Abstract
A new chlorinated phenolic compound, methyl-3-chloro-2-hydroxy-4-methoxy-6-pentylbenzoate (
Lichens are symbiotic systems consisting of a mycobiont (the dominant fungal partner), one or more photobionts (algal partner), and a complex microbial consortium comprising a wide array of heterotrophic bacteria and fungi. 1,2 More than 1000 metabolites have so far been described from lichen sources, displaying various biological activities, such as antibiotic, antimycobacterial, antioxidant, antitumor, antiviral, analgesic, and antipyretic properties. 3 The Pertusaria genus, which contains about 800 species, is globally distributed from the tropics to the Arctic and Antarctic. Phytochemical reports have identified diverse compounds, including xanthones, 4 depsides, 5 depsidones, 6 and fatty acids 7 from several Pertusaria species, including Pertusaria amara, Pertusaria albescens, Pertusaria flavicans, Pertusaria pseudocorallina, Pertusaria truncate, and other species from the genus found in China. However, the chemical and biological characteristics of Pertusaria dactylina in its Antarctic distribution have so far been poorly reported. 8 In continuation of our research focused on the chemistry and biochemistry of this Antarctic lichen, we isolated a new chlorinated-phenolic compound, together with 4 known mono phenyl derivatives. This paper reports on the isolation and structure elucidation of the new compound (Figure 1).
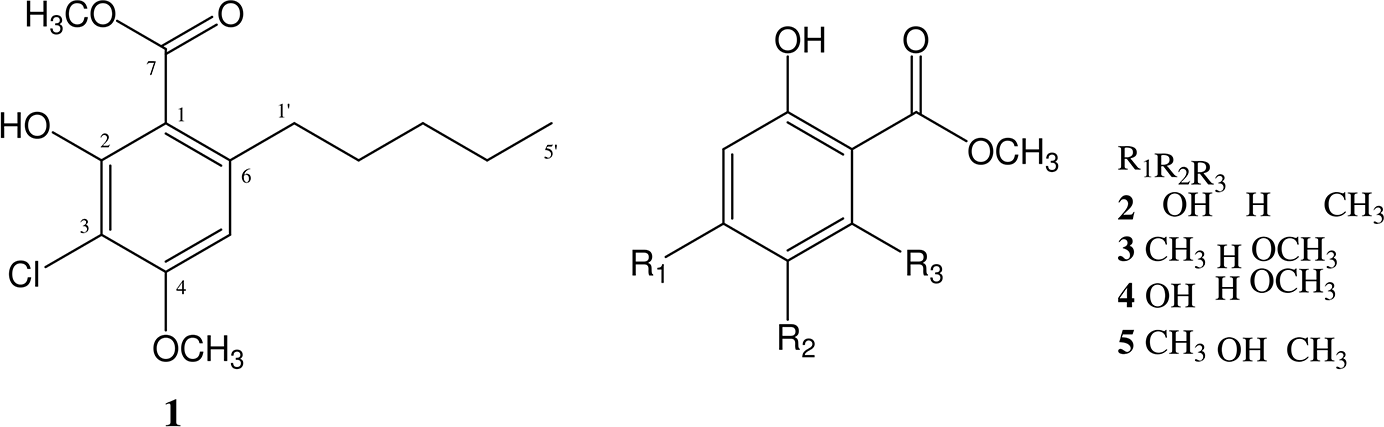
Chemical structures of compounds
The ethyl acetate partition of the Antarctic lichen, P. dactylina, was repeatedly subjected to column chromatography (CC) on silica gel, RP-18 gel, Sephadex LH-20 gel, and semipreparative high performance liquid chromatography (HPLC) to afford a new chlorinated-phenolic compound
Compound

Key HMBC correlations of compound 1.
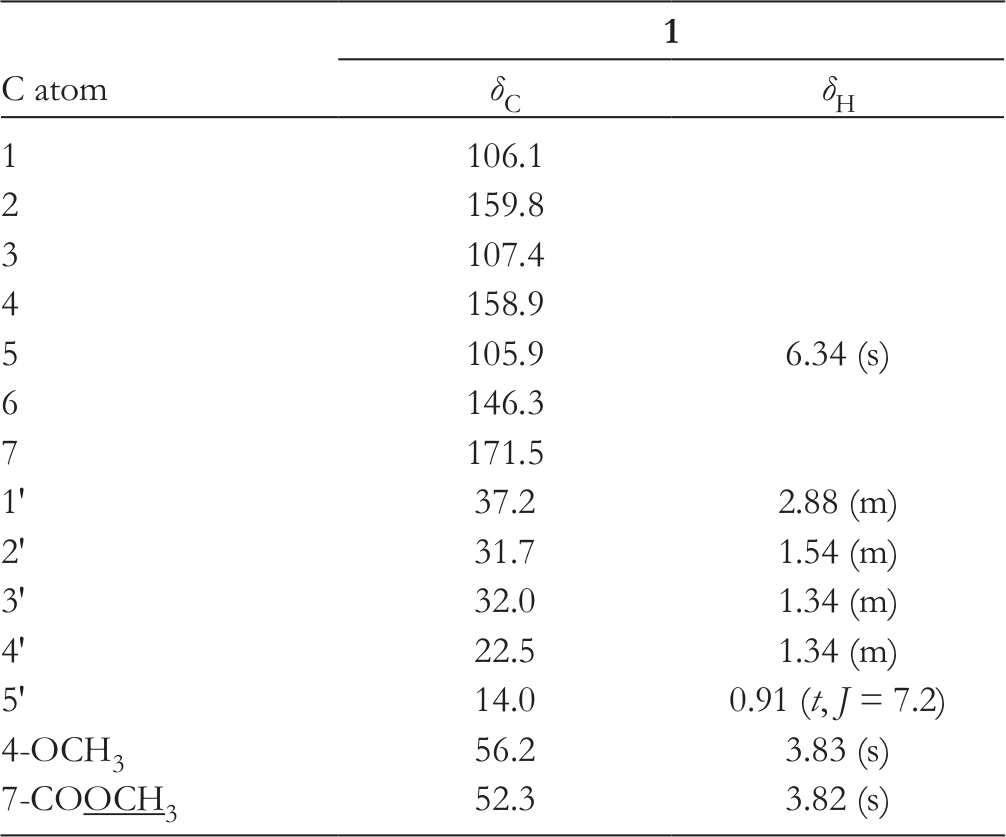
1H (600 MHz) and 13C (150 MHz) NMR Data for 1 (CDCl3, δ, ppm, J/Hz).
