Abstract
Nannozinone A is an unprecedented dihydropyrrolopyrazinone metabolite derived from myxobacterial Nannocystis pusilla strain MNa10913. In this paper, the synthesis of nannozinone A was disclosed for the first time. Nannozinone A was readily obtained in 6 steps and in 36% overall yield from commercially available N-Boc-propinal. Key feature of the synthesis includes intramolecular imine formation and subsequent spontaneous oxidation toward the unique dihydropyrrolopyrazinone skeleton. This concise synthetic route would be helpful to prepare natural dihydropyrrolopyrazinones and their derivatives.
Myxobacteria (also known as slime bacteria) are rod-shaped, Gram-negative bacteria that show unique features such as gliding motility on solid surfaces and formation of complex fruiting bodies.
1
They have been considered as one of the proficient producers of structurally diverse and unique bioactive natural compounds that might contribute to the discovery of novel therapeutic molecules.
2,3
Recently, Müller and his co-workers isolated unprecedent natural pyrazinones including nannozinone A (
As shown in Figure 1, it was envisioned that the pyrazinone moiety of nannozinone A (
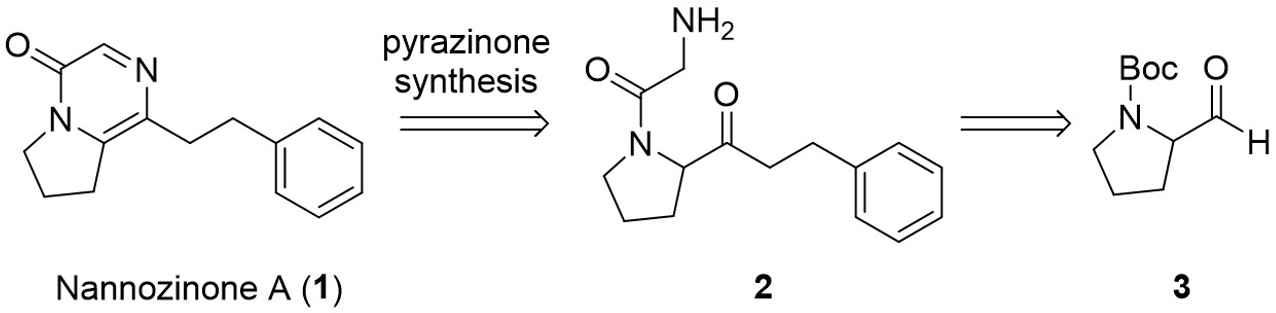
Nannozinone A (1) and its retrosynthetic analysis.
As shown in Scheme 1, the synthesis of nannozinone A (
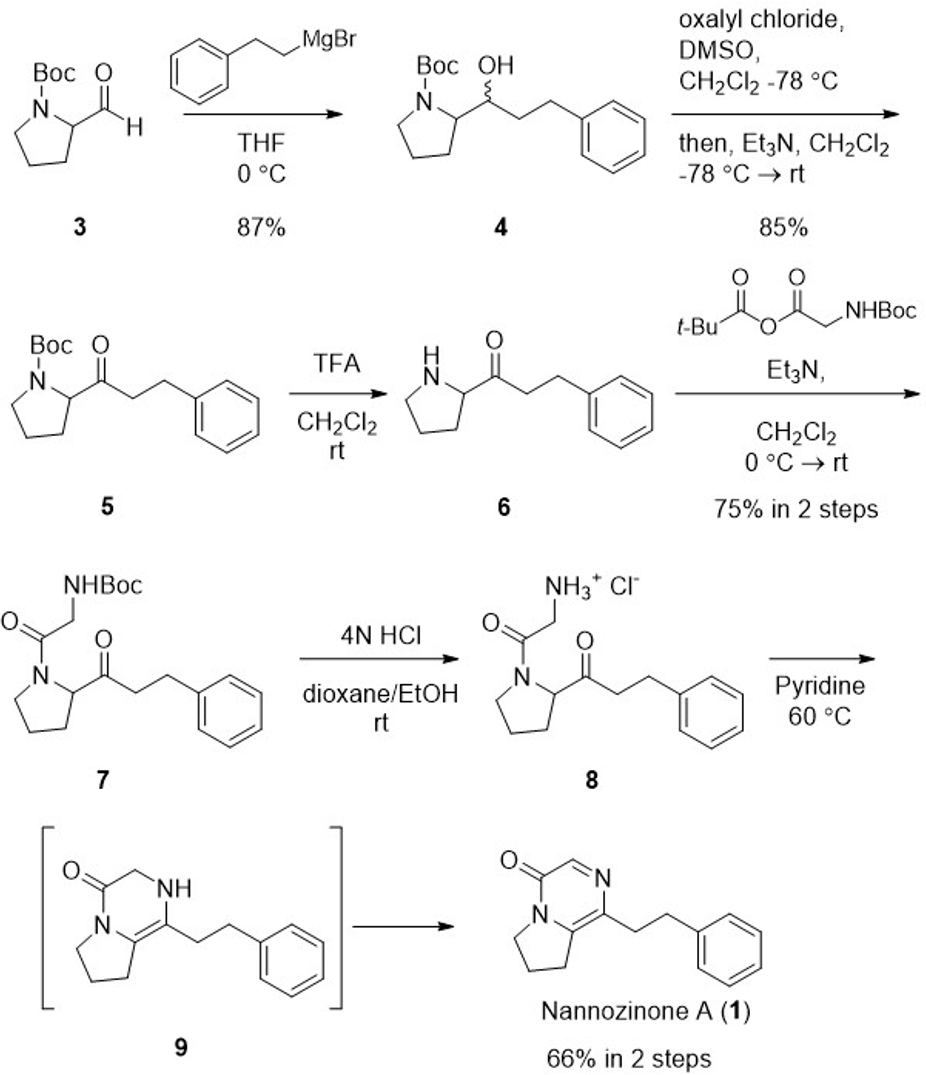
Synthesis of nannozinone A (1).
To obtained amide
In summary, this paper describes a concise synthesis of nannozinone A (
Experimental
