Abstract
Based on the strong activity dependence of paclitaxel (PTX; Taxol) or docetaxel (Taxotere) on the C-13 side chain, a small library of dehydroepiandrosterone, cholesterol, vitamin D2, and alkaloids talatisamine and songorine-PTX hybrids have been synthesized and evaluated for in vitro anticancer activity by MTT assay against human breast (MCF-7), colon (HCT116), lung carcinoma (A549), and renal adenocarcinoma (786-0) cancer cell lines. Most hybrids (

Taxol (

Structures of paclitaxel, docetaxel, and cabazitaxel.
Most of rings and side chains belonging to PTX has been modified in numerous attempts of modern chemists. 16 In contrast, less attention has been paid to the hybridization of PTX with biologically active natural products. Some PTX-thiocolchicine hybrids were synthesized by Danieli et al, 17 who found that, compared with PTX, 7-acetylpaclitaxel-thiocolchicine and N-debenzoyl-N-boc-paclitaxel- thiocolchicine have lower anticancer effect. 2′-Acetylpaclitaxel-thiocolchicine hybrid, however, has certain effects because acylation of the 2-OH group is adverse to tubulin polymerization.
Wittman et al 18 reported 3 PTX-chlorambucil hybrids and evaluated their cytotoxicity against the HCT116 human colon tumor cell lines which were not as excellent as PTX, but can provide extra antitumor effects when they were treated with PTX-resistant M109/taxlR cell lines. Six PTX-daunorubicin hybrids were synthesized by Wandless et al, 19 but none of them had more cytotoxicity than the parent monomers. Lack of analysis of the deep SAR of PTX has consistently limited the development of a synergistic combination of PTX with other drugs until Smith et al 20 designed 5 hybrids by utilizing a fully flexible (+)-discodermolide and a portion of the PTX side chain according to the inoccupation of the aromatic pocket occupied by the PTX side chain in the potential binding modes. Compared to the parent molecules, a 2- to 8-fold increase in antiproliferative activity was observed using the A549 and MCF-7 cancer cell lines. These studies indicate that active PTX-based hybrids can extend the medical utility of taxol.
Because of its complex structure, especially its baccatin component, obtaining PTX became prohibitively expensive, and it also prevents the large-scale production of synthetic PTX. Therefore, a baccatin-free compound with a taxane-like conformation which retains appreciable antitumor activity can be widely used in the field of anticancer for its simple synthetic technology and accessible materials. In our previous study, the assays of in vitro stabilizing microtubule and its antiproliferative activity indicated that PTX which mimics oxetane D-rings and side chains shows moderate antitumor activities.
6
The structures and configurations of some natural compounds like dehydroepiandrosterone (DHEA), cholesterol, vitamin D2, talatisamine, and songorine are similar to the taxane. In addition, these natural products are economical and easy to obtain. These benefits encourage us to synthesize taxane-like natural skeleton-C13 side chain hybrids to study simulated taxane skeletons and we expect to extend the therapeutic use of PTX to PTX-resistant or insensitive tumors. Herein we report the synthesis and in vitro anticancer activity of 8 PTX-natural product hybrids, in which the expensive baccatin core is structurally flexible to DHEA, cholesterol, vitamin D2, talatisamine, and songorine. Preliminary antitumor mechanism of PTX-DHEA hybrid (
Results and Discussion
We designed and synthesized a series of new hybrids which consisted of DHEA (
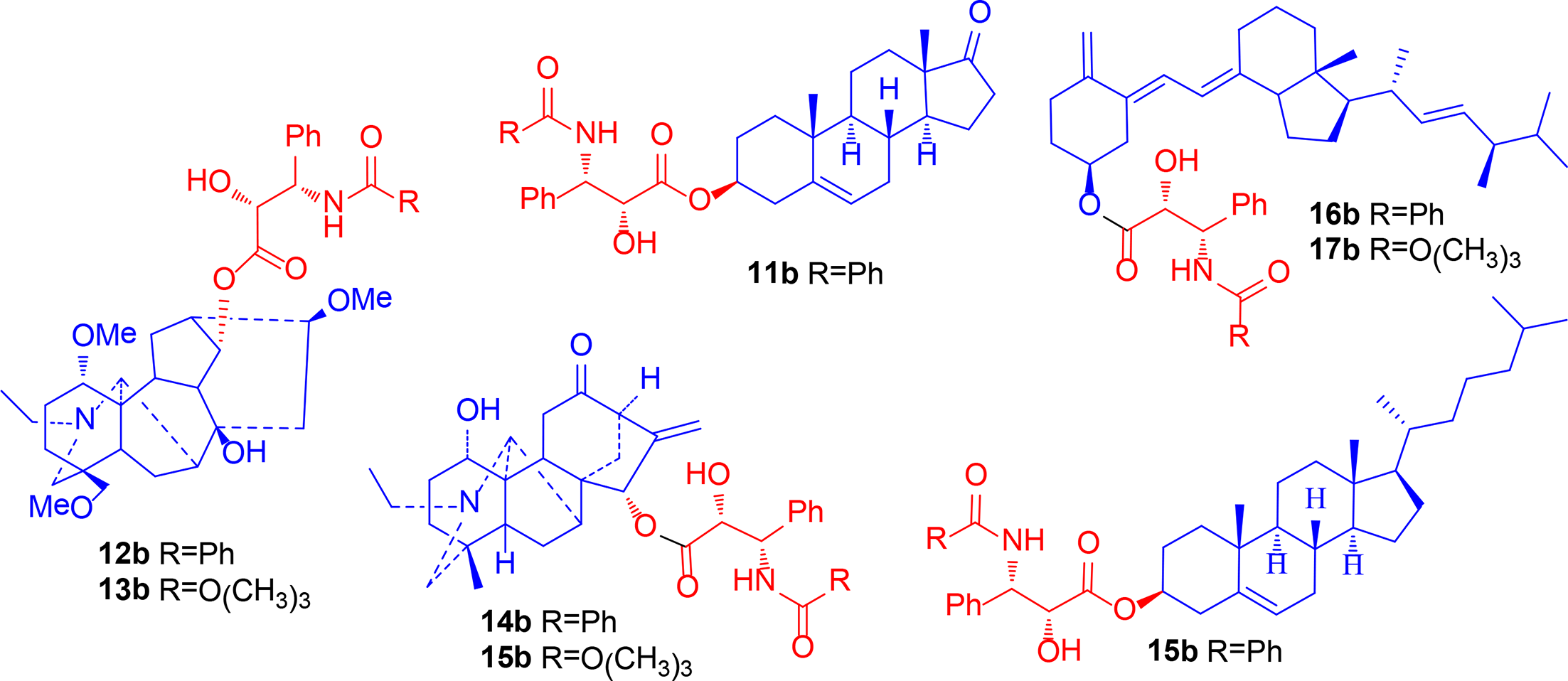
Structures of hybrids 11b to 18b.
For replacement of the baccatin core of toxoids (

