Abstract
In this study, antiphytopathogenic fungi activities of the leaf and cones essential oils and its constituents from Cunninghamia lanceolata were evaluated in vitro against 6 plant pathogenic fungi. The main compounds responsible for the antiphytopathogenic fungi activities were isolated and identified. The essential oil from the fresh leaves and cones of C. lanceolata was isolated using hydrodistillation in a Clevenger-type apparatus, and characterized by GC-FID and GC-MS, respectively. The leaf oil consisted primarily of ferruginol (10%), τ-cadinol (8.2%), and α-cadinol (6.6%); the cones oil’s main constituents were abietadiene (42.5%), abietatriene (13.1%), and α-pinene (9.6%). Comparing the antiphytopathogenic fungi activities of the oils suggested that leaf oil was the most effective one. Further fractionation of the leaf oil produced ferruginol, τ-cadinol, and α-cadinol. The 3 compounds exhibited very strong antiphytopathogenic fungi activities. For the antiphytopathogenic fungi activities of the leaf oil, the active source compounds were determined to be ferruginol, τ-cadinol, and α-cadinol.
The warm and humid island-type climate of Taiwan is rather conducive to the growth of molds or mildews; hence, they are prevalent in our environments. It causes plants in nature to be attacked by microorganisms and triggers various diseases. There are many types of microorganisms that can cause plant diseases, such as fungi, bacteria, viruses, nematodes, and among all pathogens, fungi cause the most types of diseases. 1 Plant pathogenic fungi can infect plant tissues, destroy important organs such as roots or leaves, reduce the vitality of plants, and eventually cause the death of plants. 2 -5 Increasing recognition of the importance of plant pathogenic fungi infections and the difficulties encountered in their treatment have stimulated the search for alternatives to synthetic chemical fungicides. However, these chemicals may cause secondary environmental pollutions. 6,7 In recent years, scientists began looking at plant essential oils or naturally produced chemicals as fungicides. Our research team has proven on numerous occasions the antifungal efficacies of various essential oils. 8,9
Cunninghamia lanceolata (Lamb.) Hook. (Taxodiaceae) is a traditional Chinese medicine used for the treatment of hernia, arthritis, and strangury. It is a very important tree species of silviculture in Taiwan. 10 Previous studies have reported the composition and antifungal activities of wood of C. lanceolata. 11 -17 However, only 1 report noted the composition of leaf essential oil, but this report did not evaluate the biological activity. 11 No prior study has investigated the chemical composition and biological activity of the cones essential oil. Therefore, we used hydrodistillation to collect the leaf and cones oils, and analyzed these using GC-FID and GC-MS. The second part of the study examined the antiphytopathogenic fungi activity of the essential oils and dominant constituents isolated from C. lanceolata against plant pathogenic fungi. The purpose of this study was to establish a chemical basis for the effective multipurpose utilization of the species.
Results and Discussion
Based on the dry weight of leaves and cones, hydrodistillation of C. lanceolata produced yellow colored oils with yields of 1.31 ± 0.06 and 0.98 ± 0.05 mL/100 g, respectively. All compounds are listed in order of their elution from the DB-5 column (Table 1). A total of 67 compounds were identified from the hydrodistilled leaf oil of C. lanceolata. Among the leaf oil compounds, sesquiterpene hydrocarbons were predominant (33.6%), followed by monoterpene hydrocarbons (23.1%), oxygenated sesquiterpenes (20.3%), oxygenated diterpene (12.4%), diterpene hydrocarbon (3.9%), oxygenated monoterpenes (3.8%), and others (2.0%). Among the sesquiterpene hydrocarbons, β-selinene (5.1%), germacrene D (5.0%), and α-selinene (4.8%) were the major compounds. Of the monoterpene hydrocarbons, α-pinene (10.1%) and limonene (4.6%) were the main components. Monoterpene hydrocarbons τ-cadinol (8.2%) and α-cadinol (6.6%) were the major compounds; of the oxygenated diterpene, ferruginol (10%) was the chief compound. After a thorough search, we found only 1 report pertaining to the leaf oil of C. lanceolata. 11 In the report, the main ingredients of the leaf oil were α-limonene (27.3%), α-pinene (18.5%), β-caryophyllene (9.6%), and β-myrcene (9.6%), which were different from our present results.
