Abstract
Pharbitidis Semen is a traditional Chinese medicine(TCM) with a long history for treatment of edema and fullness, fecal and urinary retention, phlegm and retained fluid, and abdominal pain due to parasitic infestation. Since Pharbitidis Semen is distributed throughout the country, the quality of the medicine from different origins may be varied. Moreover, the reported method could not control the quality comprehensively. In this article, a fingerprint of Pharbitidis Semen has been established based on a high-performance liquid chromatography (HPLC) method. In addition, the contents of the 2 main effective components were determined simultaneously. The reference HPLC fingerprint was obtained according to the chromatograms of test samples. The similarity values were calculated by the Similarity Evaluation System for Chromatographic Fingerprint of TCM (2004 A edition). Cluster analysis of 10 batches of samples was performed using statistical software (SPSS 20.0). The HPLC fingerprints of 10 batches of Pharbitidis Semen showed 25 well-resolved common peaks in each chromatogram. Two of these peaks were assigned to protocatechuic aldehyde and caffeic acid. As a result, HPLC fingerprint similarities of 10 batches of samples were more than 0.99. Pharbitidis Semen from different habitats could be divided into 3 or 2 groups. The results of cluster analysis showed that samples classified into 1 group were associated with their habitats and breeds. At the same time, quantification results showed that the contents of protocatechuic aldehyde and caffeic acid were in the range of 0.026-0.088 and 0.019-0.053 mg/g−1 respectively. HPLC fingerprint combined with multicomponent quantification and data analysis techniques can be an efficient and useful method for monitoring the quality of Pharbitidis Semen. This study also provides a practical strategy for overall quality evaluation and control of traditional Chinese medicines.
Pharbitidis Semen is the dry mature seed of either Pharbitis nil (L.) Choisy or Pharbitis purpurea (L.) Voigt, which has diuresis, catharsis, dissolving phlegm, and insecticidal actions. The medicine is usually used for the treatment of edema and fullness, fecal and urinary retention, phlegm and retained fluid, and abdominal pain due to parasitic infestation. 1 Pharbitidis Semen, also known as Heichou, Baichou, and Erchou, was first recorded in “Mingyi Bielu”. Modern research shows that the drug consists of various chemical components, including amino acids, glycosides, alkaloids, anthraquinones, phenolic acids, and carbohydrates. 2,3 It has many remarkable pharmacological effects, such as increasing the propulsive rate of the large intestine, promoting gastrointestinal peristalsis, bacteriostasis, stimulating the uterus, expelling parasites, anticancer, phlegm-resolving, anti-inflammatory, and improvement of immunity. 4,5
Fingerprints of traditional Chinese medicine (TCM) can be divided into chromatographic, spectral, and so on which can be used to reveal the chemical characteristics of the TCM. High-performance liquid chromatography (HPLC) and its various combination techniques are a primary means for research of fingerprints of TCM, 6 -9 especially HPLC. Application of this method in fingerprints has obvious advantages. The specificity, sensitivity, and accuracy of HPLC are higher than those of thin-layer chromatography (TLC) methods. 10 HPLC is also more widely applicable than capillary electrophoresis (CE) and less costly than liquid chromatography-mass spectrometry (LC-MS). 11 This method can reflect the whole information of a TCM and can be applied to its identification and classification. 12 Moreover, HPLC is considered as one of the quality control standards of a TCM in “Chinese pharmacopeia (2015 Edition)” 13 -15 and is widely used in the quality control of various TCMs. 16
Pharbitidis Semen can grow all over the country. The quality of the medicine is sensitive to breeds, origins, and storage conditions, which may lead to variable contents of its chemical components. Therefore, it is difficult to assess its quality, as well as authenticity. Few published studies of HPLC fingerprints of Pharbitidis Semen were found. At present, an HPLC fingerprint is one of the most effective and intuitive means for quality control of a TCM. In this article, an HPLC method for fingerprint and quantitation of 2 effective components in Pharbitidis Semen (including Heichou and Baichou) were established. The HPLC fingerprints of 10 batches of Pharbitidis Semen showed 25 well-resolved common peaks in each chromatogram. Two of these were assigned to protocatechuic aldehyde and caffeic acid. The developed method was found to be simple, reliable, accurate, and reproducible via method validation, which can provide methodological reference and a basis for quality evaluation, as well as quality control of Pharbitidis Semen. The proposed method can also be applied to identify Heichou, Baichou, and Pharbitidis Semen from different origins, which will provide quality assurance for a wider clinical application of the medicine.