Synthesis of paclitaxel hybrids. (a) 4 or 5, N′-(ethylcarbonimidoyl)-N,N-dimethylpropane-1,3-diaminemonohydrochloride, 4-dimethylaminopryidine, CH2Cl2, or CH3OH, 48.8% to 86.8%; (b) p-toluenesulfonic acid, MeOH, 59.3% to 78.5%.
At the beginning of our assessment, the inhibition of the 8 hybrids on human breast cancer (MCF-7), colorectal cancer (HCT116), lung cancer (A549), and renal adenocarcinoma (786-0) cancer cell lines was evaluated by MTT analysis.
22
The results showed that all compounds inhibited the growth of HCT116 and A549 cells at a concentration of 50 µM (Figure 3). Surprisingly, compared with compound

The inhibitory rates of the hybrids.
According to the desirable inhibitory effect of compound
Cytotoxicity of Compound 11b Against HCT116 Human Colon Cancer Cells After Different Exposure Times.


Dose-response curve of 11b.
The work direction of hybrid

Compound 11b inducing apoptosis by the Annexin V-FITC method.
As hybrid
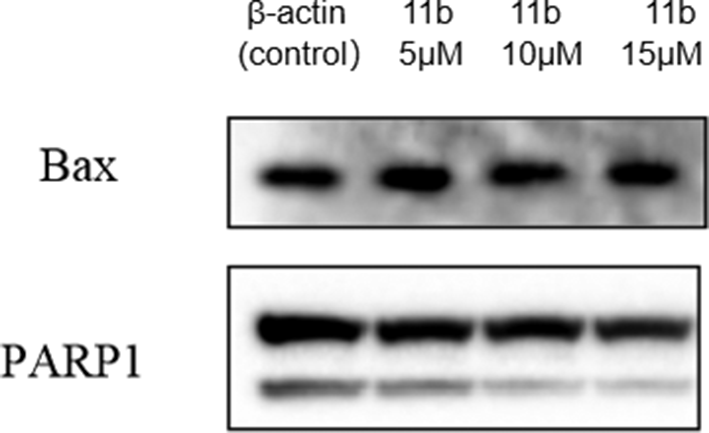
Levels of apoptosis-related proteins Bax and PARP1.
A computer modeling study was then performed to determine whether compound

Molecular docking result of compound 11b and beta-tubulin.

