Abstract
A new aurone (
Diacylglycerol acyltransferase (DGAT) is a key enzyme in triacylglycerol (TG) synthesis, which catalyzes the final step of the TG synthesis pathway. 1 There are 2 isoforms of DGAT including DGAT1 and DGAT2, and lots of studies showed that DGAT1 is responsible for the synthesis of TG. 2 Excessive accumulation of TG can lead to obesity. Therefore, inhibiting the activity of DGAT1 is a promising therapy to treat obesity.
Psoralea corylifolia L. (Leguminosae), an important Chinese folk medicine, is distributed widely in Southeast Asia. The seeds of P. corylifolia are traditionally used to treat bacterial infections, 3 osteoporosis, 3 asthma, 4 spermatorrhea, 3 and gynecological bleeding. 5 Published studies have shown that P. corylifolia has a large number of biological constituents including flavonoids, 6 meroterpenes, 7 and coumarins 8 with reported biological activities such as antioxidant, 9 anti-inflammatory, 10 and antibacterial 11 properties.
During the course of our search for DGAT inhibitors from P. corylifolia, a new aurone was isolated from the EtOAc extract of the seeds of P. corylifolia, together with 5 known compounds (
Compound
NMR Data of Compound 1 in Acetone-D 6 (1H: 500 MHz, 13C: 125 MHz).


The key HMBC correlations of compound 1.
Moreover, 5 known compounds (

Structures of compounds 1 to
All the isolates were evaluated for their inhibitory activity against DGAT1 and DGAT2, and the known DGAT inhibitor, kuraridine, was used as the positive control.
16
The result showed 6 compounds inhibited DGAT1 activity with IC50 values ranging from 54.9 ± 1.4 to 124.3 ± 1.1 µM (Table 2). Among these isolates, compound
Inhibitory Effects of Compounds 1 to
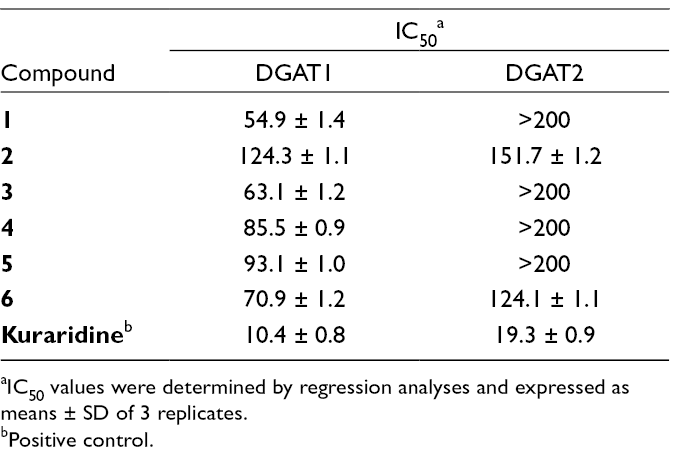
aIC50 values were determined by regression analyses and expressed as means ± SD of 3 replicates.
bPositive control.
Experimental
General Experimental Procedures
Optical rotations were determined on a Horiba SEPA-300 polarimeter (Horiba Scientific, Kyoto, Japan). UV spectra were recorded in MeOH using Shimadzu UV-2401PC spectrophotometer (Shimadzu, Tokyo, Japan). IR spectra were recorded with a JASCO FT-IR 620 spectrophotometer. NMR spectra were obtained from Bruker Avance-III 600, DRX-500, and AM-400 MHz spectrometers (Bruker BioSpin GmBH, Rheinstetten, Germany) using TMS as the internal standard. Mass spectra were obtained on a QTOF2 high-resolution mass spectrometer (Micromass, Wythenshawe, UK). Column chromatography was conducted using silica gel 60 (200 μm particle size, Yantai Xinde Chemical Co., Ltd, Yantai, China) and RP-18 (20, 45 μm particle size, Fuji Silysia Chemical Ltd., Kasugai, Japan). For thin-layer chromatography (TLC), precoated TLC silica gel 60 F254 plates from Merck were used. HPLC was carried out using a Waters 2535Q HPLC system fitted with a 2998 photodiode array detector (Waters, USA).
