Abstract
The antioxidant and antifungal activity of n-hexane and ethyl acetate extracts of stems and roots of Jatropha dioica var. dioica, a plant species from the semi-arid regions of Hidalgo, Mexico, were evaluated by the 2,2-diphenyl-1-picrylhydrazyl and 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) free-radical scavenging capacity assays and by growth inhibition of the phytopathogenic fungi, Alternaria alternata, Sclerotium rolfsii, Colletotrichum gloeosporioides, Rhizoctonia solani, and Fusarium oxysporum. The extracts exhibited a significant capacity to capture free radicals and a moderate capacity to inhibit fungal growth. A phytochemical study of these extracts led to the isolation of alkyl esters of acetic and ferulic acid, as well as the known diterpenes citlalitrione, jatrophatrione, jatropholone A, and jatropholone B, which were physically and spectroscopically characterized. These metabolites could be responsible for the observed antioxidant and fungicide activities of the extracts.
Several plants endemic in arid and semi-arid regions of Mexico have been evaluated for their action against postharvest fungal contamination of crops, due to their minimal harmful effects. 1 This type of plant, which can grow under extreme conditions, synthesizes interesting organic molecules as defense mechanisms. Jatropha dioica var. dioica (Euphorbiaceae) is endemic in arid regions of Mexico, where it is known as “sangre de grado”; infusions of its branches and roots have been traditionally used against hair loss, tooth decay, and cancer. 2 To the best of our knowledge, no biological studies have been conducted on this species, and its chemical composition is currently unknown. Therefore, this work is aimed at evaluation of the antioxidant and antifungal activity of extracts of stems and roots of this plant, as well as to identify the main secondary metabolites in them, which could be responsible for these activities.
Free-radical scavenging capacity assays were performed on the n-hexane and ethyl acetate (EtOAc) extracts of J. dioica, using the 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonicacid) (ABTS·+) and 1,1-diphenyl-2-picrylhydrazyl (DPPH·) methods (Table 1). Both tests showed statistically significant differences with respect to the extract source and the solvent used. Root extracts were more active than those from stems, independently of the extraction solvent and the method used. For both methods, hexane extracts showed a higher antioxidant activity than EtOAc extracts, disregarding the part of the plant studied. On the other hand, antifungal activity (Table 2) was higher in root extracts than in stem extracts, with Alternaria alternata and Sclerotium rolfsii being the most sensitive organisms. The hexane root extract caused a growth inhibition of 45.2% against S. rolfsii, the highest activity observed herein; the stem EtOAc extract exhibited a moderate activity (36.2%) against S. rolfsii; only the hexane root extract showed activity against Fusarium oxysporum (12.0%), and none was active against Rhizoctonia solani.
Antioxidant Activity of Extracts From Stems and Roots of Jatropha dioica var. dioica.
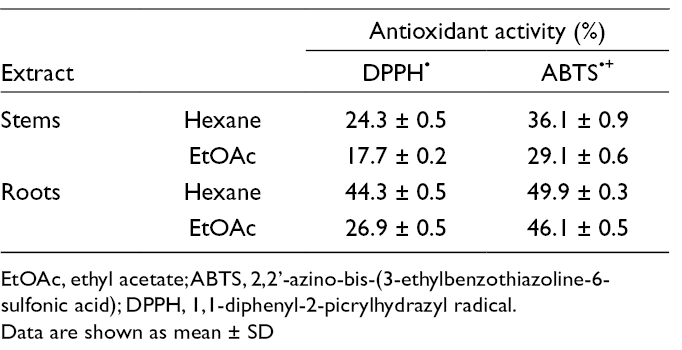
EtOAc, ethyl acetate; ABTS, 2,2’-azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid); DPPH, 1,1-diphenyl-2-picrylhydrazyl radical.
Data are shown as mean ± SD
Antifungal Activity of Extracts From Stems and Roots of Jatropha dioica var. dioica.

EtOAc, ethyl acetate.