Molecular docking pattern of compound 11b.
Conclusions
In summary, 8 PTX-based hybrids were prepared and their inhibitory levels in the growth of 4 cancer cell lines were evaluated in vitro. Most compounds (
Moreover, hybrid
In general, this work reveals a successful hybridization by analyzing the SAR. The use of baccatin-free hybrid component could be an effective strategy to establish PTX-based hybrid library. However, it was turned out that the promising compound
Experimental
General Experimental Procedures
Unless otherwise stated, reagents were commercially available and used without further purification. The side chains
General Procedure for the Esterification Reaction of Compounds 6 to 10 With Protected Side Chain
A suspension of one of the starting compounds
General Procedure for the Deprotection of the Phenylisoserine Side Chain
Ester compounds
Compound 11a (Figure S1)
The reaction was carried out with 57.6 mg (0.2 mmol) of compound
Compound 12a
The reaction was carried out with 126.3 mg (0.3 mmol) of compound
Compound 13a
The reaction was carried out with 126.3 mg (0.3 mmol) of compound
Compound 14a
The reaction was carried out with 107.1 mg (0.3 mmol) of compound
Compound 15a
The reaction was carried out with 107.1 mg (0.3 mmol) of compound
Compound 16a
The reaction was carried out with 39.6 mg (0.1 mmol) of compound
Compound 17a
The reaction was carried out with 39.6 mg (0.1 mmol) of compound
Compound 18a
The reaction was carried out with 38.6 mg (0.1 mmol) of compound
Compound 11b
The reaction was carried out with 0.05 mmol (33.6 mg) of compound
Compound 12b
The reaction was carried out with 0.1 mmol (82.2 mg) of compound
Compound 13b
The reaction was carried out with 0.1 mmol (80.2 mg) of compound
Compound 14b
The reaction was carried out with 0.1 mmol (74.2 mg) of compound
Compound 15b
The reaction was carried out with 0.1 mmol (73.8 mg) of compound
Compound 16b
The reaction was carried out with 0.05 mmol (39.2 mg) of compound
Compound 17b
The reaction was carried out with 0.1 mmol (74.2 mg) of compound
Compound 18b
The reaction was carried out with 0.1 mmol (39.9 mg) of compound
Bioassays
MCF-7, HCT116, A549, and 786-0 cells were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). MTT (M2128) was purchased from Sigma-Aldrich (St. Louis, MO, USA). Antibodies used in this study were as follows: Bax (2772, CST), PARP (9532, CST), β-actin (66009-I-Ig, Proteintech).
Culture of Cell Lines
Cells were cultured in 175 cm2 flasks that were filled with
Cytotoxicity Assay by MTT Assay
MTT assay was employed for this primary determination as we did in previous report.
1
The in vitro inhibitory activities of 8 hybrids on 4 cells were evaluated. Cells were cultured in 96-well plates with a density of 5 × 103 for 1 day and then, respectively, pre-treated at 37℃ for 24 hours with each compound
Inhibition rate = (mean OD control - mean OD drug treated)/(mean OD control - mean OD vacuity) × 100%.
Experimental Details for Obtaining IC50 Values
HCT116 human colon cancer cells were cultured in medium with 10% FBS. HCT116 human colon cancer cells were added to each well of a 96-well plate. Increasing concentrations of hybrid
Determination of Apoptosis by Annexin V and Propidium Iodide Staining
Fluorescence intensity was analyzed using a flow cytometer (exposure of apoptotic cells was measured by adding Annexin V-FITC; Immunotech Coulter). The addition of PI can distinguish apoptoticcells (Annexin positive and PI-negative) from necrotic cells (Annexin-negativeand PI-positive). The HCT116 human colon cancer cells (2 × 105 cells) were seeded on 6-well plates and incubated for 12 hours when cells are at 70% to 80% confluency. Then, cells were treated with hybrid
Western Blot Analysis
After chemical treatment, cells were washed 3 times with PBS and lysed for 30 minutes at 4°C, followed by centrifugation at 15 000 r/min for 10 minutes at 4°C. Protein concentrations of the supernatants were assayed using the BCA protein assay kit.
According to our method in previous report, 2 accoraliquots of supernatants were added to SDS denaturing buffer and boiled for 10 minutes and transferred electrophoretically onto PVDF membranes. Membranes were blocked with 5% nonfat dry milk in TTBS (Tris-buffered saline containing 0.1% Tween 20) for 1 hour at room temperature with shaking and washed 4 times with TTBS before overnight incubation with the primary antibody at 4°C. Membranes were washed 4 to 5 times, 20 minutes each, with TTBS and then incubated with the secondary antibody conjugated to horseradish peroxidase for 1 hour at room temperature. Proteins were detected with an enhanced chemiluminescence reagent and visualized by ChemiDoc MP imaging system and quantified by densitometry using Quantity One 4.52 software.
Supplemental Material
Supplementary material - Supplemental material for Design, Synthesis, and Anticancer Activity of Natural Product Hybrids With Paclitaxel Side Chain Inducing Apoptosis in Human Colon Cancer Cells
Supplemental material, Supplementary material, for Design, Synthesis, and Anticancer Activity of Natural Product Hybrids With Paclitaxel Side Chain Inducing Apoptosis in Human Colon Cancer Cells by Ling-Li Zheng, Guan Wen, Yun-Xin Yao, Xiao-Huan Li and Feng Gao in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National Natural Science Foundation of China (grant numbers 31570341 and 31870329).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