Chemical Composition of the Leaf and Cones Essential Oils of Cunninghamia lanceolata.

bKováts retention indices, experimental: n-alkanes (C9-C24) were used as reference points in the calculation of relative retention indices.
cMS, NIST and Wiley library spectra, and the literature; KI, Kováts index; ST, authentic standard compounds.
dNot detected.
Forty-seven components were identified from the cones oil. Among the component groups, diterpene hydrocarbons were the most dominant ones (62.9%), followed by monoterpene hydrocarbons (15.2%), sesquiterpene hydrocarbons (8.4%), oxygenated sesquiterpenes (5.8%), oxygenated diterpenes (4.3%), and oxygenated monoterpenes (2.2%). Abietadiene (42.5%) and abietatriene (13.1%) were the major compounds of the diterpene hydrocarbons. Of the monoterpene hydrocarbons, α-pinene (9.6%) was the chief compound. The search turns up no report on the cones oil of C. lanceolata, however. Thus, the cones oil composition on C. lanceolata represents the first such report in the literature.
The leaf and cones oils of C. lanceolata were tested against 6 plant pathogenic fungi, including 2 seedling pathogens, Fusarium oxysporum and Rhizoctonia solani; 2 leaf pathogens, Pestalotiopsis funerea and Colletotrichum gloeosporioides; and 2 root and stem pathogens, Ganoderma australe and Fusarium solani. Figure 1 shows the antifungal indices of leaf and cones parts of C. lanceolata oil (at a concentration of 500 µg/mL) against plant pathogenic fungi. The antifungal indices demonstrated clearly that the leaf oil had antifungal activities superior to those of the cones oil (Figure 1). At this concentration, the antifungal indices of the leaf oil against damping-off pathogens, F. oxysporum and R. solani, were 72.3% and 100%, respectively. As for the leaf pathogens, P. funerea and C. gloeosporioides, the antifungal indices of the leaf oil were 67.3% and 90%, respectively. The antifungal indices of the oil against root rot pathogens, G. australe and F. solani, were 100% and 100%, respectively. The results obtained showed that the leaf oil had excellent antifungal activities with values >67.3%, and suppressed totally the growth of R. solani, G. australe, and F. solani.

Antiphytopathogenic fungi activities of the leaf and cones oils (500 µg/mL) from Cunninghamia lanceolata against the plant pathogenic fungi.
In order to evaluate the half inhibition concentrations (IC50) of the leaf and cones oils, the antifungal indices of 2 oils vs fungi were regression analyzed. The results are shown in Table 2. As the table shows, the IC50 of leaf oil against 6 plant pathogenic fungi was between 83.2 and 308.9 µg/mL; that of cones oil was >500 µg/mL. Summarizing the above, the antifungal tests indicate that leaf essential oil of C. lanceolata has the strongest antiphytopathogenic bioactivity. Thus, leaf oil is singled out for further examination of its ingredients.
IC50 and minimum inhibitory concentration (MIC) Values (µg/mL) of Leaf and Cones Oils From Cunninghamia lanceolata Against the Plant Pathogenic Fungi.

To further understand the antiphytopathogenic fungi activities’ ingredients of the oil extracted from the leaf of C. lanceolata, we have conducted a silica gel column chromatography of the leaf essential oil with the eluents mixed by n-hexane (n-hex) and ethyl acetate (EA) in different proportions (n-hex/EA = 100/0-0/100). The eluents were separated into 7 fractions (CO1-CO7). Then, we carried out experiment using 7 fractions under antifungal tests and used Nystatin as a control at a concentration of 100 µg/mL. The results are shown in Figure 2. CO4 had the best antiphytopathogenic activity with the highest antifungal indices against 6 plant pathogenic fungi. Among the plant pathogenic fungi, R. solani, P. funerea, C. gloeosporioides, G. australe, and F. solani were completely inhibited by the fraction.

