Abstract
In the present study, the heartwood and the sapwood of a thick wood disc from a dated Cedrus brevifolia trunk, and the resin were analyzed for their volatile components. All samples were subjected to headspace solid-phase microextraction followed by gas chromatography mass spectrometry analysis. The main constituents were β-himachalene (22.2 %) in heartwood and (25.0 %) in sapwood, and α-pinene (37.8 %) in resin.
Driven by the rich phytochemical profile of the genus Cedrus Trew, along with its pharmacological and economic interest, we have investigated Cedrus brevifolia (Hook. f.) Henry, a long-lived species with a maximum life of 500 years. 1 It is well differentiated from the other Cedrus species based on morphological and eco-physiological traits. 2 Forests of C. brevifolia are known only from 1 location, at an altitude of 900-1 400 m in the western summits of the Troodos range. 3,4 Due to its restricted distribution, the species is characterized as Vulnerable according to the IUCN Red List of Threatened Species. 3 Essential oils (EOs) isolated from Cedrus species are important natural sources, since they are used as raw materials for the production of fragrances used in cosmetic, food, and a variety of household products. 5 Moreover, they present many pharmacological properties. The EO of Himalayan cedar wood is locally used to treat insect infections of animals and its anti-mange activity is attributed to himachalenes. 6 In addition, it is used as a spasmolytic agent, with himachalol being the active ingredient. 6 Moreover, it possesses important anti-inflammatory, analgesic, anti-arthritic, 7 -9 and anti-ulcer properties, 7,9,10 as well as larvicidal activity against larvae of Plutella xylostella. 11 It is also used as diaphoretic agent 12 and to treat insect bites. 13 Himalayan cedar gum is used to treat indigestion, insomnia, inflammations and various skin and blood disorders. 7,12
Ancient Egyptians used C. libani extracts to mummify the Pharaohs’ bodies, 14 as well as its light, durable, and resinous wood for shipbuilding. 15 The tar obtained from the burnt C. libani wood was used for several millennia in areas of SW Turkey, under the name katran for the treatment of ulcers, 16 for wound healing, and against digestive tract bacteria. 14 Furthermore, EOs and ethanol extracts derived from C. libani cones and wood exhibit antidiabetic in vitro activity. 17
C. atlantica is the main species of Moroccan forests known for the natural durability of its wood used as timber. 18,19 Its main difference from the other cedars is the characteristic wood odor. 20 Atlas cedar wood and its EO are known to be natural repellents of moths; hence, cedar is a popular lining for chests and wardrobes in which woolens are stored. Previous studies showed that C. libani, C. atlantica, and C. deodara wood EOs possess potent in vitro erythroid differentiation and antiproliferative activity against human K562 chronic myelogenous leukemia cells. 21,22
There is little interest in wood production of C. brevifolia due to its slow rates of growth, narrow distribution, and vulnerability to late frosts. 23 Actually, little is known about the chemistry of this species. In the present study, we investigated the volatile constituents of C. brevifolia heartwood, sapwood, and resin. So far, only the polar constituents of the needles have been reported. 24
A thick disc of C. brevifolia wood was examined. From the pith to the bark, 52 annual rings were counted. Based on crossdating results (Table 1) with C. brevifolia master chronology from Tripylos (TRI0CYCB), Abies cephalonica chronology from Chelmos mountain (CHELGRAC), and Pinus nigra from Pindos mountain (PINDGRPN) in Greece, the outermost, not fully completed, ring was from 2017. This finding is in accordance with the data provided by the Department of Forests. Results of crossdating (Table 1) are acceptable, given the short sequence of the sample under study (Figure 1). Being only 52 years old and in comparison, with the ages that Cedrus species can reach, 25 -27 the tree was rather young when cut. Given its age, an insignificant growth decrease would be expected due to age. Nevertheless, a linear decrease in annual growth was recorded since 1975 (Figure 2). Prolonged drought events 28 and precipitation decreases, especially during the growing season, 29 could explain this decreasing trend in annual growth. The decrease in annual growth could also be attributed to inter- and/or intraspecific competition, since cedars are light-demanding, 29,30 while significant reduction in tree growth may also be related to the presence of insect defoliators such as processionary moth. 31 Dendrochronological data of the current study do not permit a conclusion to be made as to which factor was responsible for this decrease in annual growth, but it would be interesting to know if this stress recorded in the last 42 rings is also somehow connected with the compounds identified in the sapwood.
