Abstract
A new cytisine-like alkaloid, hosimonoal (
Ormosia hosiei Hemsl. et Wils. (Leguminosae), as an ornamental plant, is mainly distributed in the Zhejiang, Fujian, and Sichuan Provinces of China.
1
Its seeds, known as “hongdou,” are used for the treatment of hernia, bellyache, and amenorrhea.
2
The previous phytochemical studies of Ormosia species have resulted in the isolation of alkaloids,
3
-5
triterpenoids,
6
and isoflavonoids,
7
-9
especially the alkaloids, known as “Ormosia alkaloids,”
5
which showed excellent biological activities. However, few phytochemistry studies have been carried out on O. hosiei, and only 4 cytisine-like alkaloids have been isolated from its roots and stems.
10
In order to study any novel alkaloids from O. hosiei, we conducted a phytochemical study of its seeds which resulted in the isolation of 3 alkaloids (
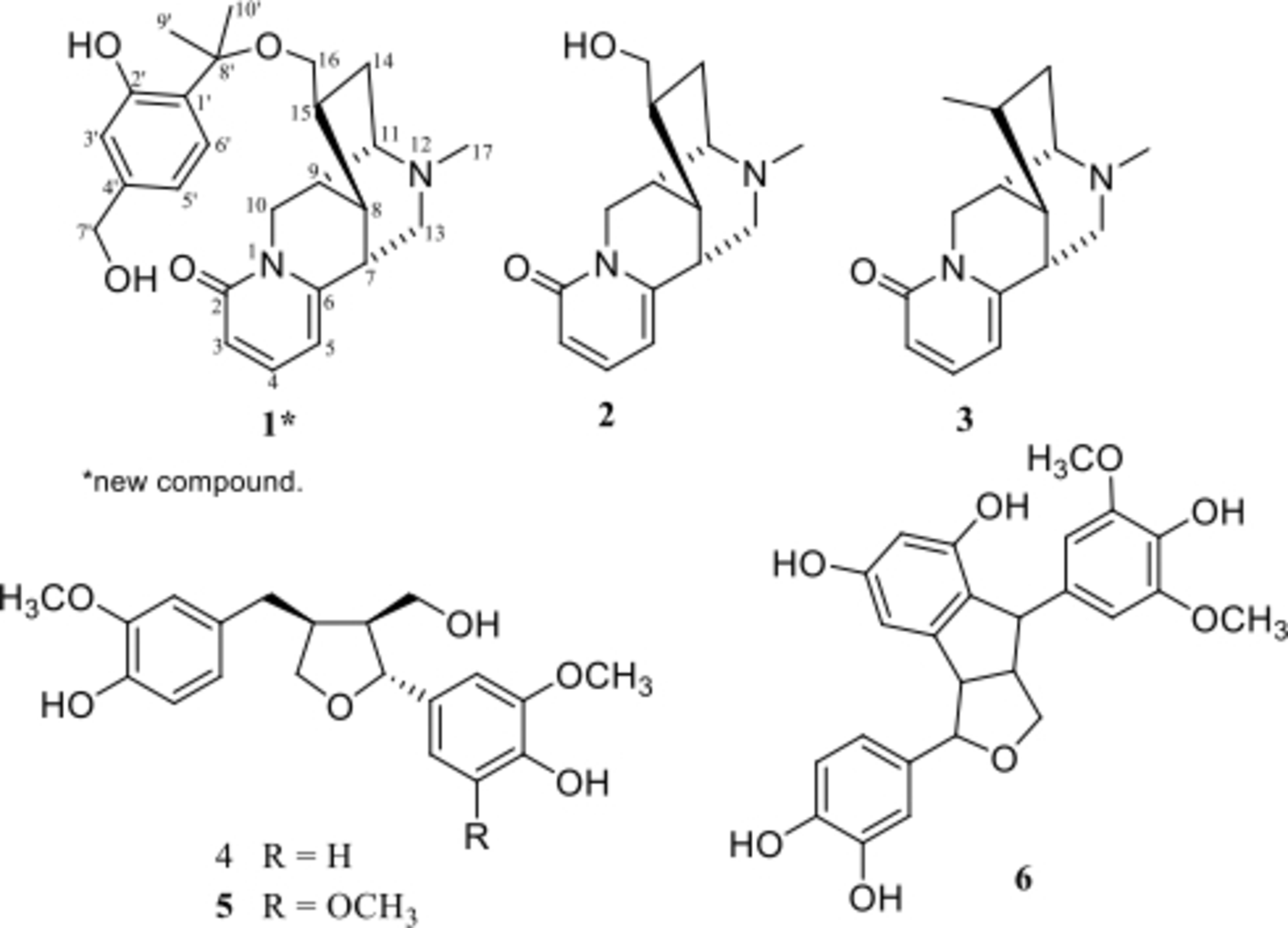
The structures of compounds 1 to
Hosimonoal (
1H and13C NMR Spectroscopic Data for Compound 1.