Plant Material
The seeds of P. corylifolia were collected in Guangyuan, Sichuan province, Republic of China, and authenticated by Professor Lei Zhang (College of Pharmacy, Sichuan University). A voucher specimen of the plant (No. 20160425) was deposited at Chengdu University of Traditional Chinese Medicine, Sichuan, China.
Extraction and Isolation
The dried seeds of P. corylifolia (5.0 kg) were extracted with 95% EtOH aqueous at room temperature for 3 days and then concentrated under vacuum at 45°C to obtain a residue (812.3 g). This extract was suspended in H2O, partitioned successively with petroleum ether, EtOAc, and n-BuOH to afford petroleum ether-, EtOAc-, and n-BuOH-soluble fractions, respectively. Part of the EtOAc-soluble fraction (100.0 g) was chromatographed over a silica gel column using a gradient of CH2Cl2/MeOH (from 0:1, 200:1, 100:1, 50:1, 25:1, 10:1, 5:1, 3:1 to 0:1) and was separated into 18 fractions (Fr.1-Fr.18). Fr.6 (12.1 g) was subjected to silica gel column eluted with CH2Cl2-MeOH (from 0:1, 100:1, 50:1, 10:1, 5:1 to 0:1) to get 8 fractions (Fr.6.1-Fr.6.8). Fr.6.5 (3.5 g) was subjected to RP-18 column (40.0 × 3.5 cm) and eluted with MeOH/H2O (3:7 to 10:0) to afford 10 fractions. Further purification of Fr.6.5.3 (851.2 mg) was subjected to RP-18 column, eluted with a stepwise gradient of MeOH/H2O (4:6 to 6:4) to afford 12 subfractions (Fr.6.5.3.1-Fr.6.5.3.12). After that, Fr.6.5.3.5 (120.1 mg) was separated by HPLC, using a gradient solvent system 50% to 70% MeOH in H2O over 80 minutes, yielded compounds
(2Z)-2-[(3′,4′-Dihydroxyphenyl)methylene]-6-Methoxy-7-Prenyl-3(2H)-Benzofurane (1)
Yellow amorphous powder.
IR (KBr) ν max: 3415, 1752, 1658, 1604 cm−1.
UV (MeOH) λ max (nm) (log ε): 255 (4.01), 397 (4.38).
1H (500 MHz) and 13C NMR (125 MHz) in acetone-d 6: Table 1.
HREIMS m/z: 352.1307 [M]+ (calcd for C21H20O5, 352.1311).
DGAT1 and DGAT2 Assays
DGAT activity assays were carried out as described previously. 17 Briefly, DGAT activity in total membranes prepared from DGAT2- or DGAT1-overexpressing Sf-9 and HEK293 Tet-on cells was determined by measuring the formation of [14C]-TG from [14C]-oleoyl CoA. The reaction mixture for the DGAT1 assay contained 175 mM Tris (pH 7.5), 100 mM MgCl2 (5 mM MgCl2 for DGAT2 assay), 200 µM sn-1,2-diacylglycerol, 20 µM [1-14C]-oleoyl CoA (5.5 µCi), 2 mg/mL BSA, and 32 µg of the membrane protein. The mixture was incubated for 20 minutes at 37°C and then the reaction was stopped by the addition of 1.5 mL of stop solution [2-propanol-heptane-water (80:20:2, v/v/v)] and vortexed with 1.0 mL of heptane and 0.5 mL of water. The top heptane phase was collected and washed with 2.0 mL alkaline ethanol solution [ethanol-0.5 N NaOH-water (50:10:40, v/v/v)]. The radioactivity of the top phase was determined by liquid scintillation counting (ALOKA LSC-8000, Aloka, Inc., Tokyo, Japan).
Supplemental Material
Supporting Information - Supplemental material for A New Aurone and Other Constituents From the Seeds of Psoralea corylifolia With Their Diacylglycerol Acyltransferase Inhibitory Activity
Supplemental material, Supporting Information, for A New Aurone and Other Constituents From the Seeds of Psoralea corylifolia With Their Diacylglycerol Acyltransferase Inhibitory Activity by Bo Ren, Jia-Yue Dong, and Ying Liu in Natural Product Communications
Footnotes
Acknowledgments
We thank Dr Lihua Wang and Dr Xin Li in the Research Center for Chemistry, Sichuan University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
