Abstract
The chemical composition of the bulbs of Pancratium maritimum L. (Amaryllidaceae) from Turkey (Pamucak, Aydın) has been determined by gas chromatography-mass spectrometry. A total of 29 compounds belonging to different skeletal types of Amaryllidaceae alkaloids were identified. Lycorine, galanthamine, crinine, and pancracine were found as major constituents. Interestingly, indole alkaloids (1-acetyl-β-carboline and galanthindole) were also detected. Acetylcholinesterase, butyrylcholinesterase, and prolyl oligopeptidase inhibitory activity potentials of the alkaloidal extract were also determined. The results of the present study show that Turkish P. maritimum comprises a rich repository for bioactive alkaloids with intriguing structural diversities.
The family Amaryllidaceae, consisting of about 1100 species and 85 genera, is well known for its ornamentally valued plants and their biologically active alkaloids. 1 Pancratium L. is a genus of bulbous monocotyledons which occurs on sand dunes and other sandy coastal habitats. 2 Although Pancratium includes about 15 species, distributed throughout the Mediterranean, Asia, and Africa, only Pancratium maritimum (sea daffodils) is indigenous to Turkey. 2,3
Galanthamine (Reminyl) is one of the most important Amaryllidaceae alkaloids, which is used for the treatment of mild to moderate Alzheimer’s disease (AD) as a cholinesterase inhibitor. 4 Acetylcholinesterase (AChE) inhibitors provide increased levels of acetylcholine at the synaptic junctions. Butyrylcholinesterase (BuChE) inhibitors play a key role, especially in the limbic system. 5 Lycorine, the most common Amaryllidaceae alkaloid, also has several biological activities such as antiviral, 6 antitumor, 7 anti-inflammatory, 8 and antiprotozoal. 9 Besides galanthamine and lycorine, other Amaryllidaceae alkaloids have crucial biologically activity potential, including tazettine, pancratistatin, narciclasine, hippeastrine, and haemanthidine. 10 -12
Prolyl oligopeptidases (POPs) are a group of enzymes belonging to the serine peptidase enzyme family, 13 and deterioration of serum POP activity has been linked with a number of neuropsychiatric problems including learning and memory problems. 13,14 Low serum POP activity has been found in patients with depression, anorexia, and alcohol addiction. On the contrary, increased serum POP levels could be associated with mania, bipolar disorder, and schizophrenia. 14,15 Recent studies have shown that POP inhibitors improve learning disability and inhibit amyloid accumulation in AD animal models. 16
The present paper describes a sensitive, practical, and fast method based on a gas chromatography-mass spectrometry (GC-MS) technique for the determination and identification of isoquinoline alkaloids in alkaloidal extract of P. maritimum bulbs. Acetylcholinesterase, BuChE, and POP inhibitory activities of the extract were also investigated. Pancratium maritimum extracts were reported to have antimalarial, 17 insecticidal, 18 antifungal, 19 anticholinesterase, 20 and cytotoxic activities. 21 In this study, BuChE and POP inhibitory activities plus alkaloid profiling by GC-MS on Turkish bulbs were carried out for the first time.
The results of GC-MS analysis of P. maritimum alkaloidal extract are given in Table 1, which includes GC-MS experimental data, retention time (RT), molecular ion peak [M+], and main fragments of the compounds. The proportion of each alkaloid in the tested extract was connoted as the percentage of the total alkaloidal content. The area of the GC-MS peaks depends not only on the concentration of the related compounds but also on the intensity of their MS fragmentation. A total of 29 compounds were identified of which 27 matched those either reported in the literature or recorded MS data of standard compounds. Tyramine (
Alkaloids of Pancratium maritimum.
