Abstract
The availability of a simple method for the determination of the quality of
Results and Discussion
Qualitative and Quantitative Analyses
To evaluate the phytochemical profile of the 3 different commercial 50% hydroalcoholic extracts of

High resolution liquid chromatography-electrospray ionization-mass spectrometry total ion current chromatograms of the AMG1, AMG2, AMG3 , and
In the negative ion mode mass spectrum of compounds
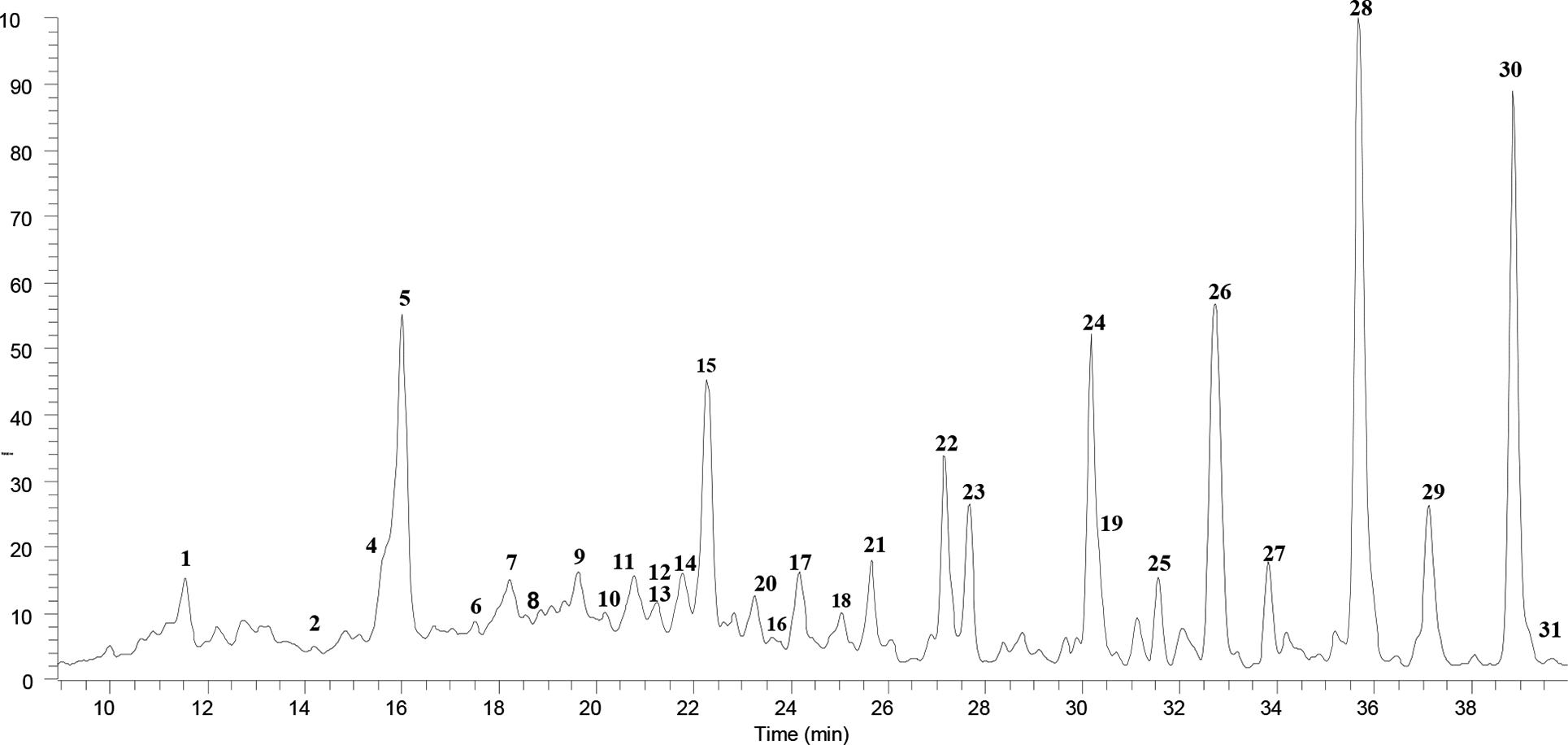
High-performance liquid chromatography-high resolution electrospray ionization-mass spectrometry profile of the polyphenol derivatives and astragalosides in
Chromatographic and MS Data of Compounds 1-
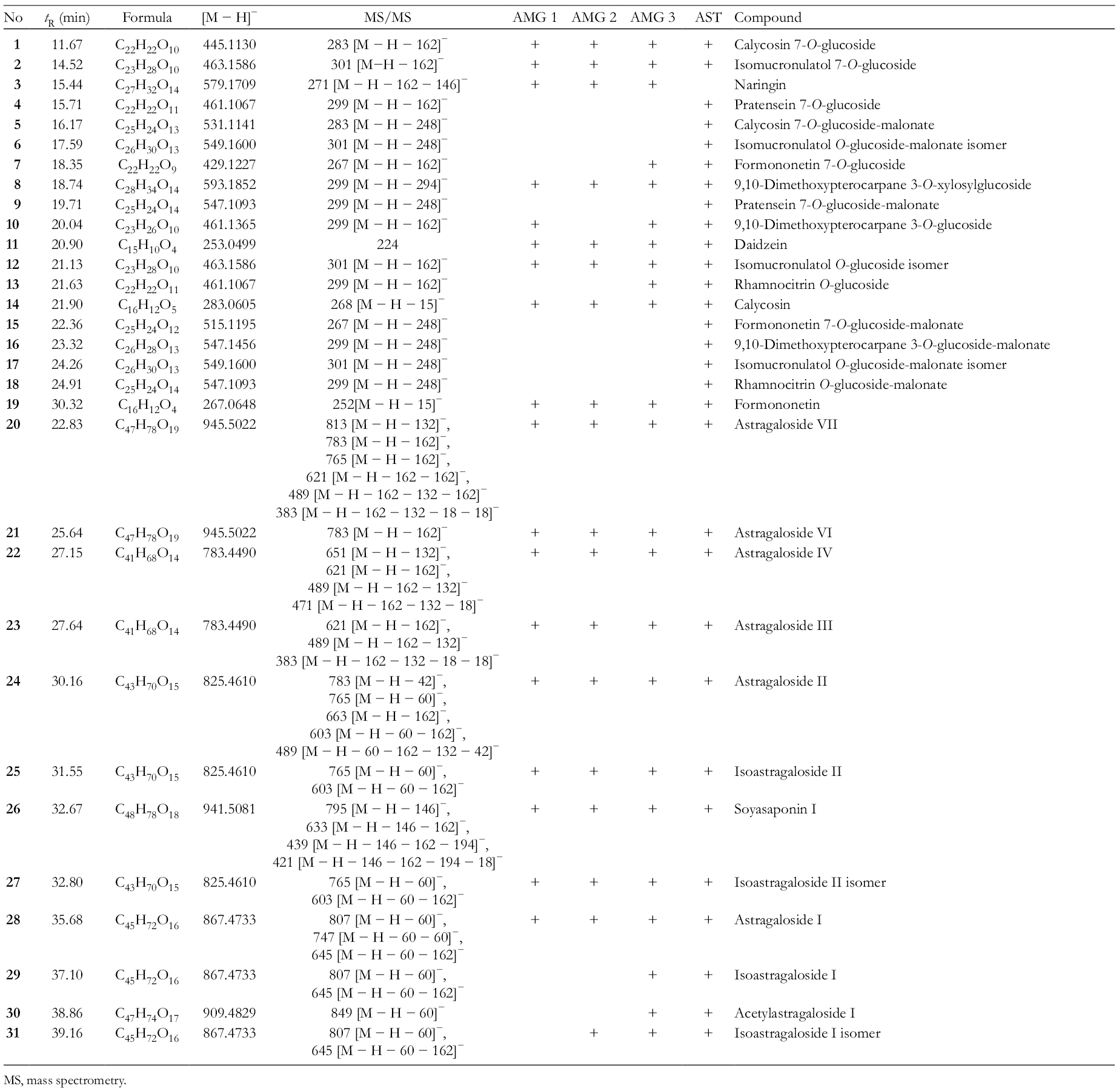
MS, mass spectrometry.
Compounds
Successively, quantitative analysis of the flavonoids and astragalosides fraction was performed in all samples. The flavonoids were quantified as formononetin, whereas astragaloside IV was used to express the saponins amount. In Table 2, the composition of total flavonoids and astragalosides as a percentage of 100 g of extracts is reported. Results of the quantitative analysis indicated that in the AMG3 (Axtragyl), saponins are more abundant compared with the other samples, whereas the flavonoids are the most represented in AST sample compared with the other three , although the flavonoids content in AMG3 was higher than in AMG1 and AMG2. Astragaloside I and astragaloside IV, being the most representative among the saponins, were selected and quantified in all samples as a marker of quality of the commercial batches. In order to obtain accurate data regarding their amounts, a selective and sensitive ultra-performance liquid chromatography-ESI-QTrap-MS/MS method was developed after the direct introduction of the standard to optimize the signal of the analyte. Data were acquired in multiple-reaction monitoring (MRM) mode in triplicate, and the results of quantitative analysis of astragaloside I and astragaloside IV in all the samples are the following: 0.0004 and 0.04 mg/g for AMG1, 0.0003 and 0.011 mg/g for AMG2, 0.24 and 0.84 mg/g for AMG3, and 0.16 and 0.13 mg/g for AST.
