Leaves of Actinodaphne pilosa were collected at 2 different seasons from the Pù Hoạt Nature Reserve, Vietnam. The leaf samples were hydrodistilled to give essential oils, which were analyzed by gas chromatography (GC)–mass spectrometry and GC-flame ionization detection. The major components in the essential oils were α-pinene, (Z)-β-ocimene, (E)-β-ocimene, β-caryophyllene, germacrene D, bicyclogermacrene, and spathulenol. The essential oils were screened for antimicrobial activity against Enterococcus faecalis, Staphylococcus aureus, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans, as well as mosquito larvicidal activity against Aedes aegypti, Aedes albopictus, and Culex quinquefasciatus. Actinodaphne pilosa leaf essential oils showed broad antimicrobial activity (minimum inhibitory concentration = 32, 64, 64, 16, and 16 μg/mL against E. faecalis, S. aureus, B. cereus, P. aeruginosa, and C. albicans, respectively) and excellent larvicidal activity (24-hour 50% lethal concentration = 19.0, 24.7, and 48.1 μg/mL against A. aegypti, A. albopictus, and C. quinquefasciatus, respectively).
Actinodaphne pilosa (Lour.) Merr. (syn. Actinodaphne cochinchinensis Meisn., Laurus pilosa Lour., Machilus hainanensis Merr., Machilus pilosa [Lour.] Nees, Tetranthera pilosa [Lour.] Spreng.) (Vietnamese names, Bộp lông, Bánh dày) is a member of the Lauraceae and is a shrub or small tree (4-12 m tall) that ranges from southern China (Guangdong, Guangxi, Hainan) as well as Laos and Vietnam.
1
-3
The leaves and bark of this tree have been used medicinally to treat skin infections, coughs, rheumatism, and swelling.
2
There are numerous problematic emerging and re-emerging infectious diseases. Examples include bacterial infections such as Escherichia coli O157:H7,
4
vancomycin-resistant Staphylococcus aureus
5
; fungal infections by Trichosporon spp.,
6
Candida auris
7
; as well as arthropod-borne viral infections such as dengue,
8
yellow fever,
9
chikungunya,
10
or Zika.
11
It is imperative to identify new agents to combat the microorganisms themselves or the vectors responsible for their transmission. In this work, we present the chemical composition, antimicrobial activity, and mosquito larvicidal activity of the leaf essential oil of A. pilosa from north-central Vietnam.
Results and Discussion
Essential Oil Composition
Two different leaf samples of A. pilosa (samples 763L and 821L) were collected from the Pù Hoạt Nature Reserve in April and August 2019, respectively. The leaves were hydrodistilled to give essential oils in 0.37% and 0.41% yield, respectively, and were analyzed by gas chromatography–mass spectrometry (GC–MS) and gas chromatography-flame ionization detection (GC-FID) (Table 1).
The major components in Vietnamese A. pilosa leaf essential oils were α-pinene (6.0%, 7.2%), (Z)-β-ocimene (14.3%, 10.1%), (E)-β-ocimene (10.4%, 6.5%), β-caryophyllene (14.9%, 9.0%), germacrene D (12.0%, 16.2%), bicyclogermacrene (11.0%, 15.9%), and spathulenol (1.0%, 6.2%). The leaf essential oil composition of A. pilosa from Guangdong, China, has been reported.
12
The major components were viridiflorene (ledene, 12.7%), γ-muurolene (12.3%), germacrene D (11.6%), β-caryophyllene (10.7%), and globulol (5.9%). Thus, there are major qualitative and quantitative differences between the samples from Vietnam and China. The minor differences in composition between the two samples from Vietnam may be due to the phenological state; sample 763L was collected during the flowering stage (April 2019), while sample 821L was collected during the fruiting stage (August 2019).
Antimicrobial Activity
The leaf essential oils of A. pilosa were screened for antimicrobial activity against 3 Gram-positive bacteria (Enterococcus faecalis, Staphylococcus aureus, and Bacillus cereus), 2 Gram-negative bacteria (Escherichia coli and Pseudomonas aeruginosa), and 1 yeast (Candida albicans) (Table 2). Sample 763L was particularly active with minimum inhibitory concentration (MIC) values of 32, 64, 64, 16, and 16 µg/mL against E. faecalis, S. aureus, B. cereus, P. aeruginosa, and C. albicans, respectively.
It is not clear what compound(s) may be responsible for the antimicrobial activities in the complex A. pilosa essential oils. (Z)- and (E)-β-Ocimene do not appear to be particularly active.
13
-16
On the other hand, several investigations have shown β-caryophyllene
17,18
and germacrene D
16
to be broadly antimicrobial. In addition, essential oils rich in both bicyclogermacrene and β-caryophyllene have shown pronounced antimicrobial activity.
19,20
In addition to the antimicrobial activities of these sesquiterpene hydrocarbons, synergistic effects may also be responsible for the antimicrobial activities observed for A. pilosa leaf essential oil.
Mosquito Larvicidal Activity
The A. pilosa leaf essential oils were screened for mosquito larvicidal activity against Aedes aegypti, Aedes albopictus, and Culex quinquefasciatus (Table 3). Based on the criteria of Dias and Moraes,
21
the leaf essential oil of A. pilosa shows good larvicidal activities, particularly against the Aedes larvae.
