Abstract
Until recently, there was a lack of information on the phytochemical composition of the taxon Senna. Recent reports show that Senna splendida, Senna gardneri, and Senna georcica are characterized by profiles dominated by flavonoids, naphthapyrones, and stilbenes. Here, we studied the phytochemical composition of Senna macranthera var pudibunda (Benth) Irwin & Barneby for comparison. A total of 26 polyphenolic compounds were identified in methanol extracts of various botanical parts of Senna macranthera var pudibunda (Benth.) Irwin & Barneby. The higher concentration (on a dry weight basis) of polyphenolic compounds was detected in the leaves (48.55 g/kg), bark (21.26 g/kg), and roots (17.08 g/kg), whereas a lower concentration of polyphenolic compounds was detected in the fruits (6.67 g/kg). The polyphenolic profiles of the various botanical parts were dominated by flavan-3-ols and flavan-3-ol dimers. The bark and roots contained only these components (100%), whereas flavonoids were also identified as minor components in the leaves (15.7%) and the fruits (5.6%), respectively. The flavan-3-ol conjugates comprised the well-known procyanidins, along with the rarely reported afzelechin, giobourtinidol and procassinidin dimers.
Keywords
There are abundant reports on the polyphenolic profiles of the genus Cassia, 1 which originally also contained the genus Senna. However since the genera were separated, 2 into both Cassia and Senna genera, there are only a few published reports of the phytochemical composition of the latter. Before this separation, several health promoting properties were published for the Cassia taxon (including Senna species) namely anticancer, antioxidant, antimicrobial, anti-inflammatory, and hepatoprotective effects among others. 3 Recently, we have shown that Senna splendida (Vogel) H.S Irwin & Barneby 4 and Senna gardneri Irwin & Barneby, and Senna georgica Irwin & Barneby 5 are characterized by polyphenolic profiles dominated by flavonoids, naphthapyrones and stilbenes.
Previous to this, the following compounds were detected in Cassia pudibunda namely 10-demethyl flavasperone, 10-demethylflavasperone-10-sulphate, 10-demethylflavasperone-10-O-β-d-apio-furanosyl-(1-6)-O-β-d-glucopyranoside and cassiapyrone-10-sulphate (7-methyl-10-demethylflavasperone-10-sulphate) 6 as well as rubrofusarin-6-O-β-d-glucopyranoside, quinquangulin-6-O-β-d-apiofuranosyl-(l-6)-O-β-d-glucopyranoside, quinquangulin-6-O-β-d-glucopyranoside and chrysophanol dimethyl ether, chrysophanol, physcion, cis-3, 3′, 5, 5′-tetrahydroxy-4-methoxy-stilbene, trans-3, 3′, 5, 5′-tetrahydroxy-4-methoxystilbene, and cassiaside B. 7 Of these, only 2 were detected by us in Senna splendida 4 namely rubrofusarin-6-O-β-d-glucopyranoside (rubrofusarin glucoside), and trans-3, 3′, 5, 5′-tetrahydroxy-4-methoxystilbene (methoxy oxoresveratrol). Here, we report on the identification and quantitation of polyphenolic compounds in various botanical parts of Senna macranthera var pudibunda (Benth.) Irwin & Barneby (Senna macranthera pudibunda).
The yields, following Soxhlet extraction with methanol, of the various botanical parts of Senna macranthera pudibunda (10 g) after delipidation with hexane were for the bark (1.72 g), for the fruits (1.36 g), for the leaves (4.03 g), and for the roots (1.35 g), respectively. High-performance liquid chromatography (HPLC) of the bark, leaves, and root methanol extracts are depicted in Figures S1-S3.
A total of 26 polyphenolic compounds were detected and identified in the methanol extracts of the botanical parts of Senna macranthera pudibunda based on their HPLC-electrospray ionization (ESI)-mass spectrometry (MS) data, in both negative and positive ion modes (Tables 1 and 2). Their structures are depicted in Figures S4-S6. The identification of the flavan-3-ols, procyanidins, and flavonoids was unequivocal by comparison to commercially available authentic standards, based on their UV spectra, HPLC-ESI-MS mode peaks, and fragmentation patterns (Table 2). In addition, the structures of the flavan-3-ols was further confirmed by gas chromatography (GC)-electron ionization (EI-MS) (Table 3). The HPLC-ESI-MS fragmentation patterns of the afzelechin and guibourtinidol dimers (the structure of
HPLC-ESI-MS Data in Negative and Positive Ion Mode at a Fragmentor Voltage (100 V) of Polyphenolic Compounds Detected in Methanol Extracts of Various Botanical Parts of Senna macranthera Pudibunda.

HPLC-ESI-MS Data in Negative-Ion Mode at a Fragmentor Voltage (300 V) of Polyphenolic Compounds Detected in Methanol Extracts of Various Botanical Parts of Senna Macranthera pudibunda.

