Abstract
A new isohopane triterpenoid (
The plant genus Rubia, a member of the Rubiaceae family, contains about 70 species. The roots of Rubia spp. are widely used as a traditional medicine for the treatment of cancer, tuberculosis, rheumatism, contusion, menstrual disorder, liver fluke, and dysentery.
1,2
For decades, phytochemicals of Rubia spp. have attracted attention due to their potent bioactivities. Anthraquinones, naphtoquinones, cyclic hexapeptides, triterpenoids, and lignans have been reported as constituents of this genus. Among them, cyclic hexapeptides, including RA-700, showed anticancer activity
1
and triterpenoids, including 2-deoxyrubianol F, inhibited PDGF-induced proliferation.
3
Rubia akane (Nakai) is widely distributed in East Asia.
4
The roots of R. akane exhibit pharmacological effects, including antiplatelet,
5
antitumor,
6
and antimicrobial
7
activities. Although many bioactive constituents of the genus Rubia have been reported, phytochemical study on R. akane is limited. The objective of this study was to explore small molecule bioactive compounds from R. akane. In this study, a new isohopane tripterpene (
Compound

Chemical structure (a) and key COSY, HMBC (b), and NOESY correlations (c) of 1.
Two known oleanane triterpenoids, 30-norhederagenin (
Effects of Compound 1 on Clotting Times and Bleeding Times
The anticoagulant activities of
Effects of Compound 1 on Thrombin-Catalyzed Platelet Aggregation and Fibrin Polymerization
Incubation of human plasma with
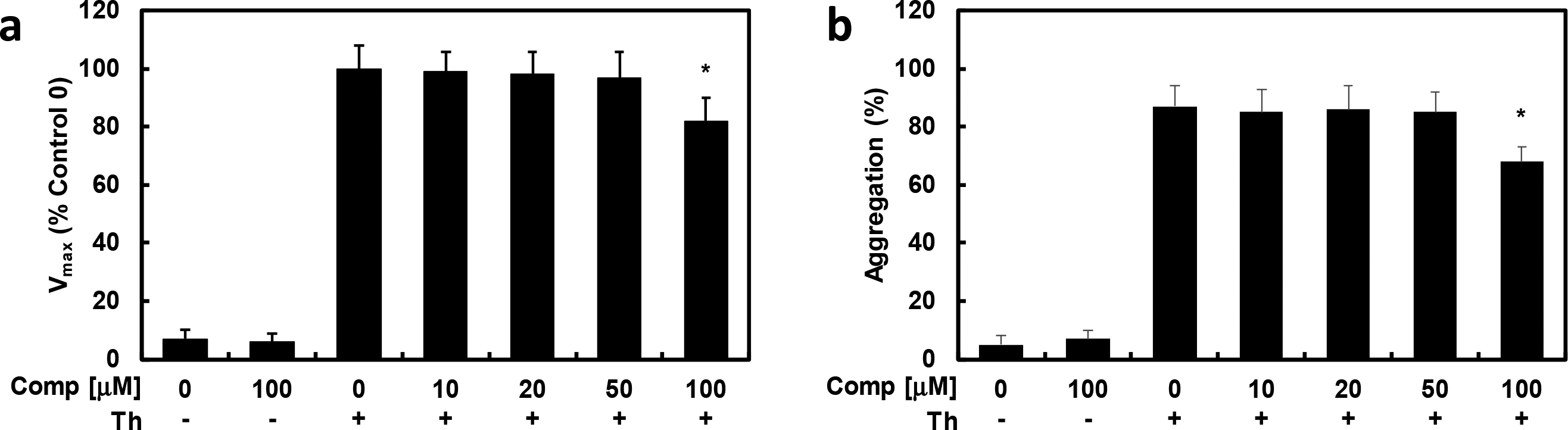
Effects of 1 on fibrin polymerization in human plasma. (a) Thrombin-catalyzed fibrin polymerization at the indicated concentrations of 1. The results are V max values expressed as percentages vs controls. (b) Effect of 1 on mouse platelet aggregation induced by 3 U/mL thrombin. D = 0.2% DMSO is the vehicle control. Data represent the mean ± SEM of 3 independent experiments performed in triplicates. *P < 0.05 vs Th alone.
Effects of Compound 1 on the Activities of Thrombin and FXa
In order to elucidate the mechanism responsible for the inhibition of coagulation by

Effects of 1 on inactivation and production of thrombin and factor Xa. (a) Inhibition of thrombin by 1 was measured using a chromogenic assay. (b) Inhibition of factor Xa by 1 was monitored using a chromogenic assay. Argatroban (a) or rivatoxaban (b) was used as positive control. D = 0.2% DMSO is the vehicle control. *P < 0.05 vs 0.0001 (a, b).
Effects of Compound 1 on Secretion of PAI-1 or t-PA Protein
In order to determine the direct effects of

