Abstract
Asana used in Ayurveda medicine originates from the heartwood of Fabaceae Pterocarpus marsupium. In our previous report, we demonstrated that a 50% ethanolic extract (PM-ext) of Asana was a promising candidate as a skin-whitening agent based on its anti-tyrosinase and antioxidative activities. Moreover, the 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging activity of PM-ext was demonstrated and one of the active constituents was identified as oxyresveratrol. However, the potency of oxyresveratrol was low and the other active constituent has not yet been investigated. Activity-guided purification led us to isolate (+)-dihydrorobinetin (DHR). DHR had a potent DPPH radical-scavenging activity (half-maximal inhibitory concentration [IC50] = 1.3 µM) and also showed suppressive activity of advanced glycation end products (AGEs) production (IC50 =132 µM), as well as superoxide dismutase (SOD)-like activity (half-maximal effective concentration=27 µM). In a PM-ext, this is the first report to identify DHR and to demonstrate the suppressive activity of AGEs production and SOD-like activity of DHR. From these results, PM-ext containing DHR is a promising candidate as a multifunctional material with multiple anti-oxidant activities.
Keywords
Asana is an Indian kino tree used in Ayurveda medicine prepared by drying the heartwood of Fabaceae Pterocarpus marsupium. A decoction of a tumbler made from Asana has been administered to treat patients with diabetes mellitus. We focused on the potential of Asana as a promising candidate of multifunctional remedy to improve various disorders related to aging. Little research has been reported on its pharmacological function against aging. Ahmad et al reported that an ethyl acetate-soluble fraction obtained from an ethanolic extract of Asana had a hypoglycemic effect. 1 The effect was demonstrated by its insulin-increasing activity in alloxan-induced diabetes model rats. According to Manickam et al, marsupin and pterostilbene were reported to be the active constituents of the hypoglycemic activity in streptozotocin-induced diabetic model rats. 2
Based on our literature research, Asana has also been utilized to treat skin diseases in Ayurveda. 3 In a few studies on skin diseases, Dhirr et al reported its antifungal activity. 4 In addition, blood fluidity improvement in the microsystem can help reduce face dullness. 5,6 We demonstrated that oral administration of 50% ethanolic crude extract obtained from Asana (PM-ext) improved the blood fluidity in disseminated intravascular coagulation syndrome model rats based on platelet anticoagulation activity. In addition, a stilbene, pterostilbene, was identified as one of the active constituents. 7 Furthermore, we revealed that PM-ext had antimelanogenesis activity due to its tyrosinase inhibitory activity. Oxyresveratrol was identified as one of the active components using tyrosinase inhibitory activity-guided purification. 8 Because auto-oxidation is another major factor in melanogenesis, we confirmed the antioxidant activities of Asana using assays on 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging activity and suppression of advanced glycation end products (AGEs) as indicators. Oxyresveratrol was determined to be one of the active components in both assays. However, the DPPH radical-scavenging activity of oxyresveratrol was inadequate in PM-ext. Therefore, we investigated the other active components.
In previous reports, PM-ext showed 16%, 33%, and 73% of DPPH radical-scavenging activity at 10, 20, and 50 µg/mL, respectively. 8 We obtained an ethyl acetate-soluble fraction (AcOEt fr), an n-butanol-soluble fraction (BuOH fr) and a water-soluble fraction (H2O fr) from PM-ext. 8 Of these, AcOEt fr possessed the most potent DPPH radical-scavenging activity of 14% and 39% at 5 and 10 µg/mL, respectively. We prepared another AcOEt fr2 from a different lot of PM-ext in the same manner as AcOEt fr to investigate the active components of DPPH radical-scavenging activity (Figure 1). Five fractions (Fr. A, B, C, D, and E) were obtained from AcOEt fr2 by silicic acid gel column chromatography. Among them, Fr. D showed the most potent DPPH radical-scavenging activity of 58% at 10 µg/mL. Another 6 fractions (Fr. D-1, 2, 3, 4, 5, and 6) were obtained from Fr. D by preparative high-performance liquid chromatography (HPLC). Fr. D-5 showed the most potent DPPH radical-scavenging activity of these fractions with 88% at 10 µg/mL. Fr. D-5 was further purified again by HPLC and 6 peaks were observed and fractionated as Fr. D-5-1, 2, 3, 4, 5, and 6. Two fractions, Fr. D-5-1 and 2, were combined since D-5-1 and 2 showed identical HPLC profiles. The content of the combined fraction was identified as (+)-dihydrorobinetin (DHR) by comparing the HPLC profile, 1H- and 13C-nuclear magnetic resonance (NMR) spectra, and optical rotation data with those of an authentic sample (Figure 2). This is the first report on obtaining DHR from PM-ext.

