Abstract
In this paper, the insecticidal activity of natural capsaicinoids (NC) and reference ingredients (imidacloprid, abamectin, and pyridaben, which are active ingredients of commercial pesticides) against 14 agricultural insects was studied in both the laboratory and field. The results of insecticidal activity in the laboratory showed that the median lethal concentration (LC50) and 90% lethal concentration values of the NC against 14 agricultural insects were greater than those of reference ingredients and the NC had a weaker relative toxicity than the corresponding reference ingredients. The NC showed impressive insecticidal activity when it was used to control Aphis gossypii from Cucumis sativus (LC50 152.82 mg L−1) but unimpressive insecticidal activity when used against Ectropis obliqua hypulina and Pieris rapae (LC50 1557.47 and 1502.48 mg L−1, respectively), which revealed that the insecticidal spectrum of NC was confined. Field experiments showed that the control effect of successively spraying the NC 2 times against insects was significantly higher than the effect of spraying 1 time.
Capsicum annuum L., known as chili pepper, belonging to the Solanaceae family, originates from tropical America. 1 Chili peppers are one of the major vegetable and spice crops cultivated worldwide. Thailand, India, China, Pakistan, and Peru are the largest dried chili producers in the world. 2 Chili peppers are popular around the world and are often used as food additives to provide a hot and pungent taste. The extremely hot or burning sensation of chili peppers is due to the presence of capsaicinoids found only in the Capsicum genus, which are biosynthesized in the placenta of the fruits by condensation of vanillylamine and medium chain length fatty acids. 3,4 The main components of capsaicinoids are capsaicin, dihydrocapsaicin, nordihydrocapsaicin, homocapsaicin, and homodihydrocapsaicin, 5 chemical structures of which are shown in Figures 1–5. Among these components, capsaicin and dihydrocapsaicin contribute approximately 80% to 90% of the total pungency in most chili peppers. 6 In addition to the above-mentioned capsaicinoids, approximately 20 other capsaicinoids have been found in different chili pepper species. 7
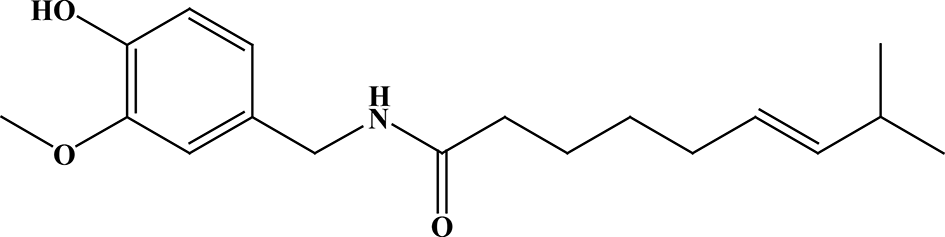
Chemical structure of capsaicin.
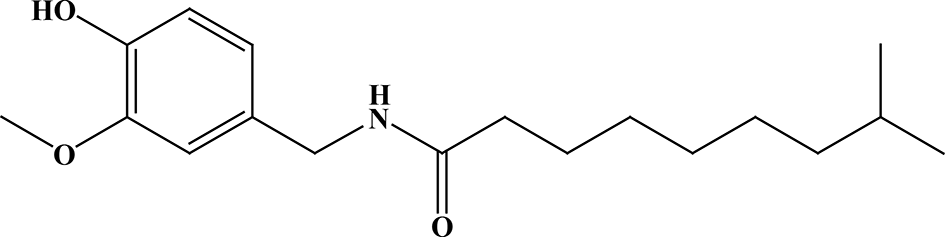
Chemical structure of dihydrocapsaicin.
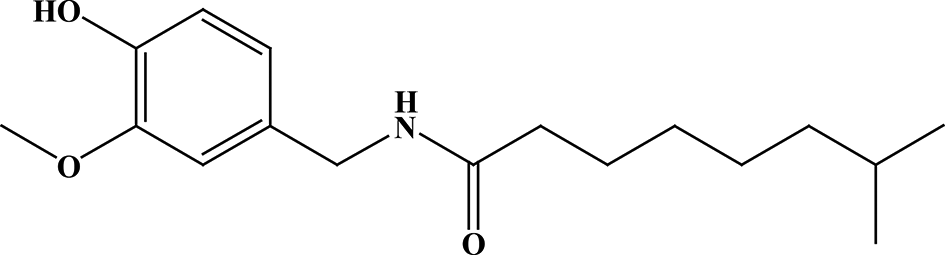
Chemical structure of nordihydrocapsaicin.

Chemical structure of homodihydrocapsaicin.