Crossdating Results of Cedrus brevifolia Sample With Master Chronologies From Cyprus (TRI0CYCB) and Greece (CHELGRAC and PINDGRPN).

OVL, overlap; Glk, Gleichlaeufigkei; CDI, crossdate index; TVBP, t-value Ballie-Pilcher; TVH, t-value Hollstein.
Mean significance for Glk (* =95.0%, **=99.0%, ***=99.9%).

Visual crossmatching of the cedar sample with master chronology from Tripylos (TRI0CYCB).
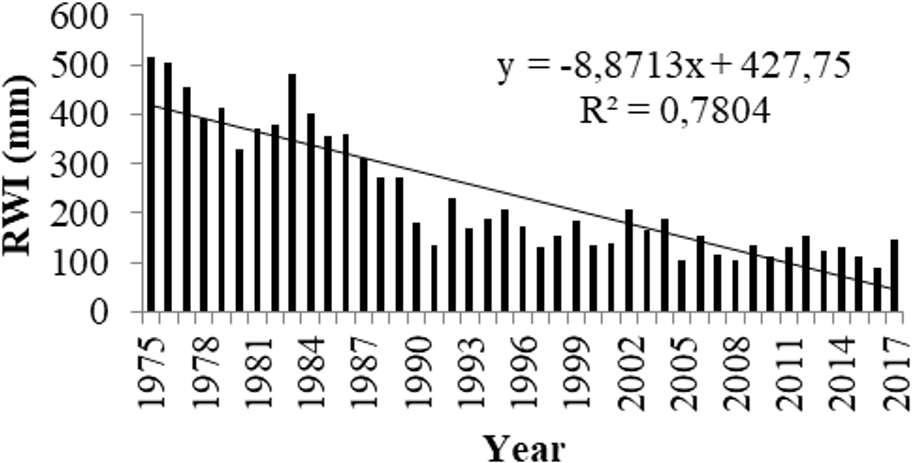
Linear decrease in annual growth, as expressed from RWI, for the period 1975 to 2017. RWI: ring width measurements.
The chemical analyses revealed that in each instance, the volatile fractions consisted of a complex mixture of different terpenoids. The identified volatile components are listed according to their chemical structures in Table 2, where their retention indices and the percent composition are included.
Qualitative and Quantitative Composition (% V/v) of Volatile Compounds of Cedrus brevifolia Wood and Resin.
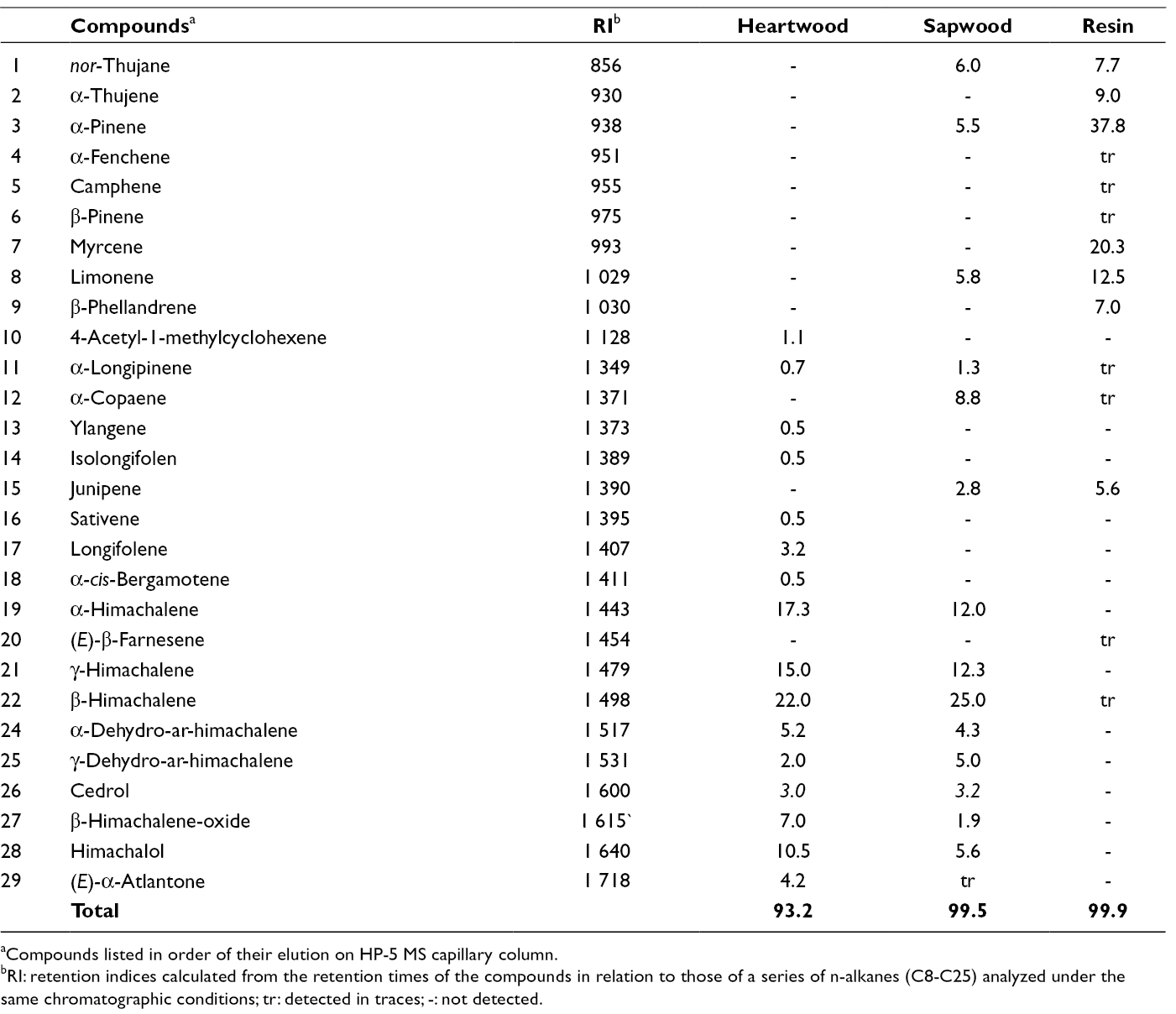
aCompounds listed in order of their elution on HP-5 MS capillary column.
bRI: retention indices calculated from the retention times of the compounds in relation to those of a series of n-alkanes (C8-C25) analyzed under the same chromatographic conditions; tr: detected in traces; -: not detected.
In total, 16 compounds in heartwood and 14 compounds in sapwood were identified covering 93.2% and 99.5%, respectively, while 7 compounds were found in resin representing 99.9%, of its total volatile constituents in addition to 7 other traces. From Tables 2 and 3, it is evident that the sesquiterpene hydrocarbons are the dominating constituents in heartwood and sapwood, representing 70.4% and 74.7%, respectively, with himachalenes being their main metabolites, while in resin, α-/γ-himachalenes are totally absent and the amount of β-himachalene is negligible. In contrast, monoterpene hydrocarbons, although major