Unless noted otherwise, all starting materials and reagents were obtained commercially and were used without further purification. All solvents utilized for routine product isolation and chromatography were of reagent grade and glass distilled, and reaction flasks were dried at 100°C before use. Air- and moisture-sensitive reactions were performed under argon atmosphere. Flash column chromatography was performed using silica gel 60 (230-400 mesh, Merck) with the indicated solvents. Thin-layer chromatography was performed using 0.25 mm silica gel plates (Merck). Mass spectra were obtained using an Agilent 6530 Quadrupole time of flight (Q-TOF) unit. Infrared spectra were recorded on a JASCO FT-IR-4200 spectrometer. UV-VIS spectra were obtained using a Shumadzu UV-1650. 1H and 13C spectra were recorded on a Bruker ADVANCE 800 (800 MHz) spectrometer in deuteriochloroform (CDCl3). Chemical shifts are expressed in parts per million (ppm, δ) downfield from tetramethylsilane and are referenced to the deuterated solvent. 1H NMR data are reported in the order of chemical shift, multiplicity (s, singlet; d, doublet; t, triplet; q, quartet; quin, quintet; m, multiplet; bs, broad singlet; and/or multiple resonances), number of protons, and coupling constant in Hertz (Hz).
Tert-Butyl 2-(1-Hydroxy-3-Phenylpropyl)pyrrolidine-1-Carboxylate (4)
To a solution of Boc-prolinal
Rf: 0.35, 0.45 (EtOAc/n-hexane = 1:2).
IR (thin film, neat): 3417, 2975, 1693, 1667, 1404, 1168 cm−1.
1H NMR (800 MHz, CDCl3);
13C NMR (200 MHz, CDCl3);
High resolution mass spectroscopy (HRMS; Q-ToF): Calcd for C18H28NO3 + (M+H+): 306.2064. Found: 306.2069.
Tert-Butyl 2-(3-Phenylpropanoyl)Pyrrolidine-1-Carboxylate (5)
To a solution of oxalyl chloride (0.4 mL, 4.26 mmol) in CH2Cl2 (16 mL), dimethyl Sulfoxide (0.6 mL, 8.51 mmol) was added at −78°C. After 30 minutes, a solution of alcohol
Rf: 0.35 (EtOAc/n-hexane = 1:4).
IR (thin film, neat): 2976, 1727, 1697, 1395, 1165, 701 cm−1.