Content in Percentage (%) of Flavonoids and Astragalosides in 100 g of AMG1, AMG2, AMG3, and

α-Glucosidase and α-Amylase Inhibition Activity
Since
AMG3 and AMG1 demonstrated the highest inhibitory activity on α-amylase among tested extracts (half-maximal inhibitory concentration [IC50] 495.6 ± 23.8 µg/mL and 679.3 ± 22.1 µg/mL, respectively) but, as expected, lower than acarbose (IC50 3.5 ± 0.2 µM) (Table 3). No activity was observed in the α-glucosidase assay for all the extracts. Among the pure compounds, the astragalosides were inactive (data not shown), while the isoflavone aglycones showed only a mild activity against α
α-Amylase and α-Glucosidase Inhibition of AMG1, AMG2, AMG3,

IC50, half-maximal inhibitory concentration.
*In this case, it was not possible to reach the IC50 value; the results were expressed as % α-amylase inhibition obtained testing extract at 50 µg/mL.
Acarbose IC50 9.59 ± 0.90 and 350.3 ± 12.6 µg/mL vs α-amylase and α-glucosidase respectively. Significant differences (
Molecular Docking of Daidzein and Formonoetin Into Human Small Intestine α-Glucosidase
To get further insight into the inhibitory effects of compounds

Docking of daidzein (11) and formononetin (19) in the
Cholinesterase Inhibition Activity
According to the published studies on the protective role of
In the present study, the inhibition of AChE and butyrylcholinesterase (BChE) enzymes was evaluated for all extracts and pure compounds (Table 4). AMG3 reported the highest AChE inhibition (IC50 27.9 ± 5.1 µg/mL) at tested concentrations. All extracts showed a similar value of inhibition vs BChE (Table 4). On the contrary, pure compounds showed a low value of inhibition of AchE activity and no activity in the BChE inhibition assay (data not shown).
AChE and BChE Inhibition of AMG1, AMG2, AMG3, and

AChE, acetylcholinesterase; BChE, butyrylcholinesterase; IC50, half-maximal inhibitory concentration.
Percentage of AChE and BChE inhibition obtained testing extracts at 50 µg/mL and of pure compounds at 5 µM; galantamine IC50 = 0.2 ± 0.0 and 6.9 ± 0.4 µg/mL vs AChE and BChE, respectively. Significant differences (
Antioxidant Activity
Several studies reported a protective effect of
Effect of Cytotoxicity in Caco-2 Cells of AMG1, AMG2, AMG3, and

Values are means ± standard deviation;
Significant differences (
Conclusions
Herbal extracts should be standardized to ensure safety, quality, and efficacy. In our study, LC-MS/MS-based approach was used to characterize specialized metabolite recovery in 3 different commercial samples of
Experimental
Reagents
Solvents for extraction were purchased from Sigma Chemicals Company (Milan, Italy). LC–MS grade solvents were purchased by Romil Ltd Pure Chemistry (Cambridge, GB). For the quantitative HPLC analysis, the following standards were used: astragaloside IV, European Pharmacopeia (EP) reference standard (97.8%), astragaloside I, Phyproof reference substance ≥98.0% (HPLC), and formononetin, analytical standard (≥98% by HPLC) from (Sigma-Aldrich, Milano, Italy). Dulbecco’s modified Eagle’s medium (DMEM), Eagle’s minimum essential medium (EMEM), and fetal calf serum (FCS), penicillin–streptomycin, from porcine pancreas and phosphate-buffered saline (PBS) tablets were purchased from Euroclone.