The larvicidal activities of A. pilosa leaf essential oil can probably be attributed to the high concentrations of the sesquiterpene hydrocarbons β-caryophyllene, germacrene D, and bicyclogermacrene. β-Caryophyllene
22
-24
and germacrene D
25,26
have shown larvicidal activity against A. aegypti. In addition, the leaf essential oil of Lantana camara, rich in bicyclogermacrene (19.5%) and β-caryophyllene (16.7%), has shown larvicidal activity against A. aegypti.
27
On the other hand, (Z)- and (E)-β-ocimene are relatively inactive; essential oils rich in these components have shown only marginal larvicidal activity.
28,29
Conclusions
The leaf essential oil of A. pilosa can be considered to be active and shows promise as a potential antimicrobial agent and as an alternative insecticidal agent against mosquito larvae.
Materials and Methods
Plant Material
Leaves of A. pilosa were collected from the Pù Hoạt Nature Reserve in April 2019, flowering stage (sample 763L: 19°32′10″N, 104°41′28″E, elev. 870 m), and August 2019, fruiting stage (sample 821L: 19°42′19″N, 104°49′40″E, elev. 640 m). The plant was identified by Do N. Dai and voucher specimens (763 and 821) have been placed in the plant specimen room, Faculty of Agriculture, Forestry and Fishery, Nghe An College of Economics. In each case, 2 kg of the fresh leaves were shredded and hydrodistilled for 4 hours using a Clevenger apparatus to give the essential oils.
GC Analysis
GC-FID analysis was performed on an Agilent Technologies HP 7890A Plus Gas chromatograph equipped with an FID and fitted with HP-5ms column (30 m × 0.25 mm, film thickness 0.25 µm, Agilent Technologies). The analytical conditions were: carrier gas H2 (1 mL/min), injector temperature (programmed temperature vaporizing) 250°C, detector temperature 260°C, column temperature programmed from 60°C (2 minutes hold) to 220°C (10 minutes hold) at 4°C/min. Samples were injected by splitting and the split ratio was 10:1. The volume injected was 1.0 µL. Inlet pressure was 6.1 kPa.
An Agilent Technologies HP 7890A Plus Chromatograph fitted with a fused silica capillary HP-5ms column (30 m × 0.25 mm, film thickness 0.25 µm) and interfaced with a mass spectrometer HP 5973 MSD was used for the GC–MS analysis, under the same conditions as those used for GC-FID analysis. The conditions were the same as described above with He (1 mL/min) as the carrier gas. The MS conditions were as follows: ionization voltage 70 eV; emission current 40 mA; acquisition scan mass range of 35-350 amu at a sampling rate of 1.0 scan/s.
Compound identification was carried out by comparison of the retention indices (RI), which were determined with respect to a homologous series of n-alkanes, under identical chromatographic conditions, and the mass spectral fragmentation patterns found in the Wiley (Wiley 9th Version) and NIST 08 libraries (on ChemStation HP), or with those in the literature.
30
The concentrations of the chemical components were calculated based on the GC peak area (FID response) without using correction factors.
Antimicrobial Screening
The antimicrobial activity of the essential oils was evaluated using 3 strains of Gram-positive test bacteria, E. faecalis (ATCC 299212), S. aureus (ATCC 25923), B. cereus (ATCC 14579), 2 strains of Gram-negative test bacteria, E. coli (ATCC 25922) and P. aeruginosa (ATCC 27853), and 1 strain of yeast, C. albicans (ATCC 10231).
MIC and half-maximal inhibitory concentration (IC50) values were determined by the microdilution broth susceptibility assay. Stock solutions of the oil were prepared in dimethylsulfoxide. Dilution series were prepared from 16 384 to 2 µg/mL in sterile distilled water in micro-test tubes from where they were transferred to 96-well microtiter plates. Bacteria grown in double-strength Mueller–Hinton broth or double-strength tryptic soy broth and fungi grown in double-strength Sabouraud dextrose broth were standardized to 5 × 105 and 1 × 103 colony-forming unit/mL, respectively. The last row, containing only the serial dilutions of the sample without microorganisms, was used as a positive (no growth) control. Sterile distilled water and medium served as a negative (no antimicrobial agent) control. Streptomycin was used as the antibacterial standard; nystatin and cycloheximide were used as antifungal standards. After incubation at 37°C for 24 hours, the MIC values were determined to be well with the lowest concentration of agents completely inhibiting the growth of microorganisms. The IC50 values were determined by the percentage of microorganisms that inhibited growth based on the turbidity measurement data of EPOCH2C spectrophotometer (BioTeK Instruments, Inc Highland Park Winooski, VT, USA) and Raw data computer software (Brussels, Belgium) according to the following equations:

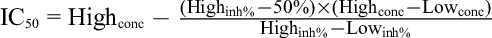
where OD is the optical density, control(–) are the cells with the medium but without an antimicrobial agent, test agent corresponds to a known concentration of the antimicrobial agent, control(+) is the culture medium without cells, Highconc/Lowconc is the concentration of test agent at high concentration/low concentration and Highinh%/Lowinh% is the % inhibition at high concentration/% inhibition at low concentration). Each of the antimicrobial screens was carried out in triplicate.
Larvicidal Screening
Mosquito colonies were obtained and maintained as previously described.
31
Larvicidal activity screening was carried out on 3rd instar larvae of A. aegypti, A. albopictus, and C. quinquefasciatus as previously described.
31
The data obtained were subjected to log-probit analysis to obtain 50% lethal concentration values, 90% lethal concentration values, and 95% confidence limits using Minitab 19 (Minitab, LLC, State College, PA, USA).