Chemical structure of homocapsaicin.
The carcinogenicity of pesticides has been well documented, and there are many reports that have linked exposure to various synthetic insecticides, herbicides, and fungicides to various types of cancers. 8 Chemical pesticides lead to poisonings in approximately 3 million people and 200 000 deaths each year worldwide. Such cases are reported more in developing countries (95%) than in developed countries. 9 According to reports, capsaicin has broad-spectrum insecticidal activity against many species of insects, eg, stored product beetles (Sitophilus zeamais and Tribolium castaneum), 10 rice grain insects (Sitotroga cerealella), 11 Alfalfa weevil, 12 Myzus persicae, 13 Bemisia tabaci, 14 and Plutella xylostella. 15,16
In light of the above information, the present study was conducted to investigate the effect of natural capsaicinoids (NC) against 14 agricultural insects as a part of the search for alternative natural products. The insecticidal activities of NC and pesticide components against 14 insects were studied in the laboratory.
The results (Table 1) showed that NC have certain insecticidal activity to the tested pests, but the sensitivity of various insects to NC is different. The toxicity regression equations of different component dilutions against tested pests were noteworthy according to their significant values, indicating that the fitting degree of each toxicity regression equation was high. According to the median lethal concentration (LC50) value, the order of insecticidal activity of NC against the tested insects is as follows: Aphis gossypii (from Cucumis sativus) >Tetranychus viennensis >Tetranychus cinnabarinus >Aphis gossypii (from Gossypium spp.) >Aphis citricola >Lipaphis erysimi >Panonychus ulmi >Brevicoryne brassicae >Myzus persicae >Aphis craccivora >Plutella xylostella >Tetranychus truncates >Ectropis obliqua hypulina >Pieris rapae. Aphis gossypii from C. sativus are sensitive to NC, but P. xylostella, E. obliqua hypulina, P. rapae, and T. truncates are insensitive. The LC50 and 90% lethal concentration (LC90) values of NC against A. gossypii (from C. sativus) were 152.82 and 769.52 mg L−1, and of imidacloprid was 1.21 and 21.92 mg L−1, respectively. The relative toxicity index of NC to imidacloprid against A. gossypii (from C. sativus) was 0.794, indicating that the insecticidal activity of NC against A. gossypii (from C. sativus) was much lower than that of imidacloprid. The LC50 and LC90 values of NC against T. truncatus were 1019.98 and 19519.22 mg L-1, and of pyridaben were 0.36 and 6.06 mg L−1, respectively. The relative toxicity index of NC to pyridaben against T. truncatus was 0.036, indicating that the insecticidal activity of NC against T. truncatus was much lower than that of pyridaben. The LC50 values of NC against P. xylostella, E. obliqua hypulina, and P. rapae were 838.47, 1557.47, and 1502.48 mg L−1, and the LC90 values were 10863.64, 17682.66, and 16844.56 mg L−1, respectively. The LC50 values of abamectin against P. xylostella, E. obliqua hypulina, and P. rapae were 3.87, 9.88, and 6.648 mg L− 1, respectively, which were much lower than that of NC, indicating that the insecticidal activity of NC was lower than abamectin. Similarly, the LC50 and LC90 values of NC against the other tested insects were greater than imidacloprid, pyridaben, and abamectin, indicating that the insecticidal activity of NC was lower than the components of the pesticides. As shown in Table 1, the LC50 values of NC against A. gossypii (from C. sativus) and A. gossypii (from G. ossypium spp.) were 152.82 and 230.89 mg L−1, and the LC90 values were 769.52 and 813.07 mg L−1, respectively. The LC50 values of imidacloprid against A. gossypii (from C. sativus) and A. gossypii (from G. ossypium spp.) were 1.21 and 1.02 mg·L− 1, and the LC90 values were 21.92 and 15.25 mg L−1, respectively. This indicated that the sensitivity of the same aphid species (A. gossypii) from different host plants (cucumber and cotton), in response to NC and imidacloprid, was different. In other words, the tolerance of A. gossypii in different growth environments on experimental components was different.
Analysis of Insecticidal Activity of NC and Pesticide Components Against the Tested Insects.