constituents in resin (86.6%), are totally absent in heartwood, while in sapwood their percentage arises to 11.3. It is also noteworthy that the quantities of some resin volatiles are significantly lower or absent from the other samples. Myrcene (20.3%), α-thujene (9.0%), and β-phellandrene (7.0%), major constituents of the resin, are absent in heartwood and sapwood, while α-pinene, the most abundant constituent in resin (37.8%), is present only in sapwood in significantly smaller amount (5.5%). Moreover, the amount of limonene is appreciable in resin (12.5%), but much lower in sapwood (5.8%) and absent in heartwood.
Grouped Components (% V/v) of Cedrus brevifolia Heartwood, Sapwood, and Resin.

Previously investigated volatiles of other cedar woods revealed that α-/β-/γ-himachalenes and (E)-α-atlantone are main constituents of C. libani and C. atlantica heartwood EOs, while himachalol is a specific marker in both EOs. 17,32 Generally, cedar EOs contain high percentages of himachalenes, although their content could differ greatly even in the same genus, as well as in different plant parts. 33 -36 It is worth mentioning that C. atlantica wood is well known for its characteristic and rare odor, attributed mainly to 4-acetyl-1-methylcyclohexene and vestitenone, 20 the first of which is also found in C. brevifolia heartwood (1.1%). Moreover, the percentages of C. brevifolia and C. libani heartwood EOs are similar, probably because both species are very close taxonomically. 32
Experimental
Plant Material
C. brevifolia is endemic to Cyprus and is located in the area of Tripylos (Cedar Valley), in the Paphos forest, where it occupies the area of the main peak and is located in scattered small clusters on the surrounding peaks. The wood cut down in July 2017 and the resin were provided by the Department of Forests, Ministry of Agriculture, Natural Resources and Environment, Nicosia, Cyprus.