aIn methanol-d 4 (100 MHz).
bIn methanol-d 4 (400 MHz).
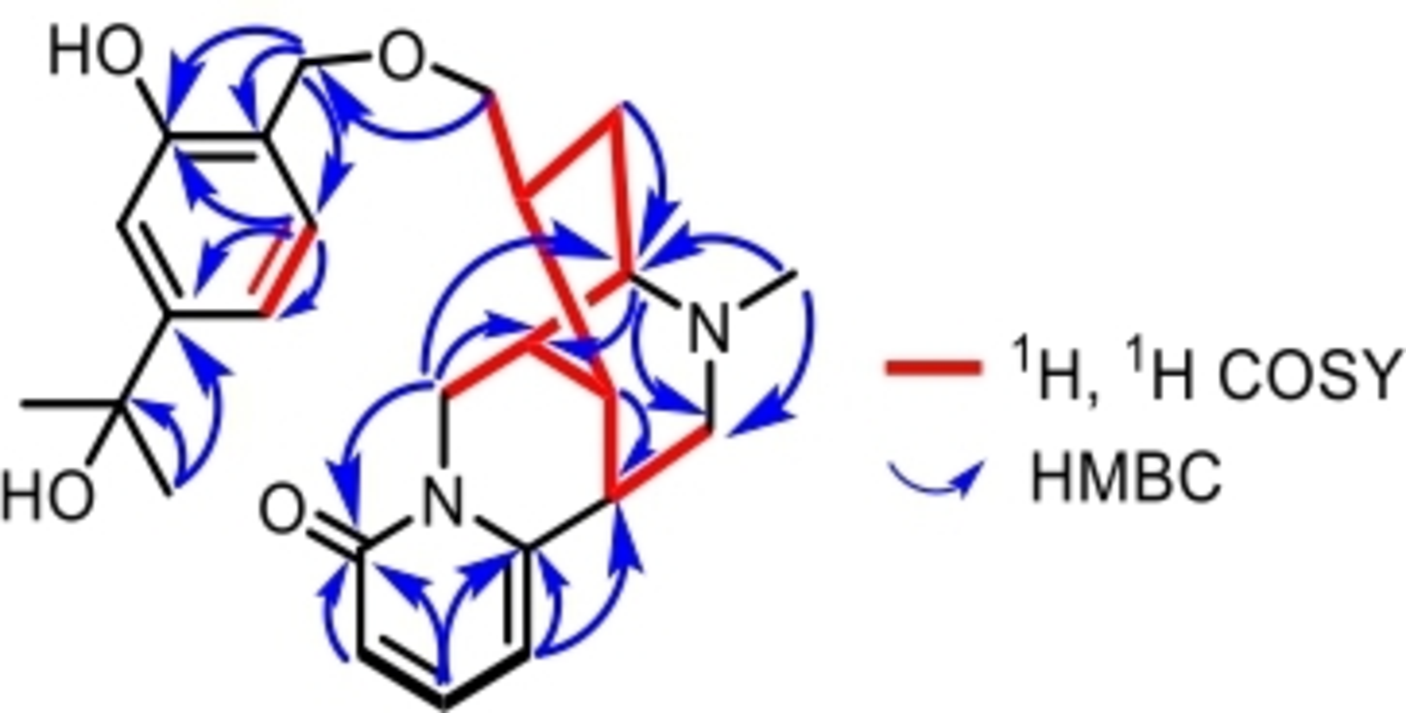
The key 1H,1H COSY and HMBC correlations of compound 1.

