Abstract
Composite nanoparticles composed of an anionic phospholipid of 1,2-dipalmitoyl-sn-glycero-3-phosphorylglycerol (DPPG) and paclitaxel (PTX) were successfully prepared by mixing them in water followed by a subsequent heating/cooling process. The size of DPPG-PTX nanoparticle could be easily tuned by ultrasonic fragmentation. Upon addition of small-sized fluorescently labeled paclitaxel (FLPTX) nanoparticles with DPPG (DPPG-FLPTX) to rat skin tissue, part of the FLPTX molecules permeated to the stratum corneum.
Paclitaxel (PTX; Figure 1) is a diterpenoid having a taxane skeleton, which can be isolated from the bark of Taxus brevifolia. Paclitaxel is known to stabilize microtubules inside cells inhibiting depolymerization of the microtubule to tubulin proteins, which enables the suppression of cell divisions of cancer cells. 1 In particular, applications of PTX for treatments of breast, ovarian, colon, brain, and lung cancers have been extensively studied, as shown in many clinical results. 2 One of the recently emerging applications of PTX is as a therapeutic agent for skin cancers. 3 Skin is frequently exposed to oxidative stress from ultraviolet radiation, which presents a risk for the development of cancers such as melanoma, squamous cell carcinoma, and basal cell carcinoma. 4 Efficient transdermal delivery of PTX would be useful to cure these serious skin cancers. Skin tissue is composed of stratum corneum, epidermis, and dermis. However, the 10- to 40-μm-thick stratum corneum, consisting of densely packed cells, provides a barrier to protect the underlying tissue from infection, dehydration, chemicals, and mechanical stress, which prevents permeation of large nanoparticles across the skin tissue. So far, strategies for transdermal delivery of PTX stabilized by either albumin proteins 5 or polyethoxylated oil have been investigated. 6 Oil-free carriers for drug delivery have also been developed. 7,8 However, invasive syringe injection is sometimes required for transdermal delivery, or surfactant molecules which solubilize PTX, but which damage skin tissue causing irritation. 9 We have developed water-soluble PTX that is little invasive to skin. For example, we successfully synthesized glycosylated PTX by a biotransformation using plant culture cells in 2018. 10 Through these works, we had an idea to utilize phospholipid to solubilize PTX. Phospholipids are promising molecules because they are synthesized in the body, and therefore, highly biocompatible. In fact, water-soluble PTX nanoparticles have been reported, stabilized by dipalmitoylphosphatidylcholine (DPPC), a component of biological membranes [5, 10]. However, frequently utilized neutral phospholipids tend to form large-sized vesicles due to membrane fusions of the self-assemblies, which sometimes result in insufficient skin penetration. Thus, preparation of phospholipid-based PTX small-sized nanoparticles is still a challenging problem.

Molecular structures of paclitaxel, fluorescently labeled paclitaxel, and DPPG.
Here, we report PTX nanoparticles stabilized with anionic phospholipids of 1,2-dipalmitoyl-sn-glycero-3-phospho-1′-rac-glycerol (DPPG; Figure 1). Small-sized DPPG-PTX nanoparticles can be easily prepared by ultrasonic fragmentation. When size-controlled nanoparticles of fluorescent Oregon Green-labeled PTX (FLPTX; Figure 1) with DPPG (DPPG-FLPTX) are added to rat skin tissue, a part of the FLPTX molecules permeate to the stratum corneum. Materials with unique functions making full use of electrostatic repulsion between colloidal particles have recently emerged. For example, anisotropic hydrogels embedding magnetically oriented charged titanate nanosheets were recently reported as a seismic isolation material. 11 Since charged particles are hard to approach each other because of electrostatic repulsion, we expected that anionic phospholipids would prevent unavoidable problems of fusion events as in the case of conventional neutral phospholipids. DPPG is an anionic phospholipid synthesized in the body and molecularly similar to DPPC. DPPG is known to control lung pressure and functions of mitochondria by using a repulsive force because of the anionic charge. In addition, DPPG molecules are supposed to form kinetically stable nanoparticles maintaining the assemblies in physiological conditions because the bilayer melting temperature (T m) of DPPG is 41°C higher than body temperature.
DPPG-paclitaxel nanoparticles could be prepared by incubating a water dispersed powder sample of DPPG and PTX above T m of DPPG. As a typical example, we dispersed DPPG powder (5.0%, w/w) and PTX powder (0.1%, w/w), and heated for 15 minutes at 60°C above T m of DPPG (41°C), and then cooled to room temperature. As a result, we observed that the PTX molecules, forming precipitates without DPPG (Figure 2(b)), were well dispersed to show a transparent solution after stabilization with DPPG (Figure 2(c)). Particle size analysis by laser diffraction revealed that the size of the PTX aggregation decreased from 5.9 to 0.15 µm after the formation of DPPG-PTX nanoparticles (Figure 2(d)). Moreover, we treated the sample by ultrasonication for 3 hours to prepare small-sized nanoparticles. As a result, the DPPG-PTX nanoparticles were fractionated to 30 nm sized nanoparticles, as confirmed by dynamic light scattering (DLS) analysis (Figure 2(e)). Transmission electron microscopy also confirmed nanoparticles with a 30 nm size (Figure 2(f)). So far, PTX nanoparticles stabilized with neutral phospholipids have been reported. 5 However, it has been hard to prepare small-sized PTX nanoparticles achieved in this work. We envisioned that repulsive forces between nanoparticles derived from anionic phospholipid of DPPG would prevent fusion events between the nanoparticles.