Note: The test times for LC50 and LC90 of P. xylostella, P. rapae, and E. obliqua hypulina were for 48 hours, but for the other tested agricultural insects were for 24 hours.
a A.gossypii from C.sativus.
b A.gossypii from Gossypium spp.,
Table 1 reveals that the insecticidal activity of NC is lower than that of imidacloprid, pyridaben, and abamectin. NC can be made into dosage forms with synergists to improve their insecticidal activities.
NC and imidacloprid were sprayed 1 time in the field, with the purpose of controlling A. citricola, Corythucha ciliata, B. brassicae, and M. persicae. As shown in Table 2, the time of the insect deaths was different due to different sensitivities to the pesticides. After spraying on the third and seventh days, the control effect of imidacloprid against A. citricola was more than 97%, which was significantly higher than that of NC. At the same time, the higher the concentration of NC is, the better the control effect, and the control effect of NC against A. citricola after spraying on the seventh day was less than that of the third day. The control effect of NC against C. ciliata was 89.07% and 86.24% on the 1st day and 11th day after spraying, respectively, which revealed that C. ciliata was sensitive to capsaicinoids. The control effects of 50 mg L−1 NC against C. ciliata on the 1st, 2nd, and 11th days after spraying in the field were significantly lower than the control effects of the same concentrations of imidacloprid. However, the differences in their control effects were inconspicuous between 300 mg L−1 of NC and 50 mg L−1 of imidacloprid against C. ciliata in the field, revealing that the capsaicinoids at high concentrations have similar control effects to imidacloprid against C. ciliata in the field. The high capsaicinoid concentration could replace chemical pesticides for controlling insects in the field. For B. brassicae and M. persicae, the higher the concentration of the capsaicinoids, the better the control effect. On the 3rd, 7th, and 11th days after spraying, the control effects of 100 mg L−1 NC against B. brassicae were 53.94%, 74.11%, and 65.86%, respectively, and of 3000 mg L−1 NC were 74.38%, 88.51%, and 77.80%, which was significantly lower than that of 50 mg L−1 imidacloprid. However, the control effects of 1000 mg L−1 NC against M. persicae on the 3rd, 7th, and 11th days were 92.52%, 98.52%, and 89.74%, respectively, which were not significantly different from 50 mg L−1 imidacloprid. The control effect of 200 mg L−1 NC against M. persicae on the 7th day was 91.20% and on the 11th day was 70.97%.
Control Effect of Pesticides Against Insects in the Field After Spraying 1 Time.

Data followed by different lowercase letters in the same column for the same insect show significant differences by ANOVA followed by Tukey’s test at the 5% level of significance, but different capital letters show significant differences at the 1% level of significance.
Based on the abovementioned findings, the control effects of NC have timelines in the field, where on the 7th day, the values were lower than those on the 3rd day against A. citricola, and the 11th day value was lower than that on the 7th day against B. brassicae and M. persicae. These results revealed that the insecticidal efficacy of capsaicinoids gradually decayed after 1 week in the field.
Therefore, to achieve a high control effect, it is necessary to repeatedly spray NC in the field, as shown in Table 3, which shows the control effects of NC on P. xylostella and T. cinnabarinus in the field after spraying NC 1 and 2 times.
Control Effect of Pesticides Against Insects in Field After Spraying 2 Times.