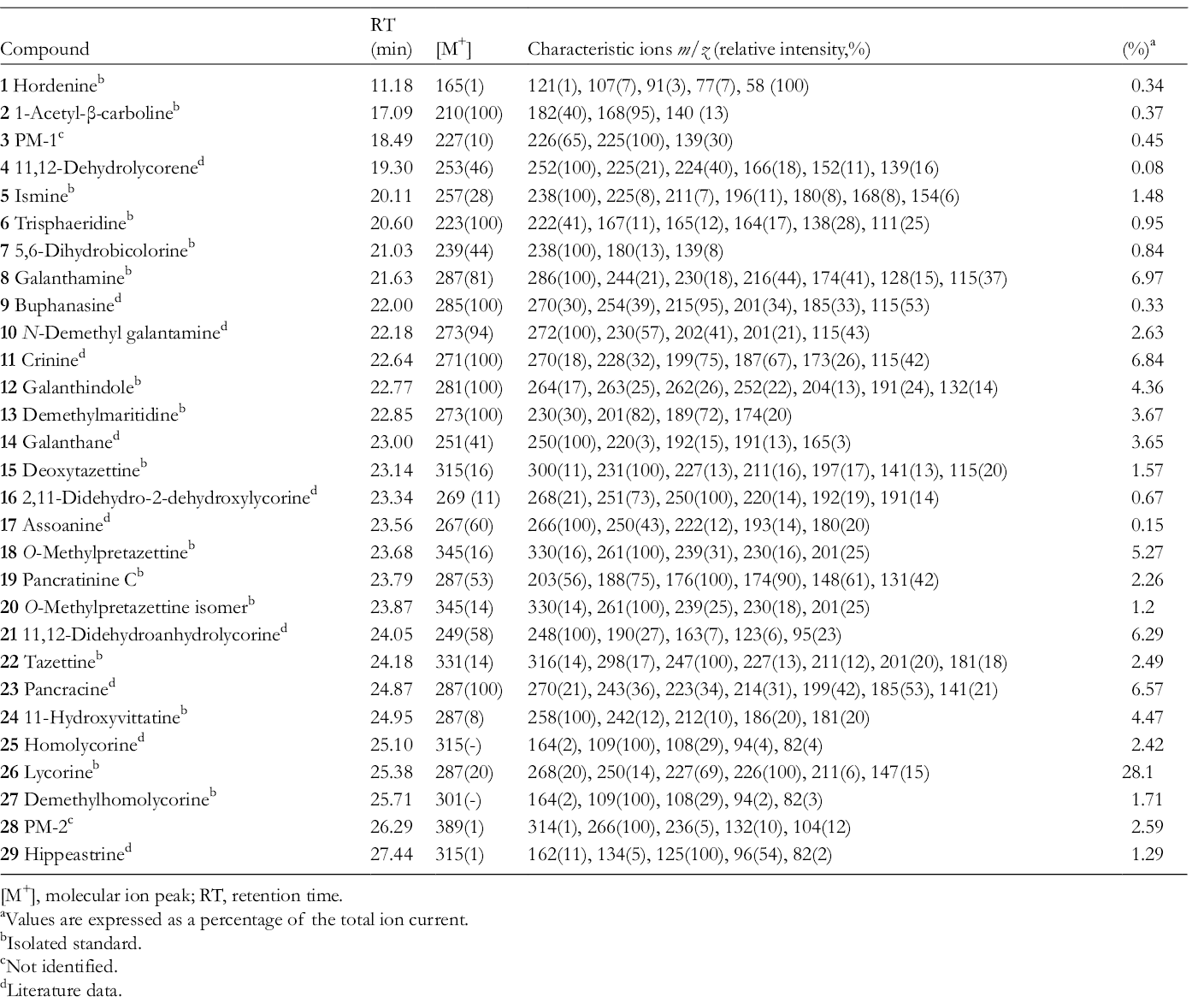
[M+], molecular ion peak; RT, retention time.
aValues are expressed as a percentage of the total ion current.
bIsolated standard.
cNot identified.
dLiterature data.
The AChE inhibitory activity (inhibitory concentration of 50%, IC50 3.49 µg/mL) of the bulbs was found to be more potent than the BuChE inhibitory activity (IC50 28.96 µg/mL). Galanthamine was used as a positive control. The IC50 value of galanthamine for AChE and BuChE inhibitory activities was 0.043 and 0.711 µg/mL, respectively. The previous studies have shown that galanthamine and some lycorine type alkaloids have potent anticholinesterase activity. 24,25 Thus, the presence of lycorine and galanthamine type alkaloids in the tested P. maritimum extract could be responsible for their high anticholinesterase activity.
Prolyl oligopeptidase inhibitory activity was carried out using a modified colorimetric method, 26,27 which is reported here for the genus Pancratium for the first time. The extract of P. maritimum bulbs showed POP inhibitory activity with an IC50 value of 78 µg/mL. (Z)-Pro-prolinal was used as a standard (IC50 0.045 µg/mL).
Experimental
Plant Material
Pancratium maritimum, collected from Pamucak-Aydın, was identified by Prof M. Ali Onur (Ege University). A voucher specimen (No. 1294) is deposited in the Herbarium of the Department of Pharmacognosy, Faculty of Pharmacy, Ege University.