Data are shown as mean ± SD
1H NMR analysis showed a similar chemical composition in hexane and EtOAc extracts of the stems. The spectra showed fatty acids, ferulic acid derivatives, and fatty alcohols as the major components of stem hexane extracts. Sterols were also detected as minor constituents. A precipitate was obtained by adding methanol (MeOH) to the extracts and letting them stand at 0°C; the precipitate was filtered and analyzed by 1H NMR. Spectra showed alkyl esters of ferulic acid and fatty alcohols as the major constituents. The filtered solutions were mainly constituted by fatty compounds. Column chromatography (CC) yielded fractions containing fatty acids, fatty alcohols, and alkyl esters of ferulic acid. Fractions A, B, and C were obtained by purifying the latter fraction by HPLC. 1-Octacosyl acetate (
1H and 13C NMR spectra of esters
A white precipitate was obtained by adding hexane-EtOAc (9:1) to the MeOH-defatted root extract, immediately separated by decantation. The residue was crystallized with CHCl3-MeOH to obtain citlalitrione (
In summary, chemical analysis of extracts of J. dioica var. dioica revealed the presence of a significant amount of alkyl esters of ferulic acid, diterpene ketones, and phenols, which could be responsible for the antioxidant and antifungal activities of the extracts. These metabolites are produced by the plant during oxidative stress caused by dehydration.
Experimental
General
Analytical-grade ABTS, DPPH, potassium persulfate (K2S2O8; Reasol, Mexico), methanol (MeOH; Meyer, Mexico), and n-hexane (J.T. Baker, USA) were used to determine antioxidant activity. Potato-dextrose agar (DIFCO, USA) and analytical-grade dimethyl sulfoxide (DMSO; Sigma Aldrich, USA) were used to determine antifungal activity. HPLC-grade formic acid (J.T. Baker, USA), MeOH, and CHCl3 (J.T. Baker) were used in mass spectrometric analysis. CC was performed on Merck silica gel 60 (Aldrich, 230-400-mesh ASTM), and HPLC analyses on a Perkin-Elmer series 200 chromatograph using a 250 × 19 mm i.d. SunFire prep silica gel column (Waters, Ireland), using CHCl3 (A) and CH3CN (B) as the mobile phase, gradient 100% A → 100% B in an overall time of 40 minutes, at a flow rate of 5 mL/min, with a UV/VIS detector (Perkin-Elmer series 200, USA) at 360 nm, at 25°C. 1H (400 MHz) and 13C (100 MHz) NMR measurements, including gCOSY, gHSQC, and gHMBC experiments, were made on either a Bruker Ascend 400 or a Varian VNMRS spectrometer from CDCl3 solutions, using the remaining signal of non-deuterated solvent centered at 7.26 ppm as the internal standard. Optical rotations were verified in CHCl3 on a Perkin-Elmer 341 polarimeter. Mass spectra were recorded on a MicrOTOF II (Bruker Daltonics) using an electrospray ionization (ESI) source at 70 eV. A MeOH-CHCl3-formic acid mixture (1:1:0.002) was used as a white sample, and a detection range of 50 to 1000 m/z was set. High-purity nitrogen was used as the nebulizing gas at a pressure of 1.0 bar and as the drying gas at a rate of 8.0 L/min with a capillary voltage of 4 500 V. The software Compass for OtofSeries ver. 1.5.3 (Bruker Daltonik GmbH) was used for data acquisition, and the source was kept at 200°C during acquisitions.
Plant Material and Extracts
Specimens of J. dioica var. dioica Sessé ex Cerv. (Euphorbiaceae) were collected during the dry season, in February 2015, in Tetepango, a town located in the State of Hidalgo (20°06'38" N, 99°09'11" W, 2100 MASL); the area has a cumulative rainfall of 543 mm and a mean annual temperature of 16.3°C. The specimens were identified and preserved in the Herbarium of the Autonomous University of Hidalgo (voucher number: AD Hernandez Fuentes 01). Roots and stems were washed with distilled water, separated, and dried at room temperature, in the dark, for 30 days. Then, the material was stored in resealable bags (Ziploc, SC Johnson, USA) and kept in the dark at 4°C in a refrigerator (Shel Lab HC30R, Sheldon Manufacturing, Inc., USA). Dried, ground stems (258 g) were extracted with hexane (2 L) and then with EtOAc (2 L) under reflux for 3 hours. Filtration and evaporation on the extract produced brown viscous oil samples, 3.85 and 3.11 g, respectively. This procedure was repeated with the roots (796 g), using hexane (4 L) and EtOAc (4 L), to obtain 14.20 and 11.71 g of brown oil samples, respectively.
