Abstract
Three monoterpene glucosides were isolated from the flowers of Brugmansia arborea L. using repeated silica gel and octadecyl SiO2 column chromatography. Based on spectroscopic data including 1d-NMR (1H, 13C, and distortionless enhancement by polarization transfer (DEPT)), 2D-NMR (gradient correlation spectroscopy (gCOSY), gradient heteronuclear single quantum coherence (gHSQC), and gradient heteronuclear multiple bond coherence (gHMBC)), Infrared Spectroscophy, and Mass Spectroscophy, the glucosides were identified as citronellol O-
β-D-glucopyranoside (
Brugmansia arborea L. (Solanaceae), commonly called angel’s trumpet, is an evergreen shrub distributed in America, Africa, Australia, and Asia.
1
This plant is commonly used for ornamental purposes
2
and also for analgesic, antirheumatic, vulnerary, decongestant, and antispasmodic purposes.
2
Recent studies report that B. arborea contains alkaloids such as atropine, scopolamine, hyroscine, hyoscyamine, 3α-tropanol, 3β-tropanol, and cuscohygrine as principal components and has shown anticholinergic activity.
3
-5
Despite these practical uses and pharmacological activities, the constituents of B. arborea flowers are rarely reported. Therefore, this study searched for their active materials. We previously reported the isolation of two benzonitriles from B. arborea flowers.
6
In this continuing study, three rarely occurring monoterpene glucosides (
Brugmansia arborea flowers were extracted in MeOH, and the concentrated extract was partitioned into EtOAc, n-BuOH, and H2O fractions. The EtOAc and n-BuOH fractions were subjected to repeated column chromatography (CC) to yield 3 compounds (
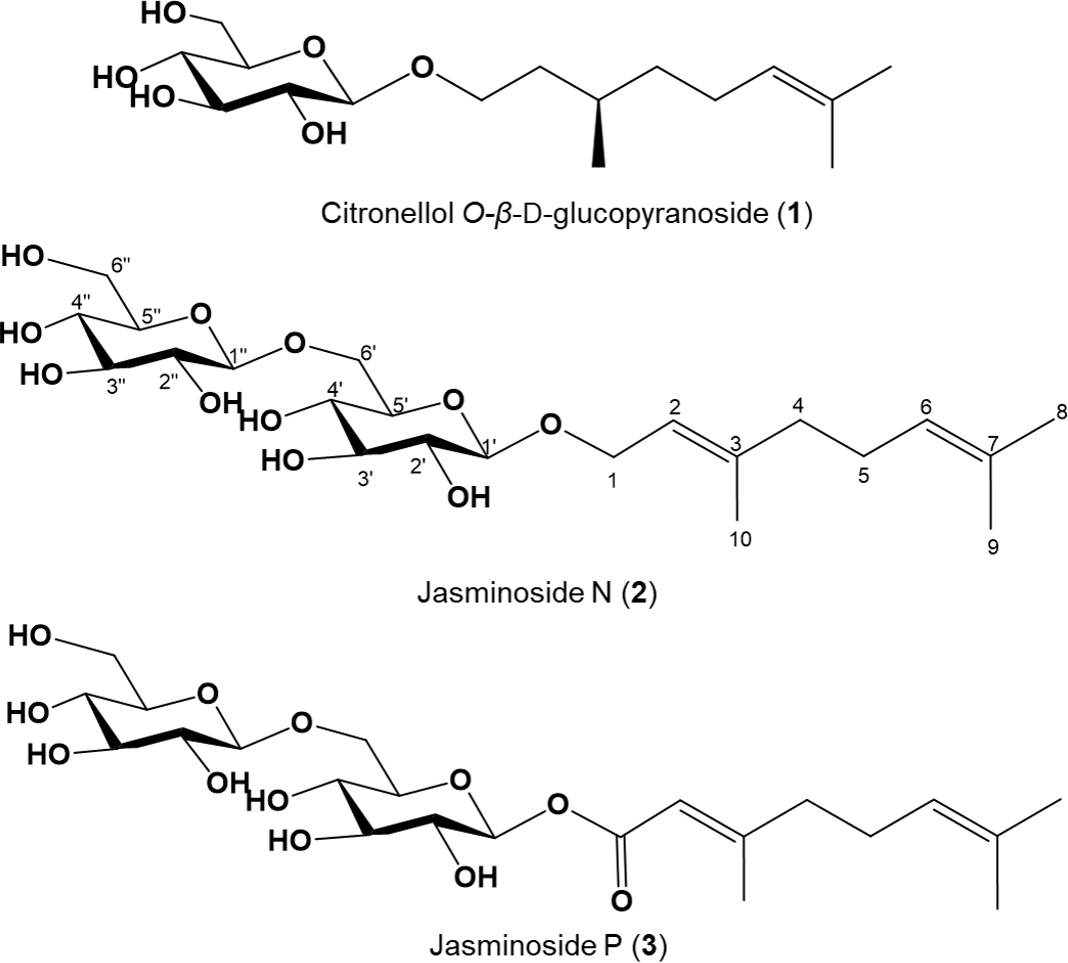
Chemical structures of monoterpene glucosides 1 to
Inhibition of Tyrosinase Activity by EtOAc (BAFE) and n-BuOH (BAFB) Fractions and Isolated Monoterpene Glucosides From Brugmansia arborea Flowers.