Activity-guided isolation of (+)-dihydrorobinetin. (a) Yield from PM-ext; (b) Yield from AcOEt fr2; (c) Yield from Fr. D; (d) Yield from Fr. D-5; (e) Yield from Fr. D-5-1 and Fr. D-5-2; (f) DPPH radical-scavenging activity at 10 µg/mL. DPPH, 1,1-diphenyl-2-picrylhydrazyl; HPLC, high-performance liquid chromatography; PM-ext, 50% ethanolic crude extract obtained from Asana.

The chemical structure of (+)-dihydrorobinetin.
DHR possessed potent DPPH radical-scavenging activity of 16%, 34%, and 89% at 0.50, 1.0, and 5.0 µM, respectively (Figure 3). The half-maximal inhibitory concentration (IC50) value for DHR was estimated to be 1.3 µM. DHR is known as a marker for aging of vinegar and wine in wooden barrels. 9,10 The antioxidant activity of DHR may contribute to the functional properties of vinegar and wine. It is suggested that DHR also contributes to the antioxidative function of Asana.

DPPH radical-scavenging activity of (+)-dihydrorobinetin. OD: Optical density at 520 nm. Each value represents the mean ± SD of triplicates. Significantly different from control at **P < 0.01.
The high potency of DHR in terms of DPPH radical-scavenging activity led us to investigate its suppressive activity on AGEs production. As a result, DHR showed 15%, 34%, and 70% suppression at 50, 100, and 200 µM, respectively (Figure 4) and the IC50 value was estimated to be 132 µM. This is the first report on the suppressive activity of DHR on AGEs production. AGEs are known to induce melanogenesis as well as generate reactive oxygen species by binding to AGE receptors. 11,12 The DPPH radical-scavenging activity and suppressive activity on AGEs production of DHR suggest that the improvement in diabetes achieved by consuming a decoction of Asana is partly attributable to DHR.
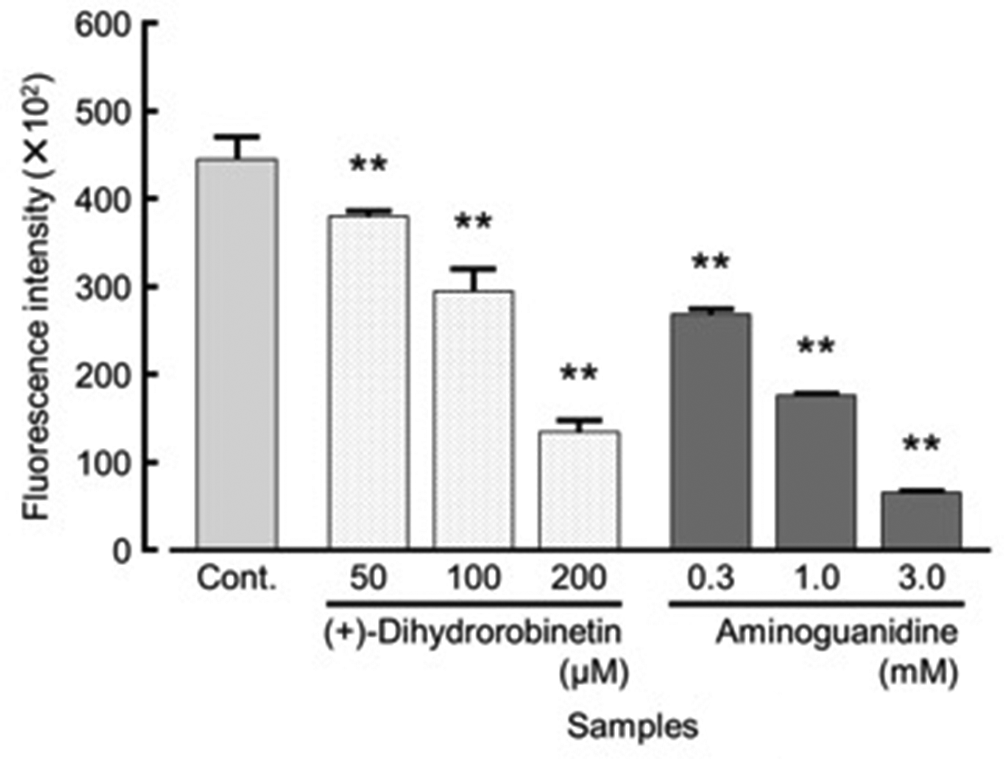
(+)-Dihydrorobinetin suppressive activity on AGEs. Fluorescence intensity was measured at 370 nm (excitation) and 460 nm (fluorescence). Each value represents the mean ± SD of triplicates. Significantly different from control at **P < 0.01. AGE, advanced glycation end product.