The nuclear overhauser effect spctroscopy correlations of compound 1.
The hosieine B unit was determined by 1H, 1H-correlation spectroscopy (1H COSY) correlations of H-3/H-4/H-5, H-7/H-13, H-7/H-8/H-9/H-10, H-8/H-15, and H-9/H-11/H-14/H-15/H-16; and 1H detected heteronuclear multiple bond correlation (HMBC) correlations from H-3 to C-2 (δC 165.5) and C-5 (δC 107.7), from H-4 to C-2 and C-4 (δC 141.5), from H-5 to C-3 (δC 116.7), C-6 (δC 153.9), and C-7 (δC 41.2), from H-10 [δH 4.05 (1H, m)] to C-2, C-9 (δC 37.8), and C-11 (δC 67.7), from H-11 [δH 3.12 (1H, t, J = 4.9 Hz)] to C-9, C-13 (δC 56.1), and C-15 (δC 35.3), and from H-17 to C-11 and C-13 (Figure 2). The 2-hydroxymethyl-5-(2-hydroxypropan-2-yl)phenol unit was confirmed on the basis of HMBC correlations (Figure 2) from H-7′ [δH 4.62 (2H, s)] to C-1′ (δC 156.0), C-2′ (δC 26.5), and C-3′ (δC 129.0), from H-3′ to C-1′, C-4′ (δC 116.6), and C-5′ (δC 151.5), from H3-9′(10′) to C-5′ and C-8′ (δC 72.8); 1H,1H COSY correlation from H-3′ to H-4′. The connection of the 2 structural units was 16-O-7′, which could be deduced by the HMBC correlation from H-16 [δH 3.47 (1H, m)] to C-7′ (δC 60.9).
The relative configuration could be established by the nuclear overhauser effect spctroscopy correlations of H-8/H2-16/H-9/H2-10 and H-7/H-15 (Figure 3). The absolute configuration of
In addition to hosimonoal (
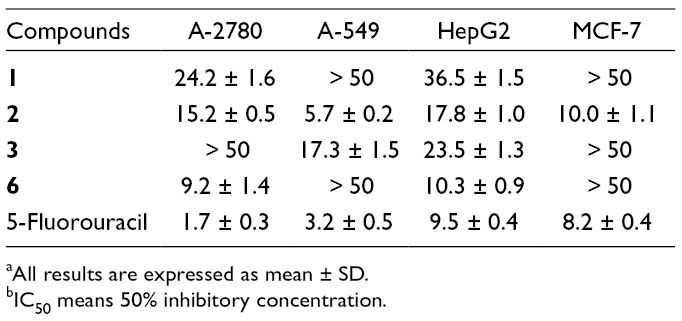
All the compounds were evaluated for their in vitro tumor cytotoxic activities against A2780, A-549, HepG2, and MCF-7 cell lines (Table 2). Compounds
aAll results are expressed as mean ± SD.
bIC50 means 50% inhibitory concentration.