To confirm the best preparation method of the test sample solution, different extraction methods (ultrasonic and reflux) 17 -19 were studied on the basis of HPLC, including the number of chromatographic peaks, and separation. Good extraction could be achieved using both ultrasonic and reflux methods, but more solvent and a longer time were required when the sample was extracted by reflux. In addition, ultrasonic extraction is faster, more accurate, more convenient, and more economical than presoaking, 20,21 liquid ammonia pretreatment, 22,23 and the co-digestion method. 24 Therefore, the ultrasonic method was adopted for the preparation process of the medicine due to easy operation and high efficiency.
Optimization of chromatographic conditions played an important part in the establishment of an HPLC fingerprint for Pharbitidis Semen. As a result, different compositions and proportions of the mobile phase and detection wavelengths were investigated. First, the detection wavelength was confirmed as 225 nm, as more chromatographic peaks and good separation could be achieved using this wavelength. Subsequently, mobile phase systems with different proportions of methanol-water, acetonitrile-water, and methanol-phosphoric acid-water were investigated using separation degree and analysis time as indexes. The results indicated that gradient elution with methanol (B)-0.1% phosphoric acid solution could achieve better separation effects with a shorter determination time.
Caffeic acid has the pharmacological effects of relaxing the bowels, resisting inflammation, preventing cough, and eliminating phlegm. 25 Protocatechuic aldehyde has anti-inflammatory as well as antibacterial effects. 26,27 The pharmacological effects above are consistent with the pesticide effects of Pharbitidis Semen. It is reported that the content of caffeic acid is also used for the evaluation of Pharbitidis Semen and its processed products. 28 Protocatechuic aldehyde is regarded as a common index component of some TCMs with anti-inflammatory and antibacterial effects. 27 At present, no research on other active components present in high contents has been reported. Some constituents with high content in Pharbitidis Semen have not been separated, identified, and determined so far due to the lack of in-depth study. The existing methods cannot completely control the quality of medicine. Thus, protocatechuic aldehyde and caffeic acid were selected as the key compounds to evaluate the quality of Pharbitidis Semen in this study after consulting the literature. The contents of these 2 components are relatively high, which can reflect the quality of Pharbitidis Semen to some degree, and can be used for the identification of the medicine.
HPLC fingerprint is an effective quality evaluation method and, combined with multicomponent quantification, is more efficient than the previous quality control method. The aim of quality control cannot be achieved by way of either HPLC fingerprint or multicomponent quantification alone because the fingerprint can reflect the differences in the whole chemical composition of the medicine, while the content of the main active ingredients has a crucial influence on the quality of the drug. Therefore, the combination of these 2 methods can evaluate the quality of Pharbitidis Semen comprehensively, which can provide a research foundation for identification, quality control, purchase, storage, and processing of the medicine.