Antiphytopathogenic fungi activities of CO1-CO7 fractions (100 µg/mL) against 6 plant pathogenic fungi.
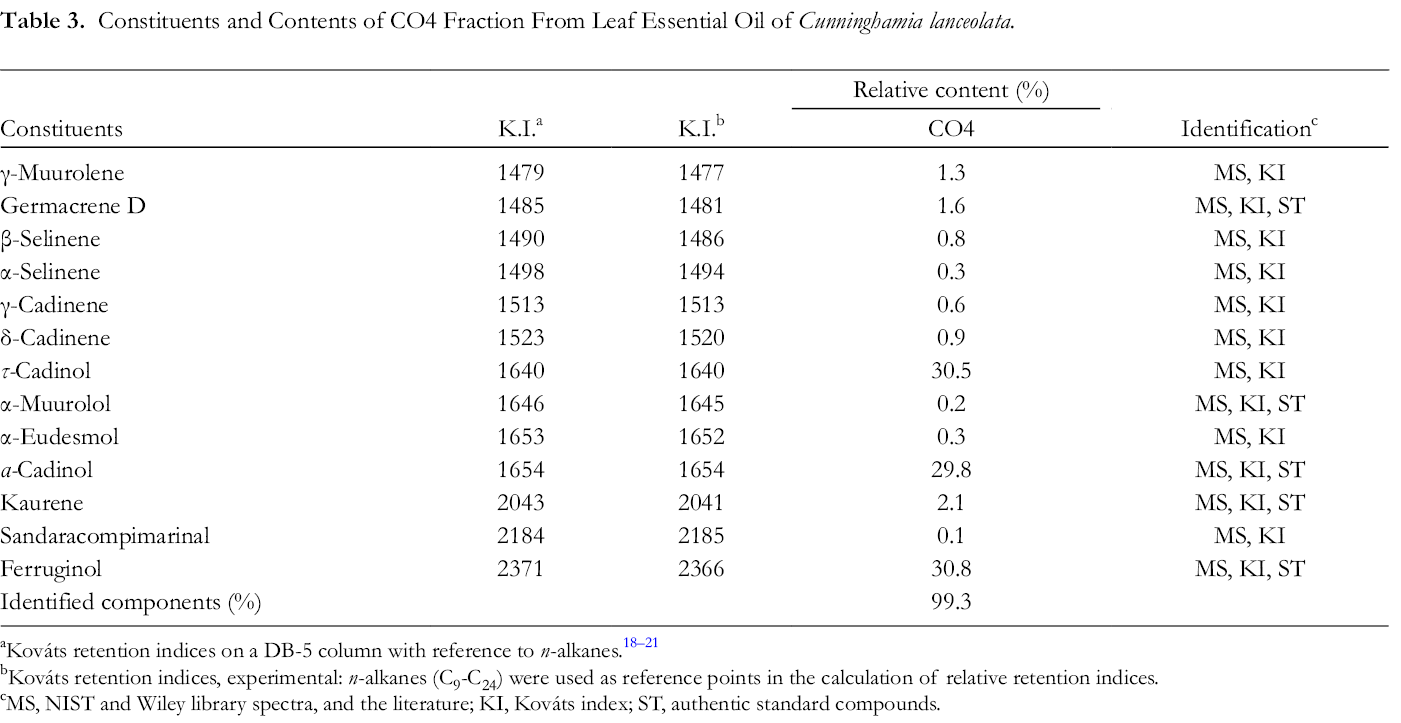
The abovementioned results have proven that CO4 exerted the best antiphytopathogenic fungi activity. We further identified the ingredients from the CO4 fraction using GC and GC-MS. The results are shown in Table 3. Thirteen compounds were identified from CO4 fraction; τ-cadinol (30.5%), α-cadinol (29.8%), and ferruginol (30.8%) were the main ones.