Dendrochronological Dating
C. brevifolia is a species suitable for dendrochronology, 37 since the ring boundaries are rather distinct and the transition from early wood to latewood is usually continuous. 38 In the current study and in order to date the sample, we cut only a small segment since the annual tree rings were relatively wide and one radius was enough for measuring the tree rings width. The surface of the sample was sanded with progressively finer grade abrasive paper (from 80 to 600 grit), until cells were clearly visible under magnification. Tree rings were counted, and ring widths were measured to 0.01 mm using the Time Series Analysis and Presentation (TSAP) software 39 and a LINTAB measuring table (Rinntech, Heidelberg, Germany). The next step was to date the sample by comparing the tree-ring sequence of the sample against a corresponding, already dated master chronology. 40 At this step, of paramount importance is the choice of the right master chronology, which should be of the same tree species or from a tree with similar growth trend, long enough to include the prospective time period covered by the sample and derived from the same wider geographical region. 40,41 In the current study, we used a chronology for C. brevifolia from Tripylo, derived from the International Tree-Ring Data Bank. 42 The above-mentioned master chronology consisted of 42 trees and covers the period from 1532 to 2002. Since our sample has been cut more recently, we additionally used chronologies from other conifers, namely Abies cephalonica from Chelmos and Pinus nigra from Pindos in Greece, 43 to have a bigger overlap with the undated cedar sample. Evaluation of crossdating results was based on the following parameters: (i) Gleichlaeufigkeit, representing the overall accordance of two series; (ii) t-value Ballie-Pilcher and t-value Hollstein, t-values that are sensitive to extreme values, such as marker years; and (iii) crossdate index, which is a combination of both. 39 The overlap was also taken into account.
Headspace solid-phase microextraction
In most cases, solid-phase microextraction (SPME) sampling has considerable advantages over other commonly used methods; the sensitivity of SPME saving time and no need for large amounts of sample are 3 of those advantages. Nevertheless, SPME has proved to be a viable alternative method for the determination of volatiles when the amount of sample is limited. 44 Previous studies have shown that headspace SPME (HS-SPME) is a convenient method for determination of volatiles from woods, 45 as in the cases of the volatiles released from wood of Picea abies (Norway spruce), Pinus sylvestris (Scots pine), and Larix decidua (European larch). 46
To determine the components, the heartwood was separated from the sapwood and each sample was comminuted. To 50 mL headspace vials sealed with a septum-type cap, 2.40 g of heartwood, 2.34 g of sapwood, and 5.88 g resin were transferred. A Carboxen/polydimethylsiloxane fiber (CAR/PDMS, 75 µm, Supelco) was used for SPME analysis. The SPME fiber was conditioned at 280°C for 1 hour in the injection port of a GC system before analyzing samples in order to remove contaminants. Subsequently, the SPME fiber was exposed in the headspace above the samples and the vials were heated to 45°C for 15 minutes. The SPME fiber was then inserted directly into the injector of the GC system for 18 minutes to allow for desorption of analytes from the fiber coating.
Gas Chromatography/Mass Spectrometry
Analyses were performed in triplicate by gas chromatography/mass spectrometry carried out using a Hewlett-Packard 6890 gas chromatograph equipped with a HP-5MS fused silica capillary column (30 m × 0.25 mm; film thickness 0.25 µm), a split-splitless injector, and a Hewlett-Packard 5973 MS detector operating in electron ionization mode at 70 eV (Agilent Technologies, Santa Clara, CA, USA). Injection was at 280°C in a split ratio 1:5; the ion source temperature and transfer line temperature were set at 230 and 250°C, respectively. The carrier gas was helium at 1 mL/min and the oven temperature was increased from 60 to 240°C at 10°C/min.
Retention indices for all compounds were determined according to the Van der Dool approach, 47 using n-alkanes as standards. The identification of the components was based on comparison of their mass spectra with those of Wiley, NIST/NBS Libraries 48 and those described by Adams, 49 as well as by comparison of their retention indices with literature data. 49 In most cases, co-chromatography with authentic markers enabled the identification of the quoted compounds.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research work was supported by the Hellenic Foundation for Research and Innovation (HFRI) and the General Secretariat for Research and Technology (GSRT), under the HFRI PhD Fellowship grant (GA. 14 674).