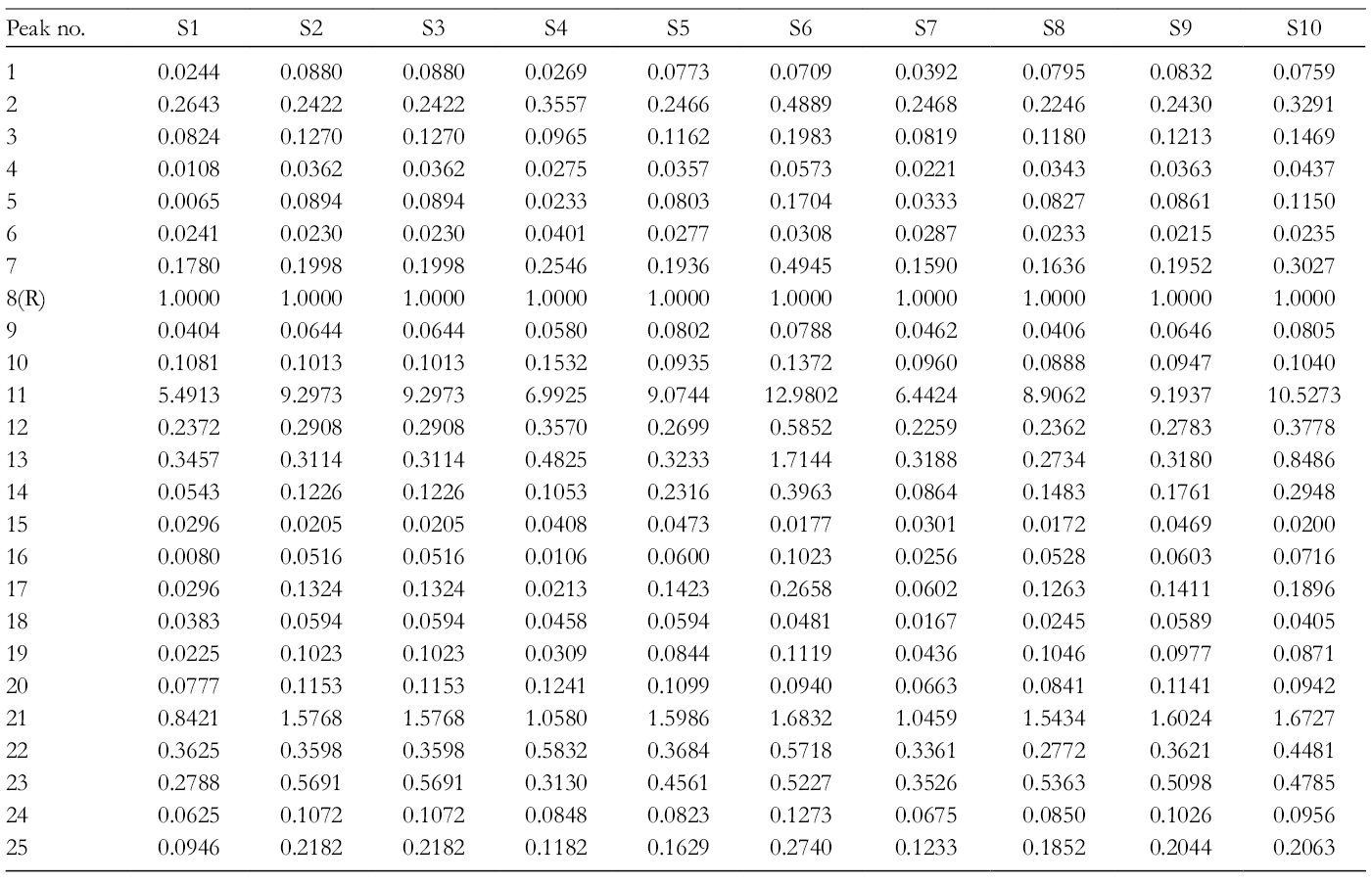
The results of software analysis are displayed in Figure 1. Twenty-five well-resolved common peaks were marked in each chromatogram (Figure 2). Two characteristic peaks were identified and attributed to protocatechuic aldehyde (peak 8) and caffeic acid (peak 13) by comparing with reference standards. Protocatechuic aldehyde was considered as the reference (R) peak due to its good separation, table peak area, and good peak shape. Relative retention times (RRTs) and relative peak areas (RPAs) of the common peaks were calculated for method validation. The results are shown in Tables 1 and 2.

High-performance liquid chromatography fingerprints of 10 batches of samples.

Common peaks of high-performance liquid chromatogram.
Relative Retention Times of Common Peaks in High-Performance Liquid Chromatograms of 10 Batches of Samples.

Relative Peak Areas of Common Peaks of High-Performance Liquid Chromatograms of 10 Batches of Samples.