Data followed by different lowercase letters in the same line show significant differences by ANOVA followed by Tukey’s test at the 5% level of significance, but different capital letters show significant differences at the 1% level of significance.
The control effect of spraying NC 1 time on the second day was lower than that on the seventh day, and on the ninth day, the effect was lower than that on the seventh day. However, the control effect on the ninth day was higher than that on the seventh day when the second NC spray occurred on the third day after the first spray. As showing in Table 3, 2 consecutive sprays of 1000 or 3000 mg L− 1 NC in the field control effects on P. xylostella and T. cinnabarinus gradually increased with the passage of time within the test time range. The control effects of spraying twice at either 1000 or 3000 mg L− 1 NC were 94.24% and 99.78%, respectively, which were significantly higher than spraying only once (the control effects were 80.16% and 62.09%, respectively). Analogously, the control effects of spraying 1000 or 3000 mg L−1 NC 2 times (41.84%, 48.15%) were significantly higher than spraying 1 time (5.32%, 15.12%). The results of Table 3 revealed that NC had a durable pesticide effect by spraying 2 times successively.
The insecticidal activity of NC against the 14 tested agricultural insects in both the laboratory and field was weaker than that of the corresponding chemical pesticides, but increasing the concentration of NC and the number of sprayings can effectively control the tested agricultural insects. In the absence of toxicological concerns from the ingestion of capsaicin because of its presence in the human diet, the Environmental Protection Agency (EPA) has waived the requirements for the submission of residue data. Therefore, no ecological effects or environmental fate data are required to support the reregistration of capsaicin. As a natural and pollution-free active ingredient of insecticides, capsaicin could be manufactured as part of green environment-friendly insecticides. In other words, insecticide containing natural capsaicin is a harmless biological pesticide, and it is possible for NC to replace some of the chemical pesticides, thus reducing the environmental pollution caused by chemical pesticides.
Experimental
Capsaicinoid crystals (96.3%, including 59.8% of capsaicin and 26.9% of dihydrocapsaicin) were supplied by Yunnan Yuesheng Agricultural Science and Technology Co., Ltd, while imidacloprid (95.3%), abamectin (93.0%), and pyridaben (97.1%) were provided by the Institute for the Control of Agrochemicals, MOA. Capsaicinoid crystals, imidacloprid, abamectin, and pyridaben were dissolved in DMF to make up their mother solutions with 10 g L− 1 active ingredients and then diluted using distilled water with 0.05% Triton X-100 to a certain concentration.
The insecticidal activity of the dilutions against adult aphids (A. craccivora, A. citricola, B. brassicae, M. persicae, A. gossypii from C. sativus, A. gossypii from Gossypium spp., L. erysimi, which belong to Homoptera insects) and adult mites (T. truncatus, P. ulmi, T. cinnabarinus, T. viennensis, which belong to Acarina insects) was tested with the immerse insects method. The definite means are as follows: Host plant leaves with 50 corresponding insects were immersed in the dilution, and the leaves were removed after jiggling for 5 seconds; filters were used to absorb excess dilution on the leaves, then the leaves were placed in Petri dish with moist blotting paper; the petri dish was covered with a breathable cling film and placed in an artificial climate box under 25°C ± 1℃. There were 3 replicates in each treatment and 50 insects in each repeat experiment; the blotting paper was moistened with 0.05% Triton X-100 as a CK. Twenty-four hours later, the tested insects were observed under an anatomical mirror; the bodies of the insects were touched with a brush tip, and insects with no response or nonnormal crawling were regarded as dead.
The insecticidal activities of the dilutions against third instar larvae Lepidoptera insects (P. xylostella, P. rapae, E. obliqua hypulina) were tested with the immerse leaves method. The definite means are as follows: Fresh tea shoots and collard leaves were soaked in a dilution for 5 seconds, the treated tea shoot was naturally air dried, and the shearing surface was wrapped in moist cotton. Tea shoots with pesticides and 50 E. obliqua hypulina were placed in 180 mm × 18 mm tubes, sealed with cotton plugs, and placed in an artificial climate box at 25°C ± 1°C. The collard leaves with dilution and 50 P . xylostella or P. rapae were placed in a 250 mm × 180 mm ×100 mm plastic container, closed with a lid with air vents, and placed in an artificial climate box at 25°C ± 1℃. There were 3 repetitions in each treatment, and each was watered with 0.05% Triton X-100 as a CK. After 48 hours, the tested insects were observed under an anatomical mirror, touching the bodies of the insects with a brush tip, and insects with no response or nonnormal crawling were regarded as dead.
Aphis citricola on apple tree, C. ciliata on sycamore, B. brassicae on broccoli, and M. persicae on tobacco were managed via spraying diluted pesticides 1 time and spraying water as a CK, and each spray was applied in 3 repetitions. The number of individual A. citricola was checked on the third and seventh days, before and after spraying diluted pesticides and water. The number of individual C. ciliata was checked on the 1st, 2nd, and 11th days, before and after spraying. The number of individual B. brassicae and M. persicae was checked on the 3rd, 7th, and 11th days, before and after spraying.
Tetranychus cinnabarinus on mulberry and P. xylostella on cabbage were sprayed with diluted NC 2 times (the second spray occurred on the 15th day after the first spray) and water as a CK, and each spray was applied in 3 repetitions. The individual numbers of T. cinnabarinus and P. xylostella were checked half an hour before the second spraying and on the second, third, fifth, seventh, and ninth days after spraying.
According to the mathematical characteristics of Equations (1)-(4), the mortality ratio, adjusted mortality ratio, relative toxicity, and control efficiency were counted using Microsoft Office Excel 2007. The LC50 and LC90 with their confidence intervals (P < 0.05) were analyzed using the probit method described by Jia 17 using the Statistical Package for the Social Sciences (SPSS) 17.0.

where D is the mortality (%), N d is the number of dead insects mouths, N t is the number of mouths.
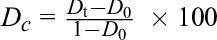
where D c is the correction of mortality (%), D t is the mortality rate of the experimental group (%), and D 0 is the death rate in the control group (%).

where T is the relative toxicity index, LC50C is the LC50 of the contrast insecticide (mg L− 1), and LC50NC is the LC50 of NC (mg L− 1).
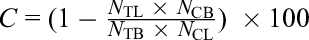
where C is the control effect, N TL and N CL are the number of viable individuals in the tested and control groups, and N TB and N CB are the initial population numbers of the tested and control groups.
Footnotes
Acknowledgments
We are grateful to the anonymous reviewers for their constructive comments and thoughtful suggestions on the manuscript. Our gratitude also goes to Dr Zhi Xiong for his kind, invaluable suggestions and copyediting of the manuscript. We apologize to all our colleagues whose studies could not be cited due to space limitations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Key R&D Program of China (Grant No. 2018YFD0200500 and 2018YFD0200300).