All compounds were examined in experiments repeated 3 times.
aConcentration for measuring inhibition ratio on mushroom tyrosinase activity was 200 µM.
bConcentration for measuring inhibitiion ratio on mushroom tyrosinase activity was 100 μg/mL.
cIC50 values are the concentration that caused 50% enzyme activity loss.
Experimental
Plant Materials, Instruments, and Chemicals
Brugmansia arborea flowers were supplied and identified as before. 6 Equipment and chromatographic materials were as previously described. 11,12
Extraction and Isolation
Dried B. arborea flowers (900 g) were extracted with 80% aqueous MeOH (27 L × 3) at room temperature for 24 hours. The concentrated MeOH extract obtained under reduced pressure (160 g) was suspended in water (1 L) and successively extracted with ethyl acetate (EtOAc, 1 L × 3) and n-butanol (n-BuOH, 0.6 L × 3); the extracts were concentrated to produce EtOAc (BAFE, 12 g), n-BuOH (BAFB, 16 g), and H2O fractions (BAFW, 132 g). The EtOAc fraction (12 g) was used for silica gel (SiO2) CC (7.0 cm × 15.0 cm) eluting with n-hexane-EtOAc (4:1 → 2:1 → 1:1, 2.0 L each) and CHCl3-MeOH-H2O (50:3:1 → 36:3:1 → 25:3:1 → 18:3:1 → 12:3:1 → 9:3:1 → 7:3:1 → 6:4:1, 1.5 L each). The eluates were monitored by Thin Layer Chromatography (TLC) to produce 16 fractions (BAFE-1–BAFE-16). BAFE-10 [966 mg, elution volume/total volume (Ve/Vt) 0.649-0.718] was subjected to octadecyl SiO2 (ODS) CC (4.5 cm × 10 cm) eluting with acetone-H2O (1:2 → 1:1, 3.7 L each) to yield 16 fractions (BAFE-10-1–BAFE-10-16). BAFE-10-15 (102.0 mg, Ve/Vt 0.736-0.990) was subjected to SiO2 CC (3.2 cm × 12 cm) eluting with CHCl3-MeOH-H2O (24:3:1, 1.3 L) to yield 14 fractions (BAFE-10-15-1–BAFE-10-15-14). SiO2 CC (1.7 cm × 6 cm) of BAFE-10-15-9 (27 mg, Ve/Vt 0.385-0.500) eluting with CHCl3-MeOH-H2O (27:3:1, 240 mL) produced 12 fractions (BAFE-10-15-9-1–BAFE-10-15-9-12), and purified compound
Tyrosinase Inhibition Assay
Mushroom tyrosinase (EC 1.14.18.1) (Sigma Chemical Co., St Louis, MO, Uited States) was used for in vitro bioassays, as described previously, with some modifications. 13,14 l-3,4-Dihydroxyphenylalanine (l-DOPA) was used as a substrate. Enzyme activity was monitored by detecting dopachrome formation at 475 nm with a UV-Vis spectrophotometer (Spectro UV-Vis Double beam; UVD-3500, Labomed, Inc., Los Angeles, CA, United States) at 30°C. All samples were dissolved in EtOH and used for experiments after dilution. First, 200 mL of 2.7 mM l-DOPA (Km = 180 µM) aqueous solution was mixed with 2687 mL of 0.25 M phosphate buffer (pH 6.8). Then, 100 µL sample solution and 13 µL phosphate buffered solution of mushroom tyrosinase (144 units) were added. Assays were conducted in triplicate. Inhibition concentration leading to 50% activity loss (IC50) was obtained by fitting experimental data to the logistic curve by the following equation: Activity (%) =100[1/(1 + ([I]/IC50))], 15 where [I] is the inhibition concentration.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was carried out with the support of "Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ014204032019)" Rural Development Administration, Republic of Korea.