The data of 10 batches of HPLC fingerprints were imported into Similarity Evaluation System for Chromatographic Fingerprint of TCM (2004 A edition) for analysis. During the analysis, the multipoint correction and chromatographic peak matching method were used to calculate the similarity of each sample (Table 3). As a result, the similarities of all samples were more than 0.991, indicating that there was little difference between these samples. The quality of 10 batches of samples was relatively stable.
Results of Similarity of 10 Batches of Samples.

The RPA data of 10 batches of Pharbitidis Semen were analyzed using statistical software (SPSS 20.0). In addition, cluster analysis (CA) was determined based on the Ward method of Euclidean Distance Hierarchical Clustering. The CA results are shown in Figure 3. Ten batches of samples were categorized into 3 groups containing 6 samples (Group 1, S2, S3, S5, S9, S8, and S7), 2 samples (Group 2, S6, and S10) and 2 samples (Group 3, S1, and S4) when the Euclidean distance was set as 5. However, when the Euclidean distance was set as 15, the 10 batches of samples were categorized into 2 groups containing 8 samples (Group 1, S2, S3, S5, S9, S8, S7, S6, and S10) and 2 samples (Group 2, S1, and S4). The results of cluster analysis indicated that Pharbitidis Semen from different places can be divided into 2 groups (Heichou and Baichou). This may be due to the different contents of chemical components in these 2 groups. Therefore, cluster analysis can be used for the identification and classification of Pharbitidis Semen under some conditions without appearance identification. Moreover, this method can lay a foundation for research on the pharmacodynamic difference and purchase of Heichou and Baichou.
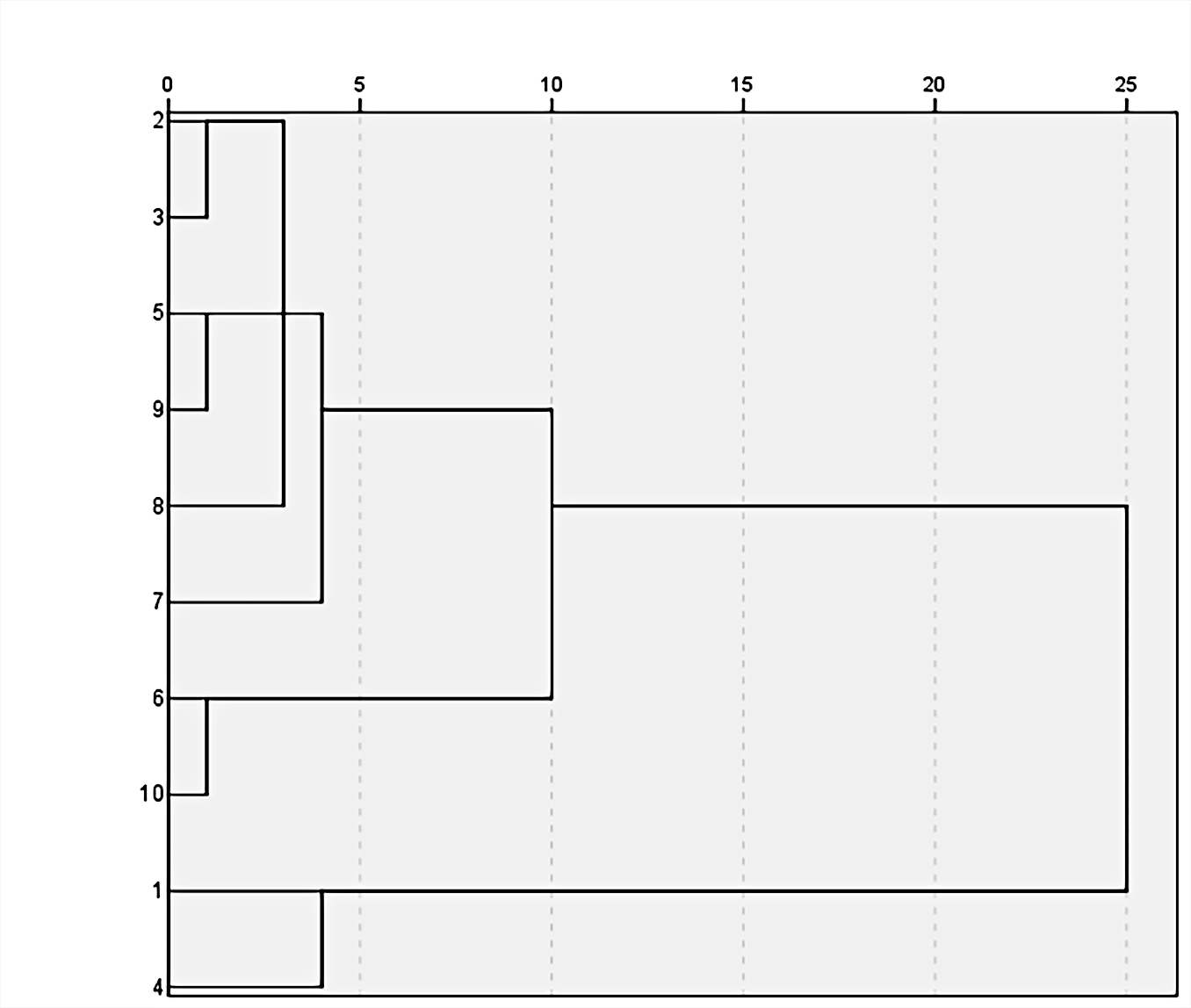
Diagram of cluster analysis.
