Abstract
A meroterpenoid, chevalone B, was obtained from the Cucumaria japonica-derived fungus Aspergillus sp. H30, which was collected from the South China Sea. The structure was determined by the analysis of nuclear magnetic resonance (NMR) spectroscopic and X-ray data. This is the first report of the crystallographic data and absolute stereochemistry of chevalone B, which was shown to have weak antibacterial activity.
Marine fungi are an exciting source for lead drugs and deserve to be studied thoroughly. Inspired by the widespread application of penicillin and the structural diversity of metabolites from fungi, such as cyclopentapeptides from Aspergillus versicolor, 1 a tricyclic lactone of an unprecedented skeletal type has been isolated from Acremonium strictum. 2 A strain of fungus was isolated from Cucumaria japonia collected in the South China Sea and identified as Aspergillus sp. H30. The ethyl acetate extract of this showed good anti-Candida albicans activity with a minimum inhibitory concentration (MIC) of 25 µg/mL and moderate anti-methicillin-resistant Staphylococcus aureus. (MASA) activity with a MIC of 50 µg/mL. Further separation led to the isolation of a meroterpenoid, the absolute stereochemistry of which was determined from NMR spectroscopic and X-ray crystallographic data and named as chevalone B. 3 The compound showed weak antimicrobial activity, but was cytotoxic to human BC1, KB, and NCI-H187 cell lines, and thus, exhibited enormous potential. 3
Chevalone B, isolated as colorless crystals, has a formula of C28H40O5, as measured from the high resolution electrospray Ionization mass spectrum (HRESIMS) [M+H]+ ion at m/z 457.2912 (calcd [M+H]+ 457.2954) see the support information Figure 4, implying 9 degrees of unsaturation. 1H and 13C NMR data see the support information Figure 2 and Figure 3 indicated 7 methyl carbon signals, and an acetoxy group at C-3 (δH 2.05, δC 21.3 and 171.0). Other details are listed in Table 1 and the X-ray Oak Ridge Thermal Ellipsoid Plot Program (ORTEP) drawing in Figure 1. Details of the absolute stereochemistry with (3S, 5R, 7R, 14R, 15R, 17S, 18S) are presented in the CIF file.
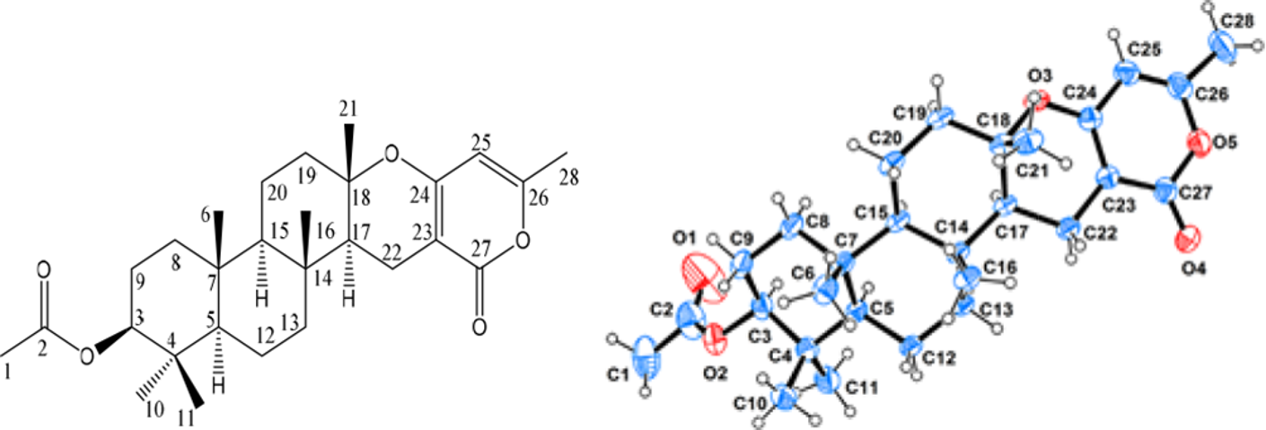
X-ray ORTEP drawing of chevalone B.
1H NMR (400 MHz) and 13C NMR (100 MHz) Data of Chevalone B in CDCl3.