Constituents and Contents of CO4 Fraction From Leaf Essential Oil of Cunninghamia lanceolata.
bKováts retention indices, experimental: n-alkanes (C9-C24) were used as reference points in the calculation of relative retention indices.
cMS, NIST and Wiley library spectra, and the literature; KI, Kováts index; ST, authentic standard compounds.
Furthermore, we used the isolated and purified 3 compounds (τ-cadinol, α-cadinol, and ferruginol) to conduct antifungal tests against 6 plant pathogenic fungi, respectively. Figure 3 shows the antifungal indices of the main compounds (100 µg/mL) against 6 plant pathogens. The results indicated that 3 compounds exhibited excellent activity against F. oxysporum, R. solani, P. funerea, C. gloeosporioides, G. australe, and F. solani with the highest antifungal indices ranging from 61% to 100%.

Antiphytopathogenic fungi activities of 3 compounds (100 µg/mL) from Cunninghamia lanceolata leaf oil against 6 plant pathogenic fungi.
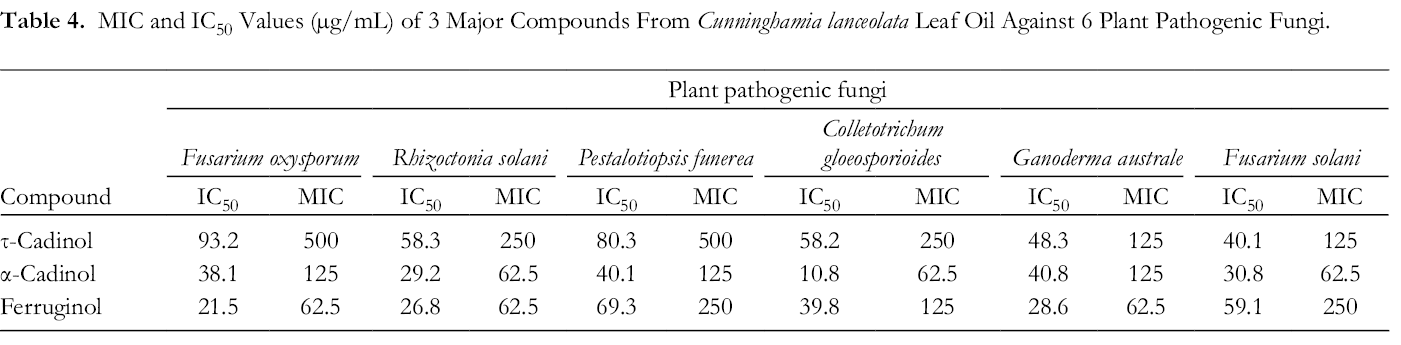
In addition, the IC50 values of 3 compounds are shown in Table 4. IC50 values of τ-cadinol against F. oxysporum, R. solani, P. funerea, C. gloeosporioides, G. australe, and F. solani were 93.2, 58.3, 80.3, 58.2, 48.3, and 40.1 µg/mL. For α-cadinol, the following IC50 values were obtained against 6 plant pathogenic fungi: 38.1, 29.2, 40.1, 10.8, 40.8, and 30.8 µg/mL. For ferruginol, the following IC50 values were obtained against 6 plant pathogenic fungi: 21.5, 26.8, 69.3, 39.8, 28.6, and 59.1 µg/mL (Table 4). Previous study have showed that these compounds are highly active in suppressing plant pathogenic fungi growth. 22 -25 Results suggest that τ-cadinol, α-cadinol, and ferruginol of C. lanceolata leaf oil could be used as potential natural fungicide for controlling fungal pathogens and worth further investigation.
MIC and IC50 Values (µg/mL) of 3 Major Compounds From Cunninghamia lanceolata Leaf Oil Against 6 Plant Pathogenic Fungi.