The established HPLC-diode-array detector method was applied to the simultaneous determination of protocatechuic aldehyde and caffeic acid in Pharbitidis Semen. The results are summarized in Table 4. The contents of the 2 components were in the range of 0.026-0.088 mg/g and 0.019-0.053 mg/g, respectively. The contents of protocatechuic aldehyde and caffeic acid in Baichou were higher than those in Heichou. In addition, the content of the 2 components in Baichou from Henan Province was higher than that from Anhui Province. The content of protocatechuic aldehyde in Heichou from Hebei Province was the lowest compared with that from the other province, while the content of caffeic acid in Heichou from Hebei and Yunnan Province was higher than that from Anhui and Henan Province.
Results of Multicomponent Quantification (mg/g, N = 2).

The reproducibility test results of the HPLC fingerprint showed that relative standard deviations (RSDs) of RRT and RPA of each chromatographic peak were less than 2.47% and 2.63%, indicating that this method had good reproducibility. The repeatability test showed that RSDs of RRT and RPA of each chromatographic peak were less than 2.40% and 2.95%, illustrating that this method was of good repeatability. The stability RSD results exhibited that the RRT and RPA of each chromatographic peak were less than 1.99% and 2.07%, suggesting that the test solution was stable within 24 hours.
The regression equations of protocatechuic aldehyde and caffeic acid were y = 21 616x + 104.6 (r = 0.9995) and y = 12 810x + 73.986 (r = 0.9996), respectively, suggesting that the 2 analytes presented good linear relationships within the ranges of 0.018-0.881 mg/mL and 0.011-0.532 mg/mL under the proposed chromatographic conditions. The reproducibility RSD results of peak areas of protocatechuic aldehyde and caffeic acid were 1.37% and 1.26%, respectively, which indicated that this method had good reproducibility. The RSD results of contents of protocatechuic aldehyde and caffeic acid were 1.83% and 1.76%, respectively, illustrating that this method was of good repeatability. The stability RSD results of area peaks of protocatechuic aldehyde and caffeic acid were 1.65% and 1.88%, respectively, suggesting that the test solution was stable within 24 hours. The results above are summarized in Table 5. The contents of the 2 constituents in the recovery test were determined and the recovery rates were calculated. The average recoveries (Table 6) of the 2 components were 99.81% and 100.13% with RSDs of 1.91% and 1.99%, respectively. The recovery results illustrated that the proposed method had good accuracy.
Results of Validation Test.

RSD, relative standard deviation.
Results of Recovery Test.

RSD, relative standard deviation.
Experimental
Chemicals and Reagents
Protocatechuic aldehyde (batch No. 110810–201608) and caffeic acid (batch No. 110885–200102) were purchased from China Pharmaceutical Biological Products Verification Institute (Beijing, China). Methanol (Fisher, USA) was of chromatographic grade. Phosphoric acid and other reagents (Beijing Chemical Industry Factory) were all of the analytical grade. Ultrapure water was purchased from Hangzhou Wahaha Co., Ltd.
