosus injection (20160702). The results showed that a protein with a molecular weight of about 60 000 from the protein extract of A. senticosus could bind to IgE in Guinea pig serum sensitized with A. senticosus injection as the immunogenic protein (Figure 5).

Western-blotting results of protein from Acanthopanax senticosus injection. 1 and 2: Serum sensitized with A. senticosus injection (20160702); 3: negative serum.
Mass Spectrometric Identification of Target Proteins
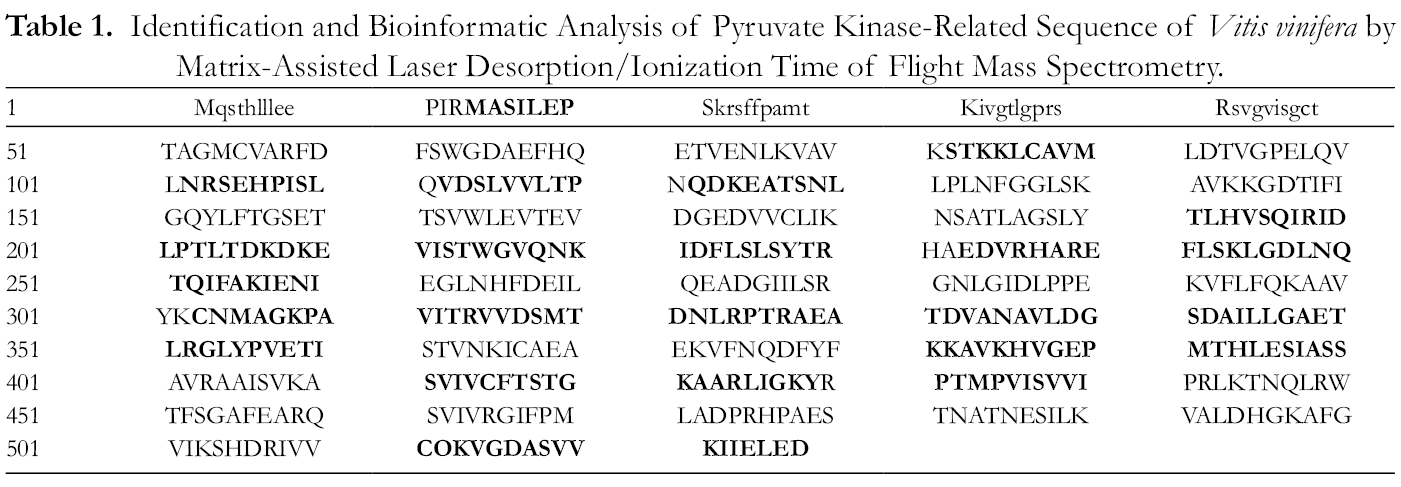
The target protein band selected from the SDS-PAGE gel was digested and identified by mass spectrometry to obtain a complete peptide fingerprint (Figure 6). The molecular weights ranged from 818.434 to 2485.305, of which 1366.695, 1592.825, 1705.904, and 1822.900 had a high signal to noise ratio. Mass spectrometric analysis of the abundance of the peptides was performed (Figure 7), followed by a Mascot search for the peptide quality information in the NCBI plant protein library. The nucleotide molecular weight information of the peptide fingerprint significantly matched the pyruvate kinase gi|225 449 659 peptides of Vitis vinifera (Tables 1 and 2), and, therefore, the target protein could be a pyruvate kinase homologous protein of A. senticosus.

First-order mass spectrometry detection results of the target protein band.

Second-order mass spectrometry detection results of the target protein band.
Identification and Bioinformatic Analysis of Pyruvate Kinase-Related Sequence of Vitis vinifera by Matrix-Assisted Laser Desorption/Ionization Time of Flight Mass Spectrometry.
Bioinformatic Analysis on the Purified 60 kDa Protein.

Conclusions
A passive cutaneous anaphylaxis test is suitable for most model animals because it is a highly sensitive drug allergic reaction detection method that can be used for the qualitative evaluation of a drug-specific immune response and also for quantitative evaluation of responses such as intensity or specificity or the amounts of IgE and IgG produced. 14 This study also examined the allergenic properties involving specific IgE binding of the protein extract, as well as more classic immunogenicity of specific IgG binding.
The results suggest that A. senticosus injection could significantly increase total IgE levels in the serum of sensitized Guinea pigs and could cause positive results in a passive cutaneous anaphylaxis test in these animals, which are associated with type I allergic reactions, consistent with the reported literature. 15 -17 In this study, due to the low content of protein in A. senticosus injection, proteins were extracted from A. senticosus extract, to increase the macromolecular protein substance levels. Pharmacological experiments indicated that the immunogenicity of A. senticosus injection may be residual protein impurities from the preparation process. To identify the immunogenicity of A. senticosus injection, the Western blotting analysis was used to identify allergic proteins and mass spectrometry to detect target bands obtained by Western blotting. This, and the mass spectrometry results, showed that the target protein, with a molecular weight of approximately 60 000 Da, should be a homologous pyruvate kinase protein of A. senticosus. This protein may be one that causes an allergic reaction to A. senticosus injection.
By clarifying the basis of the immunogenicity of A. senticosus injection, the causes can be either removed or reduced in the production process to establish a new quality standard for this TCMI. In addition, depending on the characteristics of the immunogenicity, a specific detection method for it in A. senticosus injection can be formulated.
In the subjects’ allergic reaction to a drug, the nature of the general antigen is directly related to the molecular weight. A small molecular weight substance is easily removed by the body, but when the molecular weight of an antigenic substance is more than 4000 Da, there is the capability to cause an allergic reaction. 18,19 In TCMI, proteins are the compounds that most commonly cause adverse sensitive reactions. 20 -22 Proteomics technology has been widely used in the study of allergic reactions and has become the main method for isolation, identification, and removal of immunogenicity. 23 -26 The strategy was to separate the prepared protein compounds by either SDS-PAGE or 2-dimensional PAGE and then electrotransfer to a cellulose acetate film (polyvinylidene difluoride); then the hybridization of the immunogenic experimental animal serum and the IgE antibody can be conducted. The immunogenicity can next be detected by DAB color development or enhanced chemiluminescence and the target band can be subjected to mass spectrometry. 27,28 In this study, Western blotting coupled with MALDI-TOF-mass spectrometry was used to offer a prospective method to analyze the immunogenicity of A. senticosus injections, which could provide guidance for identifying immunogenicity in other TCMI.
Footnotes
Acknowledgements
We thank International Science Editing (http://www.internationalscienceediting.com) for editing this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (No. 81703944), Outstanding Innovative Talents Project from Heilongjiang University of Chinese Medicine (2018), Heilongjiang Natural Science Foundation Project (YQ2019H031), and the Heilongjiang University of Chinese Medicine Province and Ministry Co-construction Key Laboratory of Open Fund (2017bs10).