Synthetic illustrations for the preparation of DPPG-paclitaxel nanoparticles (a). Necked-eye observation of dispersions of paclitaxel (b) and DPPG-paclitaxel nanoparticles (c). Particle size distribution of paclitaxel (red) and DPPG-paclitaxel (blue) as analyzed by laser diffraction (d). Particle size distribution of DPPG-paclitaxel before (blue) and after (green) ultrasonication for 3 hours as analyzed by dynamic light scattering (e). Transmission electron microscopy of small-sized DPPG-paclitaxel nanoparticles (f).
Then, we investigated skin permeability of DPPG-PTX nanoparticles. For this, we first checked that smaller-sized DPPG-PTX nanoparticles could be prepared even in phosphate buffered saline (PBS) buffer (close to in vivo conditions) (Figure 3(b)). Then, we prepared DPPG-FLPTX (Figure 3(c)) nanoparticles using the same method as that used for DPPG-PTX and incubated them with rat skin tissue placed on Franz diffusion cells. We prepared a histological section of the skin sample after 24 hours incubation and performed a fluorescent microscopic observation. Surprisingly, strong fluorescence derived from penetration of FLPTX molecules into the stratum corneum was observed (Figure 3(e) and (g)) as compared with the sample without DPPG-FLPTX (Figure 3(d) and (f)). It was noteworthy that we could observe fluorescence not only in the stratum corneum but also in the epidermis layer suggesting that FLPTX molecules partially penetrate even the stratum corneum. Although the molecular structure of FLPTX is not exactly the same as that of PTX, we envisaged that DPPG-PTX nanoparticles would have rather high skin permeation capability because the molecular structure of PTX is much smaller than that of FLPTX.
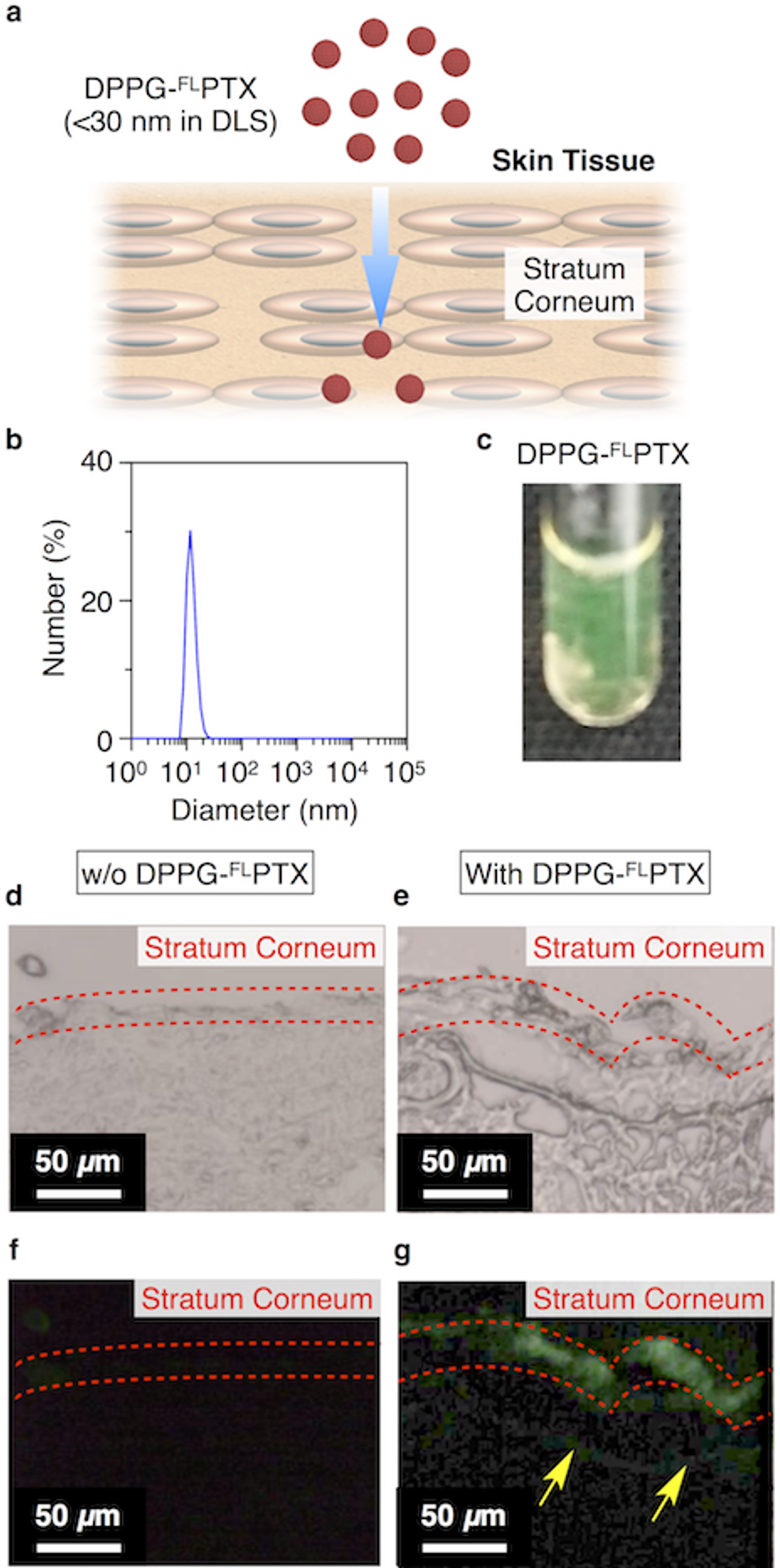
Synthetic illustrations for transdermal delivery of DPPG-fluorescently labeled paclitaxel nanoparticles (a). Dynamic light scattering profile of smaller-sized DPPG-paclitaxel nanoparticles in PBS buffer (b). Necked-eye observation of a dispersion of DPPG-fluorescently labeled paclitaxel nanoparticles (c). Bright field (d, e) and fluorescence image (f, g) of a sliced image of rat skin tissue 24 hours after incubation without (d, f) and with (e, g) DPPG-fluorescently labeled paclitaxel nanoparticles. Yellow allows indicate fluorescently labeled paclitaxel penetrating stratum corneum.