Chemicals
The standard compounds for GC-MS analysis and galanthamine for anticholinesterase activity were previously isolated from several Amaryllidaceae species in our laboratory and authenticated by means of spectral analyses. Acetylcholinesterase from Electrophorus electricus (electric eel), BuChE from equine serum, and POP (recombinant, expressed in Escherichia coli) were purchased from Sigma-Aldrich (St Louis, MO, United States). All chemicals and reagents were analytical grade, obtained from either Merck (Darmstadt, Germany) or Sigma-Aldrich.
Extraction
The alkaloidal extract prepared from P. maritimum bulbs was used for GC-MS analysis and in the biological activity assays. Air-dried, powdered plant material (500 mg) was extracted at room temperature 3 times with 5 mL methanol (30 minutes each) in an ultrasonic bath. The solvent was removed under vacuum and the residue dissolved in 10 mL 2% sulfuric acid. The nonpolar compounds were removed with diethyl ether (3 × 10 mL). The acidic aqueous phases were basified with 25% ammonia to pH 9 to 10 and the alkaloids extracted with chloroform (3 × 10 mL). These extracts were dried over anhydrous sodium sulfate and the organic solvent distilled in vacuo to give the alkaloidal extract.
Gas Chromatography-Mass Spectrometry
Gas chromatography-mass spectrometry analysis of underivatized alkaloids was performed using a Thermo GC-Trace Ultra Ver: 2.0., Thermo MS DSQ II (Thermo Fisher Scientific, San Jose, CA, United States) operating in electron impact mode. The electron energy was 70 eV. The temperature program: 80°C for 1 minute, 80°C to 250°C (10°C/min), 250°C for 2 minutes, 250°C to 300°C (10°C/min) and a 10 minute hold at 300°C. An injector temperature of 250°C was used. Helium was the carrier gas at a flow rate of 0.8 mL/min. A TR-5 MS column (30 m × 0.25 mm × 0.25 µm) was used. Mass spectra were acquired in the mass range from m/z 50 to 450. The extracts were dissolved in methanol (1 mg extract in 250 µL methanol). A split/splitless injector was used in splitless mode. The spectra of co-eluting chromatographic peaks were investigated and deconvoluted by the use of Xcalibur (version 2.07; Thermo Fisher Scientific San Jose, CA, United States). The compounds were identified by comparing their mass spectral fragmentation with standards previously isolated in our laboratory from several Amaryllidaceae species. Moreover, RTs and data obtained from the literature were used for the identification of the compounds. The percentage of the total ion current for each compound is given in Table 1.
Acetylcholinesterase and BuChE Inhibitory Activities
Acetylcholinesterase and BuChE inhibitory activities were performed spectrophotometrically using a microplate assay modified from Ellman’s colorimetric method, 28 as previously described. 29 Samples with final concentrations of 150, 100, 50, 25, 12.5, 2.5, and 0.25 µg/mL were tested. The enzyme inhibitory activity was measured as a percentage compared with a blank in a 96-well microplate reader (VersaMax Tunable Microplate reader, Molecular Devices, United States). The IC50 value was calculated using GraphPad Prism v7.0 software (GraphPad Prism Software, Inc., La Jolla, CA, United States) by means ± SD of 3 individual determinations, each performed in triplicate.
Prolyl Oligopeptidase Inhibitory Activity
The enzyme activity was assayed using a colorimetric method of Yoshimoto and Cahlikova et al 26,27 with the following modifications. The enzyme (0.2 U/mL) was dissolved in sodium phosphate buffer. Six different concentrations (0.25-600 µg/mL) of the extract were prepared. The buffer solution, test sample, and POP were added to each well of a 96-well plate. After incubation for 5 minutes (30°C), POP substrate, Z-Gly-Pro-p-nitroanilide (in 40% 1,4-dioxane), was added. The reaction was carried out for 30 minutes (30°C), and the initial rate of the enzyme was analyzed by measuring the formation of p-nitroaniline at 405 nm with a microplate reader. The enzyme inhibitory activity was calculated as a percentage compared with the blank. The IC50 value was obtained using the software package GraphPad Prism v7.0 software by means ± SD of 3 individual determinations, each performed in triplicate.
Footnotes
Acknowledgment
The authors acknowledge the assistance of Prof. M. Ali Onur for the collection of plant material and FABAL (Ege University Faculty of Pharmacy Pharmaceutical Sciences Research Centre) for facilitating GC-MS analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
