Abstract
In the course of the search for cancer cell growth inhibitors, 3 new diterpene alkaloids, designated ceylonamides G-I (
Cancer is the fastest growing health problem and leading cause of death worldwide. Although a number of new anti-cancer drugs are approved yearly, more efficacious drugs with fewer side effects are greatly needed. Natural products are the primary source of anti-cancer drugs, as about half of the present cancer therapy drugs are either natural products or their derivatives. 1 Recently, marine invertebrates such as marine sponges have emerged as a valuable resource for discovering anti-cancer drugs owing to their diverse content of secondary metabolites. Trabectedin (Yondelis) isolated from Ecteinascidia tubinata, and Eribulin (Halaven), a halichondrin B derivative, isolated from Halichondria okadai are typical examples. 2 More than 20 compounds from marine medicinal resources are in the clinical trial phase. 3 We have also isolated some marine natural products showing unique effects on the cancer cells. For instance, furospinosulin-1 (furanosesterterpene) and dictyoceratin-C (sesquiterpene phenol) are isolated from Dactylospongia elegans, which are selective growth inhibitors of hypoxia-adapted human prostate cancer DU145 cells. 4 -7 N-methylniphatyne A (new 3-alkylpyridine alkaloid) and biakamides (new unique polyketides) isolated from Xestospongia sp. and Petrosaspongia sp., respectively, inhibited the growth of pancreatic cancer PANC-1 cells adapted to nutrient-starved conditions. 8,9
On the contrary, candidate compounds with promising results in cell-based assays are often ineffective in tumor xenograft animal models, mostly due to the reliance on established two-dimensional (2D) monolayer cultures for cellular response studies. Albeit enormously useful for screening of cytotoxic compounds, the 2D monolayer culture falls short in recapitulating the complex native environment of living tumor tissue. This situation has then led to the pursuit of three-dimensional (3D) cell cultures that have the potential to improve the physiological relevance of cell-based studies and increase the successful translation of cell-based drug screening. 10 Among the 3D cell culture models, spheroid cultures are one of the well-characterized methods for screening of cytotoxic compounds due to its simplicity and reproducibility. 11
In this study, 3 new diterpene alkaloids, designated ceylonamides G-I (
Ceylonamide G (
1H- (600 MHz) and 13C-NMR (150 MHz) Spectral Data for Compounds 1 and 2 in CD3OD (δ in ppm).


Chemical structures of ceylonamides 1 to

HMBC and COSY correlations for compounds 1 to
1H- (600 MHz) and 13C-NMR (150 MHz) Spectral Data for Compounds 3 and 4 in CD3OD (δ in ppm).

Ceylonamide H (
Ceylonamide I (
The relative stereochemistry of compounds
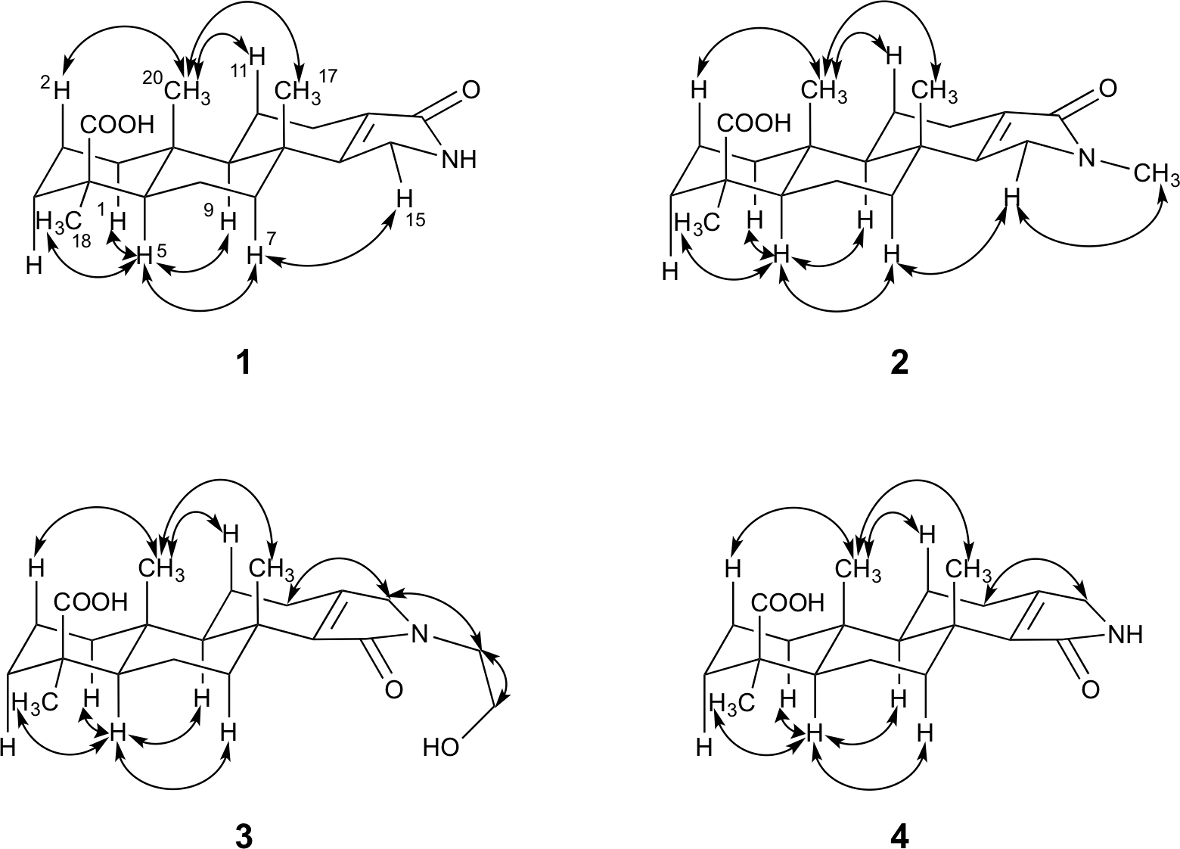
ROESY correlations for compounds 1 to
We next evaluated the cytotoxic activity of compounds