In this work, we have reported PTX nanoparticles stabilized by anionic phospholipids of DPPG. The DPPG-PTX nanoparticles can be easily prepared by heating followed by cooling of aqueous mixtures of DPPG and PTX, and the nanoparticles can be fractionated by an ultrasonication treatment to prepare small-sized nanoparticles. Moreover, upon addition to rat skin tissue, the small-sized DPPG-PTX particles penetrated into the stratum corneum. Although PTX is a well-known anticancer agent, applications of PTX to skin diseases have been challenging compared with diseases in other tissues because of difficulty in transdermal delivery of PTX. The DPPG-PTX nanoparticles having skin permeability demonstrated in this study would be a new candidate as a cancer therapeutic agent.
Experimental
General
Ultrasonication was performed by using a QSonica model ultrasonic homogenizer. Sonication was performed using a Branson model sonicator. Particle sizes were measured using either a Malvern model Zetasizer Nano ZSP zeta potential analyzer (DLS) or a Horiba model LA-960 laser diffraction particle size analyzer (SALD). Transmission electron microscopy was performed using a JEOL model JEM-1230 transmission electron microscope operating at an anode voltage of 80 kV, and the sample was stained with gadolinium acetate. Paclitaxel was purchased from Funakoshi (purity: >99%). Paclitaxel labeled with Oregon Green (FLPTX) was purchased from Thermo Fisher Scientific (purity: >92%). 1,2-Dipalmitoyl-sn-glycero-3-phospho-(1-rac-glycerol), sodium salt (DPPG) was purchased from Avanti Polar Lipids (purity: >98%).
Preparation of DPPG-PTX and DPPG-FLPTX Nanoparticles
Paclitaxel (0.1%, w/w) was mixed with DPPG powder (5%, w/w) in water and sonicated for 2 minutes to disperse homogeneously, and then heated at 60°C for 15 minutes, when the solution turned clear. The resulting mixture was kept at room temperature for 1 hour before use. DPPG-fluorescently labeled paclitaxel was prepared in the same way, except for using FLPTX (0.0025%, w/w) instead of PTX (0.1%, w/w). To tune the size of DPPG-PTX and DPPG-FLPTX nanoparticles, the samples were ultrasonicated at 50 W for 3 hours at 4°C.
Transdermal Delivery
In vitro skin permeation tests were performed using a vertical Franz diffusion cell with an effective diffusion area of 0.95 cm2. Skin tissues were obtained from abdominal hair of rats, as in a previous report. 12 The subcutaneous fat and other extraneous tissues of rat skin were trimmed and removed. A piece of excised skin (area 3.14 cm2, diameter 20 mm) was mounted on the Franz diffusion cell with the stratum corneum facing the donor compartment. One circular SS Nikasol or SS HGA patch (area 0.785 cm2, diameter 10 mm) was applied to the stratum corneum side of the skin. The receptor compartment was filled with 3 mL of water and maintained at 32°C using a circulating water bath stirred with magnetic bars. For microscopic observations, skin tissue was embedded into optimal cutting temperature (OCT) compound, frozen, and cryosectioned.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