Experimental
Plant Materials
Fresh leaves and cones of C. lanceolata were collected in January 2018 from Guanyin Mt in central Taiwan (Taichung County, elevation 600 m, N 24°28′69″, 120°90′51″). The samples were compared with specimen no. ou-9886 from the herbarium of National Chung-Hsing University (NCHU) and were positively identified by Prof. Yen-Hsueh Tseng of NCHU. The voucher specimen (CLH-068) was deposited in the NCHU herbarium. Leaves and cones of the species were collected for subsequent extraction and analysis.
Isolation of the Leaf and Cones Essential Oil
Leaves and cones of C. lanceolata (1 kg) were distilled for 3 hours using a Clevenger-type apparatus and hydrodistillation technique. The essential oil obtained was dried with anhydrous sodium sulfate. The oil yields and all test data are the average of triplicate analyses.
Essential Oil Analysis
A Hewlett-Packard (HP) 6890 gas chromatograph equipped with a DB-5 fused silica capillary column (30 m × 0.25 mm × 0.25 μm film thickness, J&W Scientific) and a FID detector was used for quantitative determination of the oil components. The percentage composition of the oil was computed from the GC peak areas without any corrections. Oven temperature was programmed as follows: 50 °C for 2 minutes, rising to 250 °C at 5 °C/min; injector temperature: 270 °C; carrier gas: helium with a flow rate of 1 mL/min; detector temperature: 250 °C, split ratio: 1:10. Diluted samples (1.0 µL, 1/100, v/v, in EA) were injected manually in the split mode. Identification of the oil components was based on their Kováts indices and mass spectra obtained from GC-MS analysis on a HP 6890/HP 5973 equipped with a DB-5 fused silica capillary column (30 m × 0.25 mm × 0.25 μm film thickness, J&W Scientific). The GC analysis parameters are listed above and the MS were obtained (full scan mode: scan time was 0.3 seconds and mass range was m/z 30-500) in the EI mode at 70 eV. All data were the average of triplicate analyses.
Component Identification
Identification of the essential oil constituents from the leaf and cones was based on comparisons of the Kováts retention indices, 18 and mass spectra with those obtained from authentic standards and/or the NIST and Wiley libraries spectra, and the literature. 18 -21
Isolation and Purification of Leaf Oil Components
The leaf essential oil (20 g) mixed with silica gel (60 g) (Merck 7734, Merck Co., Germany) was chromatographed on a silica gel open column (600 g) and eluted with a stepped gradient consisting of n-hex and EA (ranging from n-hex/EA = 100:0-0:100). The samples collected were screened by thin-layer chromatography (TLC) (Silica gel 60F254, Merck Co., Germany) profile, and fractions having similar TLC patterns were combined to give 7 fractions (CO1-CO7). The yield of each fraction was 0.3% (CO1), 6.8% (CO2), 15.3% (CO3), 45.3% (CO4), 15.3% (CO5), 11.2% (CO6), and 3.1% (CO7).