Plant Material
The roots of
Extraction and Sample Preparation
The dried roots of
LC–MS Qualitative Analysis
The separation system adopted was an Accela (Thermo Fisher Scientific, Milan, Italy) HPLC interfaced through an ESI source to a linear ion trap coupled to a high-resolution mass analyzer (LTQ-Orbitrap XL, Thermo Fisher Scientific, Milan, Italy). The MS data were acquired, in negative ion mode, at first in full-mass and data dependent-scan mode, then, tandem MS experiments were done in order to identify the specialized metabolites. Capillary temperature was set at 350°C, flow rate of sheath gas and auxiliary gas were set at 30.0 and 10 arbitrary units, capillary voltage was −48.0 V. A C18 column (Luna C18, Phenomenex, 100 × 2.0 mm, 2.5 µm) and a binary mobile phase composed of eluent A (ultrapure water–formic acid 0.1% v/v) and eluent B (ultrapure acetonitrile–formic acid 0.1% v/v) were used. The separation conditions are from 10% to 95% of B in 60 minutes. Flow rate was 0.0200 mL/min and the injection volume 10.0 µL.
LC–MS Quantitative Analysis
Quantification of astragalosides I and IV was carried out using an API6500 Q-Trap (ABSciex Foster City, CA, USA) coupled with an A NexeraX2 UHPLC apparatus (Shimadzu, USA), working in negative MRM mode. All the instrumental parameters were optimized directly injecting solutions containing pure compounds. Samples were loaded on a Kinetex column (Phenomenex) (C18 100 A, 50 mm × 2.6 µm × 2.1 mm), and compounds were separated using a linear gradient from 30% to 55% of acetonitrile (eluent B) and water containing 0.1% formic acid (eluent A) over 10 minutes. The flow rate was 0.35 mL/minute, and the injection volume was 3 µL for standards and samples. To perform accurate quantitative analyses, 9 points (in the range 0.10-2 µg /mL) calibration curves were built for the two astragalosides. The mean values ± standard deviation from at least three experiments showing similar results were reported.
α-Amylase Inhibition Assay
The α-amylase inhibition assay was performed using the iodine/potassium iodide method.
27
Each sample (25 µL) was mixed with the α
α-Glucosidase Inhibition Assay
Different concentration of each sample was incubated with the α
In Silico Molecular Docking
Molecular docking of conformationally flexible
AChE and BChE Inhibition Assay
The inhibition of AChE was determined based on Ellman’s method, as reported by Faraone et al. 31 In this assay, 25 µL of 0.05 U/mL AChE, 125 µL of DTNB (3 mM), 25 µL of buffer B (50 mM Tris–HCl, pH 8 containing 0.1% BSA), and 50 µL of the sample at different concentrations were incubated for 10 minutes. Then, 25 µL of acetylthiocholine iodide (5 mM) was added, and the absorbance was measured at 405 nm after 10 minutes. The BChE inhibition assay was performed in a similar way using 25 µL of butyrylthiocholine chloride (5 mM) as substrate and 0.05 U/mL of BChE as enzyme. Three independent assays were performed in triplicate at different concentrations. Results are expressed as % of enzyme inhibition measured at certain concentrations.
Cell Cultures
PBMC cells were isolated from buffy coats of healthy donors and cell viability evaluated as reported before.
32
Caco-2 cells were maintained in DMEM, containing 200 mL/L FCS, 10 mL/L of 100× nonessential amino acids, 2 mmol/L
Induction of Oxidative Stress and Neutral Red Assay
An iron-free medium (EMEM) was used for the oxidative stress induction experiments. The oxidative stress was induced in the apical compartment of the transwell insert by the addition of H2O2. To assay the capacity of AMG1, AMG2, AMG3, and AST to protect Caco-2 cells from ROM-mediated oxidative injury, cells were preincubated for 4 hours with extracts that were added to the apical side of monolayer. After the end of the preincubation time, the medium was changed before the addition of the oxidative stress-inducing agents. The cytotoxicity of ROM on Caco-2 was assessed by the viability test of neutral red uptake, performed according to the procedure of Fautz et al. 34 After oxidative stress induction, the medium in the insert was removed and replaced with 0.1 mL of fresh medium containing 1.14 mmol/L neutral red. At the end of 3 hours of incubation, the medium was removed and cells were washed twice with PBS; finally, the incorporated neutral red was released from cells by incubation for 15 minutes at room temperature in the presence of 1 mL of cell lysis buffer containing acetic acid (1%, v/v) and ethanol (50% v/v). To measure the dye taken up, the cell lysis products were centrifuged and supernatants spectrophotometrically measured at 540 nm.
Statistical Analysis
Data were expressed as mean ± standard deviation. Statistical analysis was performed by analysis of variance followed by Tukey’s test using GraphPad Prism 5 Software, Inc. (San Diego, CA, USA) and a
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by FARB 2018, University of Salerno.