The superoxide dismutase (SOD)-like activity of DHR was also investigated. This enzyme is important in the defense against oxidative stress and is known as an intravital factor that removes oxidants. DHR showed 42%, 68%, and 77% SOD-like activity at 20, 50 and 100 µM and the half-maximal effective concentration value was estimated to be 27 µM (Figure 5). This is also the first report on the SOD-like activity of DHR.

SOD-like activity of (+)-dihydrorobinetin. OD: Optical density at 550 nm. Each value represents the mean ± SD of triplicates. Significantly different from control at *P < 0.05, **P < 0.01. SOD, superoxide dismutase.
In this study, the active constituent of PM-ext involved in DPPH radical-scavenging activity was first determined to be DHR. DHR’s suppressive activity on AGEs production and SOD-like activity were also demonstrated for the first time. From these results, PM-ext containing DHR is a promising candidate as a multifunctional material with the multiple antioxidant activities. Further investigation of the active constituents is now underway in our laboratory.
Experimental
Reagents
Reagents used in this study were purchased from FUJIFILM Wako Pure Chemicals Corp. (Osaka, Japan), Nacalai Tesque (Kyoto, Japan), or Sigma-Aldrich (St. Louis, MO, USA), unless otherwise stated.
Preparation of Extracts From Heartwood of P. marsupium
Heartwood of P. marsupium was harvested in Kerara, India on 10th April 2014 and provided by INABATAKORYO Co., Ltd. in May 2014. The voucher specimen of this plant is deposited at the Faculty of Pharmacy, Kindai University (Voucher No. PM-20140410).
Pulverized heartwood (400 g) was extracted with 50% ethanol (EtOH, 4 L) under reflux for 2 hours. The filtrates were combined and the solvent was evaporated under reduced pressure and lyophilized to give a brown powder (PM-ext, 46.4 g, 11.6%).
Fractionation of PM-Ext
The PM-ext (40 g) was suspended in water (600 mL) and extracted with ethyl acetate (600 mL × 3). The yields of AcOEt fr2 and Insoluble fr were 10 and 30 g, respectively. The AcOEt fr2 (10 g) was subjected to silicic acid column chromatography (280 g, Merck, 2.25 internal diameter [i.d.] × 40 cm). Elution was performed with mixtures consisting of A: n-hexane (Hex), B: AcOEt (A: B = 5:1, 3:1, 1:1, and 1:3) and methanol (MeOH), and 5 fractions were obtained as Fr. A, B, C, D, and E. Fr. D (750 mg) was then subjected to preparative HPLC under the following conditions: column; Waters SunFire C18 column (10 i.d. × 250 mm), mobile phase; water/MeOH = 19:1 (0 minutes) to 9:11 (30 minutes) in a linear gradient, flow rate; 4.0 mL/min, detection; UV 280 nm to obtain 6 fractions (Fr. D-1 to 6). Fr. D-5 (48 mg) was then subjected to preparative HPLC under the following conditions: column; Waters SunFire C18 column (10 i.d. × 250 mm), mobile phase; water/methanol = 4:1 (0 minutes) to 1:9 (30 minutes) in linear gradient, flow rate: 3.0 mL/min, detection; UV 280 nm to give DHR (8.3 mg). The chemical structure of DHR was determined by comparison of 1H- and 13C-NMR spectra and optical rotation with those from an authentic sample.