According to the antiphytopathogenic fungi activity assay, the CO4 exerted the best antiphytopathogenic fungi activity. Ferruginol, τ-cadinol, and α-cadinol were isolated and purified from CO4 fraction by semipreparative HPLC (column: Si-60 column, mobile phase: EtOAc/n-C6H14 = 30/70, flow rate: 1 mL/min). Ferruginol (retention time (RT): 14.3 minutes), τ-cadinol (RT: 22.1 minutes), and α-cadinol (RT: 31.2 minutes) were separately obtained. The structures of 3 compounds were confirmed by comparing physical and spectral data (including 1H-NMR, 13C-NMR, and EI-MS) with the previously reported values. 26,27 Ferruginol: Yellow oil, EI-MS for C20H30O (EI-MS: 286), 1H NMR (in CDCl3): δ (ppm) 0.88 (3H, s, H-18), 0.91 (3H, s, H-19), 1.16 (3H, s, H-20), 1.22 (3H, d, J = 7.0 Hz, H-16), 1.30 (3H, d, J = 7.0 Hz, H-17), 2.76 (1H, ddd, J = 17.0, 10.5, 7.0 Hz, H-7a), 2.83 (1H, ddd, J = 2.0, 6.5, 17.0 Hz, H-7b), 3.13 (sept, J = 7.0 Hz, H-15), 6.62 (1H, s, H-11), 6.83 (1H, s, H-14); 13C NMR: δ (ppm) 19.10 (C-6), 19.19 (C-2), 21.45 (C-19), 22.52 (C-16), 22.68 (C-17), 24.63 (C-20), 26.48 (C-15), 29.61 (C-7), 33.16 (C-18), 33.21 (C-4), 37.26 (C-10), 38.65 (C-1), 41.58 (C-3), 50.21 (C-5), 110.88 (C-11), 126.46 (C-14), 126.25 (C-8), 131.68 (C-13), 148.18 (C-9), 150.96 (C-12). τ-Cadinol: Yellow oil, EI-MS for C15H26O (EI-MS: 222), 1H-NMR (in CDCl3): δ (ppm) 0.75 (d, J = 7.0, H-12), 0.87 (d, J = 7.0, H-13), 1.16 (s,H-14), 1.68 (s, H-15), 2.15 (m, H-11) 5.50 (s, H-4). 13C-NMR: δ (ppm) 15.18 (C-12), 19.79 (C-7), 21.40 (C-13), 22.57 (C-1),23.76 (C-15), 26.15 (C-11), 28.44 (C-14), 30.88 (C-2), 37.71 (C-5), 40.28 (C-8), 46.64 (C-6), 47.91 (C-10), 70.69 (C-9), 122.63 (C-4), 134.35 (C-3). α-Cadinol: Colorless needle crystal, m.p. 74 to 75 °C, EI-MS for C15H26O (EI-MS: 222), 1H-NMR (in CDCl3):δ (ppm) 0.75 (d, J = 7.0, H-12), 0.88 (d, J = 7.0, H-13), 1.06 (s, H-14), 1.63 (s, H-15), 2.14 (m, H-11) 5.46 (s, H-14). 13C-NMR: δ (ppm) 15.12 (C-12), 20.76 (C-14), 21.50 (C-13), 22.00 (C-1), 22.66 (C-7), 23.80 (C-15), 26.02 (C-11), 30.96 (C-2), 39.86 (C-5), 42.21 (C-6), 46.75 (C-6), 50.06 (C-10), 72.46 (C-9), 122.33 (C-4), 134.95 (C-3).
Antiphytopathogenic Fungi Activity Assays
The plant pathogenic fungi used were F. oxysporum f. sp. melonis Snyder & Hansen (BCRC32121), R. solani Kuhn (BCRC31626), P. funerea (Desmazieres) Steyaert (BCRC35266), C. gloeosporioides Penzig (BCRC35003), Ganoderma australe (Fries) Paterson (BCRC36246), and F. solani (Martius) Saccardo (BCRC32458). Each fungal strain was cultured in potato dextrose agar (PDA, Difco Company). Microbial strains were obtained from the Bioresource Collection and Research Center (BCRC) of Taiwan.
The method of Ho et al 28 was adopted. Essential oil concentration (500 µg/mL) and different concentrations of main compounds (0-500 μg/mL) were added to sterilized PDA in 9 cm Petri dishes. After transferring the mycelium of 1 fungus strain, the testing dishes were incubated at 27 °C and 70% relative humidity. When the mycelium reached the edge of the control plate, the antifungal index was calculated as follows:
Antifungal index (%) = (1 – D a/D b) × 100
where D a is the diameter of the growth zone in the experimental dish (cm) and D b is the diameter of the growth zone in the control dish (cm).
Each test was repeated 5 times and the data were averaged. The IC50 values (the concentration in mg/mL that inhibited 50% of mycelium growth) were calculated by a probit analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We appreciate the financial support (108-2313-B-002-022 and 106-2313-B-054-003-MY3) from the Ministry of Science and Technology.