Effects of compounds 1 and 4 on the spheroids prepared from DU145 cells. Minimum effective concentrations of compounds 1 and 4 were determined on the basis of the morphological change in the spheroids, as described in Experimental section. Taxol was used as a positive control. “*”: Minimum effective concentrations of the compounds.
Cytotoxicity of Compounds 1 to

2D, two-dimensional; MEC, minimum effective concentration.
Although investigating the mode of action and the effect of the compounds on the microenvironment of spheroids is necessary, our findings suggested that compounds
Experimental
General
NMR spectra, referenced to tetramethylsilane or residual solvent peak, were measured on an Agilent NMR system (1H: 600 MHz, 13C: 150 MHz). ESI-TOF-MS was recorded on a Q-Tof Ultima API mass spectrometer (Waters Co., MA, United States). IR spectra and specific rotations were obtained with a JASCO FT/IR-5300 (KBr pellets) and JASCO P-2200 digital polarimeter (L = 50 mm) (JASCO Corporation, Tokyo, Japan), respectively. UV spectra were obtained with UV-2450 spectrophotometer (SHIMADZU, Kyoto, Japan). Column chromatography was performed on Silica gel BW-200 (Fuji Silysia Aichi, Japan), COSMOSIL 5C18-AR-II (Nacalai tesque, Kyoto, Japan), and COSMOSIL Cholester (Nacalai tesque, Kyoto, Japan). HPLC was performed using a Hitachi L-6000 pump equipped with Hitachi L-4000H UV detector (Hitachi High-Tech Science Corporation, Tokyo, Japan). Thin-layer chromatography (TLC) analysis was carried out using pre-coated TLC plates (Merck, 60F254). Spots on the TLC plates were detected by spraying acidic p-anisaldehyde solution (p-anisaldehyde: 25 mL, c-H2SO4: 25 mL, AcOH: 5 mL, and EtOH: 425 mL) or phosphomolybdic acid solution (phosphomolybdic acid: 25 g and EtOH: 500 mL) with subsequent heating. The following reagents and materials were used for cell culture and bioassay: RPMI 1640 medium (Nacalai tesque, Inc., Kyoto, Japan); fetal bovine serum (FBS; lot 30-2215) (Equitech-Bio Inc., Kerrville, TX, United States); taxol (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan); and kanamycin, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and other chemicals (Sigma-Aldrich, St Louis, MO, United States or Nacalai tesque, Inc., Kyoto, Japan).
Extraction and Isolation
The specimen of marine sponge was collected at Biak, Indonesia, and was identified as a Spongia sp. on the basis of morphological characteristics. The dried marine sponge of Spongia sp. (330 g) was extracted with MeOH. The MeOH extract (44 g) was partitioned into a water-EtOAc mixture. Following guidance of bioassay, the active EtOAc soluble portion (11 g) was subjected to silica gel column chromatography by eluting with n-hexane-EtOAc (10:1), EtOAc, chloroform-MeOH (10:1), and MeOH. The active chloroform-MeOH elute (0.7 g) was separated using ODS-HPLC (COSMOSIL 5C18-AR-II, 10 mm i.d. × 250 mm, 60% MeOH aq. containing 0.1% trifluoroacetic acid [TFA]) to afford ceylonamide F (17.8 mg,
Ceylonamide G (1)
A white amorphous powder. [α]D 25: −24.2° (c = 0.1, MeOH). IR νmax (KBr) cm−1: 3276, 2928, 2848, 1693, 1452, 1213. UV λmax (MeOH) nm (log ε): 217 (3.2), 223 (0.1). ESI-TOF-MS: m/z 354 [M + Na]+. High resolution (HR)-ESI-TOF MS: Calcd for C20H29NO3Na: m/z 354.2045. Found 354.2060. 1H NMR (600 MHz, CD3OD δ H), 13C NMR (150 MHz, CD3OD, δ C) spectra: as shown in Table 1.
Ceylonamide H (2)