The physical and spectral properties of the known compounds we isolated were compared with the published values, and identified as methyl 2,4-dihydroxy-6-methylbenzoate (
Antimicrobial activities of compounds
Experimental
General
Optical rotations were measured on a Rudolph Research Autopol IV multiwavelength polarimeter. UV spectra were recorded on a Shimadzu PharmaSpec-1700 Ultraviolet (UV)-visible spectrophotometer. Infrared (IR) spectra were measured on a Bruker Tensor-27 spectrophotometer. 1D and 2D NMR spectra were recorded on a Bruker AVANCE (600 MHz) spectrometer. High-resolution electrospray ionization mass spectra (HREIMS) were obtained with an Agilent 6530 liquid chromatography quadrupole time-of-flight (LC-qTOF) High Mass Accuracy mass spectrometer operated in the positive- and negative-ion modes. Thin-layer chromatography (TLC) was performed on silica gel 60 F254 (0.25 mm, Merck, Germany). Silica gel (230-400 mesh, Merck, Germany) and C-18 (YMC·GEL ODS-A, 12 nm, S-150 μm) were used for CC. Semipreparative HPLC was conducted on YL9100 HPLC system (Young Lin, South Korea) equipped with a UV/Vis detector using an Alltech reversed-phase YMC-Pak C-18 column (10 µm, 20 × 250 mm) with a flow rate of 2 mL/min.
Lichen Material
The lichen, P. dactylina, was collected in January 2017 from King George Island, Antarctica, (62°12′53.69″ S; 58°55′23.87″W), and identified by Dr Ji Hee Kim and Miss Jae Eun So. A voucher specimen (no. Ant-053) was deposited at the Natural Product Chemistry Laboratory of the Korea Polar Research Institute.
Extraction and Isolation
An air-dried and powdered lichen, P. dactylina (80 g), was used for extraction by maceration in methanol (MeOH) (3 × 0.5 L) at room temperature. The solvent was concentrated in vacuo, yielding 3.0 g of a crude extract which was then suspended in distilled water (0.2 L) and extracted successively with n-hexane (2 × 0.2 L), ethyl acetate (EtOAc) (2 × 0.2 L), and n-butanol (2 × 0.2 L). The ethyl acetate partitions (1.2 g) were separated by CC over a silica gel column and eluted with Hex:EtOAc (90:10-50:50) to obtain 15 subfractions (Es1-Es15). The subfractions from Es4 to Es7 (250 mg) were combined based on their TLC patterns, subjected to a Sephadex LH-20 (30 g) gel CC using a solvent mixture (MeOH:H2O, 50:50). They were then purified with HPLC on a semipreparative C-18 gel column, using an MeOH:H2O mixture (60:40), and yielded
Methyl-3-Chloro-2-Hydroxy-4-Methoxy-6-Pentylbenzoate (1)
White amorphous powder.
UV (MeOH) λ max (log ε): 262 (4.0) nm.
1H- (600 MHz, CDCl3) and 13C-NMR (150 MHz, CDCl3) data, see Table 1.
HRFABMS: m/z 287.1050 [M+H]+ (calcd for C14H20ClO4, 287.1050)
Antibacterial Assays
The antimicrobial activities were tested using S. aureus KCTC 3881 (bacteria) and C. albicans KCTC 27242 (fungi) (Korean Collection for Type Cultures, Daejeon, Korea) in a 96-well plate. The cell culture was diluted up to 0.5 McFarland Standard with sterilized media. For C. albicans, the culture broth was 100 times more diluted before use. Each well was filled with 95 μL of culture broth. The compounds dissolved in DMSO were added until the final concentrations (0.5, 1, 2, 5, 10, 20, and 50 ug/mL), and the final volume of each well was 100 μL. 13 The plate was incubated at 25°C for 16 hours. Cell inhibition was measured at 600 nm (for S. aureus) and 530 nm (for C. albicans) using Multiskan GO Microplate Spectrophotometer (Thermo Scientific, Waltham, MA, United States). The IC50 value was calculated using an exponential trend line in Excel software (Microsoft, Redmond, WA, United States), and the values are mean ± standard errors of 3 determinations. Kanamycin and nystatin were used as positive controls against the bacterium and yeast, respectively.
Supplemental Material
Supplementary material - Supplemental material for A New Chlorinated Phenolic Compound From the Antarctic Lichen, Pertusaria dactylina
Supplemental material, Supplementary material, for A New Chlorinated Phenolic Compound From the Antarctic Lichen, Pertusaria dactylina by Man H. Koo, Min J. Kim, Jae E. Seo, Ji H. Kim, Se J. Han, Il C. Kim, Jun H. Lee and Ui J. Youn in Natural Product Communications
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant to the Korea Polar Research Institute, KOPRI, under a project (PE19210).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