Antioxidant and Antifungal Activity
Antioxidant activity was determined by DPPH· free-radical scavenging capacity 14 and ABTS·+ assays. 15 Hexane and EtOAc extracts (50 mg) from stems and roots were mixed with hexane (10 mL) and vortexed for 5 minutes. The samples were sonicated at 40 kHz in an ultrasonic bath (Ultrasonic Cleaner, Mod. 32V118A, Illinois, USA) in three 10-minute intervals with two 5-minute intersonication periods, and then centrifuged at 10 000 × g for 10 minutes at 5°C. To perform the DPPH assay, 0.3 mL of supernatant was added to 2.7 mL of a DPPH· cold methanolic solution 6 × 10−5 M and allowed to stand in complete darkness for 60 minutes at 4°C. Then, the absorbance at 515 nm was measured. For the ABTS method, the radical cation (ABTS·+) was obtained by mixing 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) 7 mM with potassium persulfate 2.45 mM and incubated at 25°C in the dark for 16 hours. Once formed, the ABTS·+ radical was diluted in MeOH to an absorbance of 0.700 (±0.1) at 734 nm. The ABTS·+ solution was mixed with the supernatant and stored in the dark for 6 minutes; then, absorbance at 734 nm was measured. Results were expressed as percentage of ABTS·+ and DPPH· inhibition according to equations (1) and (2), respectively.


where Cabs is the absorbance of the control and Sabs is the absorbance of the sample.
Alternaria alternata, Sclerotium rolfsii, Colletotrichum gloeosporioides, Rhizoctonia solani, and Fusarium oxysporum were used as model phytopathogenic fungi. A quantitative determination of the antifungal activity of hexane and EtOAc root extracts was performed as described in. 16 The effect of the treatments on the radial growth of phytopathogenic fungi was assessed using extract solutions at 250 µg/mL, dissolved in 0.5% DMSO. Five milliliters of each extract solution was mixed with 1% PDA, poured into 5 cm Petri dishes, and allowed to solidify. Then, a 0.5-cm-diameter inoculum of the test microorganism, previously incubated in PDA for 10 days, was placed in the center of each Petri dish and incubated from 1 to 4 days, except for S. rolfsii, which was incubated for 7 days at 28°C under a 12-hour light/darkness cycle. At the end of the incubation period, the diameter of the mycelial colony in each phytopathogenic species replicate was measured in 2 perpendicular directions. Inhibition percentage was determined with respect to the growth of controls cultured in parallel. One percentage PDA and 1% PDA-0.5% DMSO were used as negative controls, while the fungicide Captan® at 250 µg/mL was used as a positive control. Antioxidant and antifungal activities were studied in a completely randomized experimental design. The results were analyzed by one-way analysis of variance (α = 0.05), and significant differences among treatments were analyzed by the Tukey’s test, using the software SAS 9.0.
Phytochemical Studies
The addition of MeOH (100 mL) to stem hexane and EtOAc extracts, followed by heating at 50°C, standing at 0°C for 12 hours, and filtration, yielded fatty residues (1.03 and 1.1 g, respectively). Both residues were combined and subjected to separation by CC using silica gel as the stationary phase, and hexane-CHCl3 (1:1, 250 mL) and CHCl3-EtOAc (19:1, 250 mL) as eluents. Fractions of different polarity were collected and analyzed by thin-layer chromatography (TLC) and 1H NMR. Fatty acids and fatty alcohols were identified in the fraction eluted with hexane-CHCl3, while fatty alkyl esters of acetic and ferulic acid (200 mg) were identified in the CHCl3-EtOAc fraction. This latter fraction was dissolved in CHCl3 (3 mL) and purified by HPLC, as described in the general section, to afford 3 fractions, A, B, and C. 1-Octacosyl acetate (
After the addition of MeOH at 50°C to the root hexane extract (6.6 g), and letting it stand at 0°C for 12 hours before filtering and concentrating in a rotary evaporator under vacuum, a defatted extract (4.0 g) and a fatty fraction (1.138 g) were obtained. When hexane-EtOAc (9:1, 10 mL) was added to the defatted extract a white precipitate was obtained, which was then separated by decantation and crystallized from CHCl3-MeOH to obtain citlalitrione (
Footnotes
Acknowledgments
The authors thank M.C. Victoria Ayala Escobar (Phytopathogenic Fungi Laboratory, Postgraduate School) for donating the fungal strains. J.G.T. thanks CONACYT-Mexico for fellowship grant No. 228146.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for theresearch, authorship, and/or publication of this article: This study was partially supported by CONACYT-Mexico (grant No. 238206) and FOMIX-CONACYT-Mexico (grant No. 195462)..
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