Effects of 1 on secretion of plasminogen activator inhibitor type 1 and tissue-type plasminogen activator. (a) Human umbilical vein endothelial cells were cultured with 1 in the absence or presence of tumor necrosis factor-α (10 ng/mL) for 18 h and plasminogen activator inhibitor type 1 concentrations in culture media were determined. (b) Human umbilical vein endothelial cells were cultured with 1 in the absence or presence of tumor necrosis factor-α (10 ng/mL) for 18 h and tissue-type plasminogen activator concentrations in culture media were determined. (c) Plasminogen activator inhibitor type 1/tissue-type plasminogen activator ratio in tumor necrosis factor-α activated human umbilical vein endothelial cells from (a) and (b). D = 0.2% DMSO is the vehicle control. *P < 0.05 vs tumor necrosis factor-α alone or D; n.s., not significant.
The genus Rubia has been studied extensively, and there are several reports on the chemical constituents of R. akane and their bioactivities. Anthraquinone, lignan, and cyclopeptide have been isolated from R. akane. The arborinane- and oleanane-type triterpenoids, such as rubiarbonol D, rubiprasin A, and rubiprasin B, were discovered from the species.
2
However, isohopane triterpenoid is the first report on the chemical constituent in R. akane. In this study, we demonstrated the antithrombotic, anticoagulant, and antiplatelet effects of new isohopane triterpenoid
According to the previous studies, antiplatelet activity by thrombin, ADP, and PAF was confirmed with anthraquinones isolated from R. akane.
5
In the current study, a new isohopane triterpenoid
Experimental
General
IR spectrum was recorded on a Bruker ALPHA FT-IR spectrometer. NMR experiments were conducted using a Bruker DMX 300 (1H-300 MHz, 13C-75 MHz) and Bruker DMX 600 (1H-600 MHz, 13C-150 MHz) spectrometers (Karlsruhe, Germany). Optical rotations were recorded using a JASCO DIP-1000 (Tokyo, Japan) and mass spectrometric data were obtained utilizing a SYNAPT G2 Waters mass spectrometer (Manchester, United Kingdom). Vacuum-liquid chromatography (VLC) was performed using silica gel (Merck, 70-230 mesh). MPLC was carried out employing Biotage Isolera reversed phase C18 SNAP Cartridge KP-C18-HS and normal phase SNAP Cartridge KP-Sil (Biotage AB, Uppsala, Sweden). HPLC separation was performed using a Gilson system with a UV detector and Phenomenex C18 column (250 × 21.2 mm, 5 µm). TLC was performed on glass plates precoated with silica gel 60 F254 and RP-18 F254 (Merck).
Plant Material
The roots of Rubia akane were purchased from Korea traditional medicine market (Daejeon, Korea) in 2017. A voucher specimen (CNU 20170726) was deposited at the Pharmacognosy Laboratory of the College of Pharmacy, Chungnam National University (Daejeon, Korea).
Extraction and Isolation
The roots of R. akane (4.9 kg) were extracted with EtOH (8 L × 3) at room temperature for 6 days. The EtOH extract (158.2 g) was suspended in H2O and partitioned with CH2Cl2. The CH2Cl2 fraction (78.9 g) was subjected to silica gel VLC and eluted with n-hexane/EtOAc (10:0, 5:1) and CHCl3/MeOH (40:1, 20:1, 10:1, 5:1, and 0:10) to obtain 6 fractions (MC1-MC6). Fr. MC4 (9.8 g) was further divided into 9 fractions (MC4-1-MC4-9) using MPLC (C18 SNAP Cartridge KP-C18-HS, 400 g) with MeCN/H2O (37:63, 37:63 → 100:0). Fr. MC4-7 (404.8 mg) was applied to C18 HPLC with isocratic MeOH/H2O (78:22) to afford
3β-Hydroxy-16-Oxo-21β-Isohop-22(29)-En-24-Oic Acid (1)
White amorphous solid.
FT-IR (ATR) Vmax : 3319, 2943, 2831, 1450, 1415, 1020 cm−1.
UV/Vis λmax (MeOH) nm (log ε): 200 (3.89).
1H NMR (600 MHz, pyridine-d 5): 4.82 (2H, d, J = 16.93 Hz, H2-29), 4.67 (1H, t, H-3), 3.00 (1H, m, H-21), 2.64 (1H, d, J = 11.51 Hz, H-17), 2.50 (1H, d, J = 12.42 Hz, H2-15), 2.20 (1H, m, H-13), 1.92 (1H, d, J = 12.29 Hz, H-5), 1.86 (1H, d, J = 12.42 Hz, H2-15), 1.75 (3H, s, H3-30), 1.72 (1H, m, H2-1), 1.66 (3H, s, H3-23), 1.55 (1H, m, H2-19), 1.40 (1H, m, H2-19), 1.36 (1H, m, H-9), 1.15 (1H, m, H2-1), 1.06 (3H, s, H3-26), 0.97 (3H, s, H3-27), 0.94 (3H, s, H3-25), 0.72 (3H, s, H3-28)
13C NMR (150 MHz, pyridine-d 5): 210.7 (C-16), 181.3 (C-24), 148.2 (C-22), 109.5 (C-29), 75.8 (C-3), 65.6 (C-17), 54.9 (C-4), 52.6 (C-18), 51.9 (C-5), 50.7 (C-8, 9, 15), 49.3 (C-13), 43.2 (C-14), 42.3 (C-21), 41.1 (C-19), 39.6 (C-1), 37.1 (C-10), 33.7 (C-7), 28.3 (C-11, 20), 24.1 (C-2), 22.0 (C-6), 21.9 (C-30), 21.4 (C-12), 17.9 (C-27), 17.5 (C-26), 16.7 (C-25, 28), 12.4 (C-23)
HRESIMS: m/z 493.3291 [M+Na]+ calcd for C30H46O4Na: 493.3294
Biological Section
Detailed methods for the biological evaluation in this study are provided in Supplemental data.
Supplemental Material
Supplemental data - Supplemental material for Antithrombotic and Antiplatelet Activities of New Isohopane Triterpene From the Roots of Rubia akane
Supplemental material, Supplemental data, for Antithrombotic and Antiplatelet Activities of New Isohopane Triterpene From the Roots of Rubia akane by InWha Park, Wonhwa Lee, Hyelim Kim, Khong Trong Quan, DaYoung Kim, Jong-Sup Bae, and MinKyun Na in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research fund from Chungnam National University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