Materials
Ten batches of Pharbitidis Semen samples were purchased from different origins of China. Detailed information is displayed in Table 7.
Origins of Pharbitidis Semen Samples.

Instrumentation
Chromatographic separation was achieved on an Agilent 1260 HPLC system (including quaternary low pressure mixing pump, autosampler, column oven, diode array detector, and Chemstation workstation). An AB135-S electronic balance was obtained from Mettler Toledo International Co., Ltd. KQ-250, an ultrasonic cleaner from Kunshan Ultrasonic Instrument Co., Ltd., and an R series rotatory evaporator from Shanghai ShenSheng Technology Co., Ltd.
Preparation of Standard Solutions
Appropriate amounts of protocatechuic aldehyde and caffeic acid were weighed precisely and dissolved in methanol to obtain stock standard solutions with concentrations of 0.881 mg/mL and 0.532 mg/mL, respectively. These standard solutions were stored at 4°C. One milliliter stock standard solution was placed into a 10 mL volumetric flask and diluted with methanol (final adjusted volume 10 mL) to obtain a mixed standard solution with a concentration of protocatechuic aldehyde of 88.1 µg/mL and caffeic acid of 53.2 µg/mL. The solution above was shaken well and filtered through a 0.22 µm membrane filter before HPLC analysis.
Preparation of Test Solutions
About 5.0 g of comminuted Pharbitidis Semen was weighed accurately and placed in a stoppered conical flask. Then 40 mL methanol was added to the flask and ultrasonically (250 W, 40 kHz) extracted for 30 minutes. The methanol extract was filtered after cooling. The sample was extracted twice, and the extracts were combined and evaporated to dryness using a roller evaporator at 60°C. Finally, the residue was redissolved in methanol (final adjusted volume 5 mL) and the solution filtered through a 0.22 µm microporous membrane filter after shaking, and the subsequent filtrate was collected as the test solution.
Chromatographic Conditions
The analysis was achieved on an Agilent ZORBAX SB-C18 column (250 mm× 4.6 mm, 5 µm) with a column temperature of 30°C. The mobile phase consisted of methanol (A) and 0.1% phosphoric acid solution (B). The detection wavelength was 225 nm. The flow rate was maintained at 1.0 mL/min. The injection volume of the sample was 10 µL. The gradient elution conditions were as follows: 0-5 minutes, 2%→15%A; 5-13 minutes, 15%→20%A; 13-23 minutes, 20%→20%A; 23-28 minutes, 20%→30%A; 28-30 minutes, 30%→30%A; 30-37 minutes, 30%→35%A; 37-60 minutes, 35%→45%A; 60-75 minutes, 45%→90%A; 75-80 minutes, 90%→97%A; 80-90 minutes, 97%→97%A; and 90-95 minutes, 97%→100%A.
Establishment of Fingerprint and Multicomponent Quantification
The standard solutions and 10 batches of Pharbitidis Semen solutions were injected into the HPLC to obtain their chromatograms and peak areas, followed by calculation of contents of 2 analytes via an external standard method. Then the recorded chromatograms were imported to the Similarity Evaluation System for Chromatographic Fingerprint of TCM (2004 A edition) for analysis.
Method Validation
The details of this section can be found in the Supplementary Material.
Supplemental Material
Supplementary Material - Supplemental material for HPLC Fingerprint Combined With Multicomponent Quantification as an Efficient Method for Quality Evaluation of Pharbitidis Semen
Supplemental material, Supplementary Material, for HPLC Fingerprint Combined With Multicomponent Quantification as an Efficient Method for Quality Evaluation of Pharbitidis Semen by Huiwei Bao, Huailei Yang, Feng Wang, Kaixuan Zhou, Yanan Yang, Yang Xu and Lijing Li in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to Changchun Medical College, Changchun University of Chinese Medicine, Changchun Hospital of Traditional Chinese Medicine, Jilin University, Baicheng Medical College, and Chinese Institute of Jilin Ginseng for providing equipment and funding supports.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