Experimental
General Experimental Procedures
A JASCO P-2000 polarimeter (JASCO, Easton, MD, United States) and JASCO V650 (Thermo Scientific, Waltham, MA, United States) spectrophotometer were used to collect optical rotations and UV spectra, respectively. A Bruker-400 spectrometer (Bruker-Biospin, Billerica, MA, United States) was used to measure the 1D and 2D NMR spectra. An Agilent 6520 High Performance Liquid Chromatography-Quadrupole-Time-of- flight mass spectrometry (Agilent, Waldbronn, Germany) was used to obtain the HRESIMS. High Performance Liquid Chromatography-diode array detector analysis was performed using a Waters 2690/5-W2998 series system with a Dikma Diamonsil (C18 250 × 4.6 mm, 5 µm). Preparative High Performance Liquid Chromatography (HPLC) was performed on a Shimadzu LC-8AD instrument with a SPD-20A detector (Shimadzu Corp., Tokyo, Japan), using 2 types of YMC-Pack ODS-A column (20 × 250 mm, 5 µm and 10 × 250 mm, 5 µm). Column chromatography (CC) was performed using silica gel (200-300 mesh, Qingdao Marine Chemical Inc., Qingdao, People’s Republic of China), D101 macroporous resin (Changfeng Technology Co. Ltd, Jiangsu, People’s Republic of China), and Sephadex LH-20 (GE, United States).
Plant Material
The seeds of O. hosiei Hemsl. et Wils. were gathered from 3 trees in the Jin’an District, Fuzhou, Fujian Province, on April 15, 2017. The trees were more than 100 years old and identified by Professor Bizhu He and Shasha Wu from Fujian Agriculture and Forestry University (FAFU), People’s Republic of China.
Extraction and Isolation
The air-dried and powdered seeds of O. hosiei (600 g) were subjected to reflux extraction with 70% EtOH (2 × 6 L) at 85°C and the solution was evaporated under reduced pressure in a rotary evaporator at 50°C to yield 100 g extract. This was fractionated by PRP-512A macroporous resin CC, using a gradient solvent system of ethanol-H2O (10%, 30%, 50%, 70%, and 100%; v/v; 2.0 L for each gradient) to yield 5 fractions (A1-A5). Fraction A2 (26.2 g) was subjected to silica gel CC using a gradient solvent system of CHCl3-MeOH (1:0-0:1) to afford fractions B1-B5. Fraction B1 (3.5 g) was subjected to Sephadex LH-20 CC eluting with CHCl3:MeOH (1:1) to give fractions C1-C7. Fraction C4 (170.8 mg) was further purified by preparative HPLC [MeOH-H2O-TEA (25:75:0.05), v/v, 8.0 mL/min, 210 nm] to yield compound
Biological Activities
A2780 (ovarian neoplasm cell line), A-549 (lung cancer cell line), HepG2 (liver cancer cell line), and MCF-7 (breast cancer cell line) were used to test for cytotoxic activities. The cytotoxicity assay was carried out using the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide method, as previously described. 15,16 5-Fluorouracil (Sigma, St. Louis, MO, United States) was used as a positive control, and the IC50 values were calculated by Graphpad prism 7.0.
Hosimonoal (1)
Yellow gum.
IR (Microscope Transmission): 3353, 2932, 1647, 1547, 1420, 1364, 1176, 1148, 1026, 804, 654 cm−1.
UV (CH3OH) λmax (log ε): 203.0 (4.07), 282.0 (3.31), 310.0 (3.41).
CD (MeOH) λmax (Δε): 233.5 (+4.31), 308.5 (−3.14);
1H NMR (methanol-d 4, 400 MHz) and 13C NMR (methanol-d 4, 100 MHz): see Table 1.
HRESIMS m/z: 425.24258 [M+H]+ (calcd for C25H33O8N2, 425.24348).
Supplemental Material
Supplemental data - Supplemental material for Chemical Constituents and Their Activities From the Seeds of Ormosia hosiei
Supplemental material, Supplemental data, for Chemical Constituents and Their Activities From the Seeds of Ormosia hosiei by Huiyou Xu, Yatie Qiu, Jingxin Chen, Yan Shi, Xiaoqin Zhang, Qiang Song, Mingqing Huang, Zujian Wu, and Lin Ni in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 31700292) and the Foundation of Pharmaceutical Engineering Competition Steering Group of FAFU (No. 103-111416040).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.