1H NMR (800 MHz, CDCl3, mixture of rotamers) δ 7.28-7.25 (m, 2H, Ar-H), 7.20-7.17 (m, Ar-H), 4.34-4.33 and 4.22-4.20 (m, 1H, -NCH), 3.53-3.40 (m, 2H, -NCH2), 2.93-2.89 (m, 2H, ArCH2), 2.81-2.68 (m, 2H, -CH2-), 2.13-2.11 and 2.03-2.02 (m, 1H, -CH2-), 1.81-1.78 (m, 2H, -COCH2), 1.68-1.65 (m, 1H, -CH2-), 1.46(s, 4H, tert-Bu), 1.38(s, 5H, tert-Bu).
13C NMR (200 MHz, CDCl3, mixture of rotamers) δ 209.4/209.2 (1C, C=O), 154.8/154.0 (1C, C=O), 141.5/141.2 (1C, Ph), 128.7/128.6/128.5 (4C, Ph), 126.3/126.2 (1C, Ph), 80.3/80.0 (1C, O
HRMS (Q-ToF): Calcd for C18H26NO3 + (M+H+): 304.1907. Found: 304.1914.
Tert-Butyl (2-Oxo-2-(2-(3-Phenylpropanoyl)Pyrrolidin-1-yl)Ethyl) Carbamate (7)
To a solution of Boc-pyrrolidine
MP: 101°C-104°C.
Rf: 0.15 (EtOAc/n-hexane = 1:2).
IR (neat): 3421, 3061, 2977, 1715, 1656, 1453, 1169 cm−1.
1H NMR (800 MHz, CDCl3) δ 7.29-7.25 (m, 2H, Ar-H), 7.20-7.16 (m, 3H, Ar-H), 5.38 (bs, 1H, NHBoc), 4.59-4.57 (m, 1H, -NCH), 4.00-3.97 (m, 1H, -NCH2), 3.91-3.88 (m, 1H, -NCH2), 3.55-3.52 (m, 1H, -NCH2), 3.46-3.43 (m, 1H, -NCH2), 2.93-2.91 (m, 2H, ArCH2), 2.88-2.86 (m, 1H, -CH2-), 2.82-2.78 (m, 1H, -CH2-), 2.05-2.02 (m, 1H, -CH2-), 1.97-1.91 (m, 2H, -COCH2), 1.72-1.69 (m, 1H, -CH2-), 1.44-1.43 (m, 9H, tert-Bu).
13C NMR (200 MHz, CDCl3) δ 207.4 (1C, C=O), 167.4 (1C, C=O), 155.9 (1C, C=O), 141.1 (1C, Ph), 128.6/128.6/128.5/128.5 (4C, Ph), 126.3 (1C, Ph), 79.8(1C, O
HRMS (Q-ToF): Calcd for C20H29N2O4 + (M+H+): 361.4615. Found: 361.4620.
Nannozinone A (1)
To a solution of N-Boc-ethylcarbamate
MP: 112°C-115°C.
Rf: 0.2 (EtOAc/n-hexane = 2:1).
IR (neat): 2925, 1657, 1596, 1171, 702 cm−1.
UV (MeOH) λmax (log ε) 233 (4.03), 333 (3.78).
1H NMR (800 MHz, CDCl3) δ 8.05 (s, 1H, N=CH), 7.25-7.23 (m, 2H, Ar-H), 7.19-7.17 (m, 1H, Ar-H), 7.06-7.05 (m, 2H, Ar-H), 4.05 (t, 2H, J = 7.4 Hz, NCH2), 2.95 (t, 2H, J = 7.3 Hz, ArCH2), 2.80 (t, 2H, J = 7.3Hz, -CH2-), 2.57 (t, 2H, J = 7.7 Hz, -CH2-), 2.00 (quin, 2H, J = 7.6 Hz, -CH2-).
13C NMR (200 MHz, CDCl3) δ 155.5 (1C, C=O), 145.0 (1C, N=CH), 141.1 (1C, Ar), 139.4 (1C, N
HRMS (Q-ToF): Calcd for C15H17N2O+ (M+H+): 241.1335. Found: 241.1339.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared no financial support for the research, authorship, and/or publication of this article.