Radical-Scavenging Activity
Radical-scavenging activity was measured according to the method of Blois with minor modification. 13 The test sample was dissolved in dimethylsulfoxide (DMSO) and diluted with 0.5 M acetate buffer (pH 5.5) to a final DMSO concentration of 5% v/v. A mixture of test sample solution (100 µL), EtOH (64 µL), 0.5 M acetate buffer (pH 5.5) (16 µL), and 1.0 mM DPPH/EtOH solution (20 µL) was allowed to stand for 30 minutes at room temperature. The optical density (OD) of the resulting mixture at 520 nm was determined with a microplate reader. L-Ascorbic acid was used as a reference agent. The scavenging activity of each sample was expressed as a percentage of the decrease in OD compared with that of a control DPPH solution.
Suppression Activity of AGEs Production
AGEs formation activity was measured according to the method of Shimoda et al and Itoh et al with minor modification. 14,15 The test sample was dissolved in DMSO and diluted with sodium phosphate buffer (0.2 M KH2PO4, 0.2 M NaOH, pH 7.2) to a final DMSO concentration of 1% v/v. A reaction mixture of glucose (10% w/v) and bovine serum albumin (BSA, 1% w/v) dissolved in phosphate buffer (900 µL) was incubated for 48 hours at 60°C in microtubes (2 mL) with or without a test solution. After incubation, the fluorescence (F) associated with AGEs in the reaction mixture was monitored at an excitation wavelength of 370 nm and emission of 450 nm using a multilabel counter (PerkinElmer 2030 ARVO X4,PerkinElmer Life and Analytical Sciences, Waltham, MA, USA). Aminoguanidine hydrochloride was used as a reference agent. The inhibitory ratio of the sample was calculated using the following formula:
where F control is the fluorescence with phosphate buffer containing glucose and BSA, F normal is the fluorescence with PBS containing glucose and BSA without incubation (stored at 4°C), F sample is the fluorescence with sample solution in phosphate buffer containing glucose and BSA, and F sample is blank fluorescence with sample solution in phosphate buffer. Each assay was performed in triplicate.
SOD-Like Activity
SOD-like activity was measured according to the method of Oyanagui with minor modification. 16 The test sample was dissolved in DMSO and diluted with 0.5 mM disodium dihydrogen ethylenediamine tetraacetate (EDTA)-PBS buffer (pH 8.2) to a final DMSO concentration of 1% v/v. SOD, originating from cow’s milk (Roche Co., Tokyo, Japan), was dissolved with 0.5 mM EDTA-PBS buffer (pH 8.2) as a reference. A mixture of 0.5 mM EDTA-PBS buffer (pH 8.2) (0.2 mL), 0.5 mM hypoxanthine in EDTA-PBS buffer (pH 8.2) (0.2 mL), reagent A solution (10 mM hydroxylamine hydrochloride and 1 mg/mL hydroxylamine-O-sulfonic acid in water) (0.1 mL), water (0.2 mL), and the sample solution (0.1 mL) was preincubated at 37°C for 10 minutes. Five milliunits per milliliter xanthine oxidase (Roche Co., Tokyo, Japan) solution in 0.5 mM EDTA-PBS buffer (pH 8.2) (0.2 mL) was added to the above solution, and the mixture was incubated at 37°C for 30 minutes. Reagent B (30 µM N-1-naphthylethylenediamine・2 HCl, 3 mM sulfanilic acid, and 25% acetic acid in water) (2 mL) was added to the reaction mixture. The resulting mixture was allowed to stand for 30 minutes at room temperature, and then OD was measured at 550 nm with a UV-Visible spectrophotometer (Shimadzu, UV-2450, Kyoto, Japan). The SOD-like activity of each sample was expressed as a percentage of the decrease in OD compared with that of control.
Statistical Analysis
The experimental data were statistically analyzed with Statcel3 (The Publisher OMS, Tokorozawa, Japan), add-in software for Excel®, using one-way analysis of variance. Statistical significance was analyzed by Bonferroni/Dunn’s multiple-range tests and significance was detected at P < 0.05 and P < 0.01.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