Experimental
General
1H NMR and 13C NMR spectra were measured on a BRUKER ASCEND 400 AVANCE III HD with CDCl3 as solvent, and HRESIMS on an Agilent QTOF-6530 (Agilent Technologies, United States). Column Chromatography (CC) was carried out on silica gel (200-300 mesh, Qingdao Haiyang Chemicals) and Sephadex LH-20 (Amersham Biosciences). Precoated silica gel plates (Qingdao Marine Chemical Co; GF-254) were used for thin layer chromatography, and an Agilent 1260 Infinity instrument equipped with a diode array detector (DAD) detector for high performance liquid chromatography (HPLC).
Fungus Material
Aspergillus sp. H30 was isolated from C. japonica from the South China Sea. Voucher specimens are stored in the Biology Institute, Shandong Academy of Sciences, Jinan, PR of China.
Identification of the Entophytic Isolate
The fungus was grown on potato dextrose agar (PDA) for 7 days at 28°C. Genomic DNA was extracted and purified using Fungal DNA Kit 50 (OMEGA, United States), according to the manufacturer’s instructions, suitably modified. For identification and differentiation, the Internal Transcript Spacer (ITS) regions (ITS4 and ITS5) of the fungus were amplified by polymerase chain reaction (PCR) with universal ITS primers. The PCR products were then purified using the EZ Spin column PCR product purification kit (BBI) and sequenced. The sequencing results were aligned with the nucleotide-nucleotide database (BLASTn) of the U.S. National Center for Biotechnology Information (NCBI) for final identification of the fungus. The 480 base pair ITS sequence had 100% sequence identity with that of Aspergillus sp.
Fermentation, Extraction, and Isolation
The fungal strain was cultivated on solid medium at 28°C for 28 days in 50 × 500 mL Erlenmeyer flasks each containing 50 g rice in 80 mL of distilled water. The system was extracted 4 times with ethyl acetate. After filtration, the filtrate was concentrated to dryness under 42°C and the extracts subjected to silica gel CC. The desired compound, distributed in fraction 3, was further purified by Sephadex LH-20 chromatography using a mobile phase of CH2Cl2:CH3OH (1:1); fraction 3.2 was further subjected to semipreparative reverse-phase HPLC (octadecylsilane, ODS) (YMC, 250 × 10 mm) (85%MeOH-H2O, v/v, elution 28 minutes), and chevalone B (52.1 mg) was obtained with a t R = 21.1 minutes.
Crystallographic Data
Single crystals of chevalone B were obtained from CH3OH containing a small amount of CH2Cl2 at room temperature. The crystallographic data of chevalone B were collected on a Rigaku XtaLAB PRO single-crystal diffractometer equipped with graphite monochromated Cu-Kα radiation (λ = 1.54184 Å). The crystal was kept at 293(2) K during data collection. Structure determination was solved by direct methods using SHELXS-97 and refined with full-matrix least-squares on F2 using SHELXLS-97. 4 Positional and anisotropic displacement parameters were refined for all nonhydrogen atoms. Hydrogen atoms were placed in geometrically calculated positions and refined as part of a riding model. Crystallographic data are deposited at the Cambridge Crystallographic Data Centre (CCDC No. 1900770) and can be obtained free of charge from the CCDC Web site (www.ccdc.cam.ac.uk). Crystal data of chevalone B: C28H40O5 (M = 456.60 g/mol), orthorhombic, P212121, a = 7.18860(10) Å, b = 11.3685(2) Å, c = 31.2088(5) Å, α = 90°, β = 90°, γ = 90°, V = 2550.50(7) Å3, Z = 4, T = 293 (2) K, μ = 0.637 mm−1, D calc = 1.189 g/cm3, 8835 reflections measured (8.278° ≤ 2θ ≤ 148.582°), 4537 unique (R int = 0.0515); the final R 1 = 0.0405, w R 2 = 0.1030, (Δρ)max = 0.21 and (Δρ)min= −0.23 e/Å3. The Flack parameter was −0.02 (12). The selected bond lengths and bond angles are listed in Tables 2,3, respectively.
Bond Lengths for Chevalone B.

Bond Angles for Chevalone B.

Biological Assays
Antimicrobial activity was performed by the broth microdilution method in 96-well microtiter plates at 37°C for 16 to 24 hours, as well as in Sabouraud dextrose agar at 28°C for 48 hours. The test samples were dissolved in 0.1% dimethyl sulfoxide (DMSO). Amphotericin B was used as a positive control (MIC = 1.5625 µg/mL) for the in vitro test against C. albicans SC5314, and vancomycin against Staphylococcus aureus and MRSA (MIC = 1.25 µg/mL); DMSO (99.9%) was the negative control. MICs were over 100 µg/mL.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos. 81202452, 31320103911, 31430002, and 31770024), the Qinghai Key R&D and Transformation Project (Qinghai Science and Technology Department) (2017-NK-C25), the Open Project of Qinghai Key Laboratory of Qinghai-Tibet Plateau Biological Resources, Grant no. 2017-ZJ-Y10, the Priority Academic Program Development of Jiangsu Higher Education Institutions (No. 1107047002), the Natural Science Foundation from Shandong Province (Nos. ZR2017MH025 and ZR2017ZB0206), and the Taishan Scholar Project from Shandong Province to Lixin Zhang.