A white amorphous powder. [α]D 25: −23.1° (c = 0.1, MeOH). IR νmax (KBr) cm−1: 3326, 2898, 1672, 1522. UV λmax (MeOH) nm (log ε): 216 (3.4), 222 (3.5), 225 (3.5). ESI-TOF-MS: m/z 368 [M + Na]+. HR-ESI-TOF MS: Calcd for C21H31NO3Na: m/z 368.2202. Found 368.2197. 1H NMR (600 MHz, CD3OD δ H), 13C NMR (150 MHz, CD3OD, δ C) spectra: as shown in Table 1.
Ceylonamide I (3)
A white amorphous powder. [α]D 25: −32.0° (c = 0.1, MeOH). IR νmax (KBr) cm-1: 3226, 2978, 1701, 1517. UV λmax (MeOH) nm (log ε): 202 (2.8), 203 (2.8), 210 (2.8). ESI-TOF-MS: m/z 398 [M + Na]+. HR-ESI-TOF MS: Calcd for C22H33NO4Na : m/z 398.2307. Found 398.2315. 1H NMR (600 MHz, CD3OD δ H), 13C NMR (150 MHz, CD3OD, δ C) spectra: as shown in Table 2.
Cytotoxicity of the Compounds on the DU145 Cells Under 2D Culture Conditions
Human prostate cancer DU145 cells were maintained in RPMI 1640 supplemented with heat-inactivated 10% FBS and kanamycin (50 µg/mL) in a humidified atmosphere of 5% CO2 at 37°C. In order to evaluate the activity of the compounds, the DU145 cells were plated in 96-well plates (1 × 104 cells/200 µL/well) and incubated for 12 hours in a humidified atmosphere containing 5% CO2 at 37°C. The test compounds were later added, and the plates were incubated for an additional 24 hours under the same conditions. Cell proliferation was detected according to a previously established MTT method. 14 The growth inhibition rate was calculated as a percentage relative to negative controls. The IC50 values were determined by linear interpolation from the growth inhibition curve.
Spheroid Preparation and Cytotoxicity Evaluation
The preparation of spheroids from DU145 cells was performed based on the description by Friedrich et al. 11 Briefly, sterilized 1.5% agarose in RPMI 1640 medium (50 µL) was added into each well of 96-well plates, and the plates were kept at room temperature to solidify the agarose. Then, DU145 cells in the RPMI 1640 medium supplemented with heat-inactivated 10% FBS (1 × 104 cells/well/100 µL) were inoculated onto the agarose coated 96-well plates and cultured for 7 days in a humidified atmosphere of 5% CO2 at 37°C to allow the size of spheroids to be approximately 600-700 µm. After forming spheroids, the test compounds were added, and the plates were incubated for an additional 72 hours. To evaluate the effect of the compounds on the spheroids, their concentrations that resulted in dead cells appearance around the spheroid were defined as a MEC.
Supplemental Material
Supplementary data - Supplemental material for Cytotoxicity of New Diterpene Alkaloids, Ceylonamides G-I, Isolated From Indonesian Marine Sponge of Spongia sp.
Supplemental material, Supplementary data, for Cytotoxicity of New Diterpene Alkaloids, Ceylonamides G-I, Isolated From Indonesian Marine Sponge of Spongia sp. by Takahiro Jomori, Andi Setiawan, Miho Sasaoka, and Masayoshi Arai in Natural Product Communications
Footnotes
Acknowledgments
This research is dedicated to the late Professor Hideaki Matsuda (Kindai University, Japan). The authors thank Dr Kazutake Tsujikawa (Osaka University) for providing DU145 cells.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) from AMED (Grant no. JP19am0101084), Kobayashi International Scholarship Foundation, and a Grant-in-Aid for Scientific Research B (Grant nos. 18H02096 and17H04645) from JSPS.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