*Base peak.
Gas Chromatography (GC)-electron ionization (EI)-MS Data for the TMS-Ether Derivatives of the Flavan-3-Ols Detected in the Botanical Parts of Senna Macranthera pudibunda.
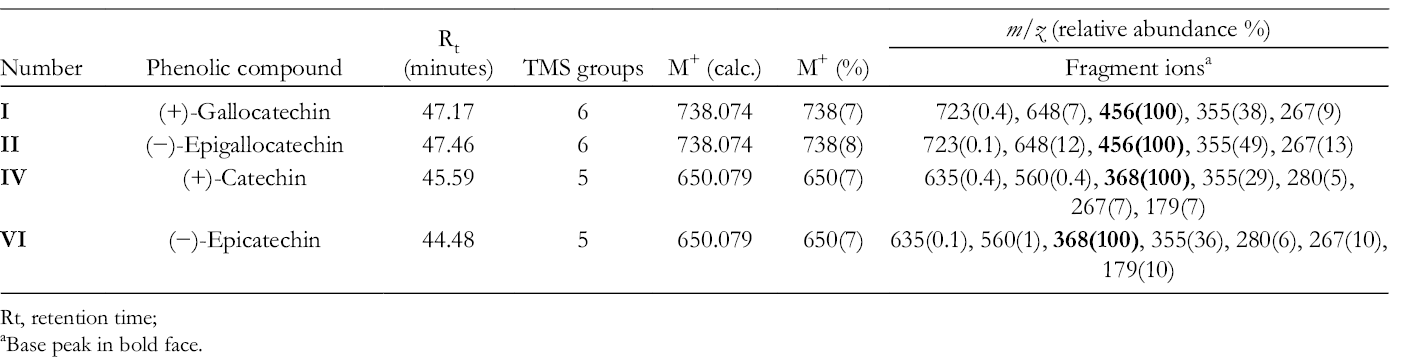
Rt = retention time.
aBase peak in bold face.
Of the polyphenolic compounds, 40% were detected in 1, 44% were detected in 2, and only 16% in 3 botanical parts. None were common to the 4 botanical parts studied. The concentration of the flavan-3-ols, procyanidins, and flavonoids in the methanol extracts were calculated against standard curves of authentic commercial standards. However, the concentrations of afzelechin, guibourtinidol. and cassiaflavan dimers were calculated against a standard curve of procyanidin B1 with relevant molecular weight corrections.
Based on the standard curve data (Table S1), the higher concentration (on a dry weight basis) of polyphenolic compounds was detected in the leaves (48.53 g/kg), bark (21.28 g/kg), and roots (17.10 g/kg), whereas the concentration in the fruits (6.66 g/kg) was considerably lower (Table 4). The polyphenolic profiles of the various botanical parts of Senna macranthera pudibunda were dominated by flavan-3-ols and flavan-3-ol dimers. The bark and roots contained only these phytochemicals (100%), whereas in addition other flavonoids were also identified as minor components in the leaves namely (
Concentration (G/kg Dry Weight) of Polyphenolic Compounds Detected in Methanol Extracts of Various Botanical Parts of Senna Macranthera pudibunda.

nd, not detected; SMB, S . macranthera pudibunda bark; SMF, S . macranthera pudibunda fruits; SML, S . macranthera pudibunda leaves; SMR, S . macranthera pudibunda roots.
*
*COV% of duplicate extractions and work-up procedures <5.
The major component (Table 4) in Senna macranthera pudibunda bark was cassiaflavan-(Cat) Epicatechin-1 (5.47 g/kg), in the fruits cassiaflavan-(Cat)Epicatechin-1 (1.60 g/kg), in the leaves cassiaflavan-(Cat)Epicatechin-4 (8.34 g/kg), and in the roots cassiaflavan-(Cat)Epicatechin-1 (5.62 g/kg). The composition (%) of flavan-3-ols, procyanidins, afzelechin, guibourtidinol and procassinidin dimers, plus other flavonoids detected in methanol extracts of the botanical parts of Senna macranthera pudibunda is shown in Table 5.
Composition (%) of Flavan-3-Ols, Procyanidins and Afzelechin, Guibourtidinol, Procassinidin Dimers and Other Flavonoids Detected in Methanol Extracts of Various Botanical Parts of Senna Macranthera pudibunda.

Previously, the polyphenolic profiles of the various botanical parts of Senna splendida 4 were published. The composition of polyphenolic compounds in the bark, flowers, and leaves of Senna splendida were dominated by flavonoids at >98%. However in the roots, the major polyphenolic components were naphthopyrones (54%) and stilbenes (39%). Similar findings for Senna gardneri and Senna georgica were also reported recently. 5
The profiles of the polyphenolic compounds in the various botanical parts of Senna macranthera pudibunda are by contrast very different from other Senna species reported previously by us. 4,5 The polyphenolic composition of all botanical samples was dominated by flavan-3-ols and flavan-3-ol dimers. The bark and roots contained only these compounds, whereas in the fruits and leaves, other flavonoids were also detected (6.73 and 5.66% respectively).
The flavan-3-ol dimer profiles present in Senna macranthera pudibunda initially appeared to be novel. However, the components of the methanol extracts have considerable concordance with a very recent report by Sobeh et al. 9 Sobeh et al 9 studied the phytochemical profile of methanol extracts of Senna singueana Delile, which is a traditional African medicinal plant, and reported the presence of 36 phytochemicals. In our study, 26 polyphenolic compounds were detected in the botanical parts of Senna macranthera pudibunda of which 13 were also identified in the bark of Senna singueana Delile by Sobeh et al 9 We identified 4 apfelechin-(Cat)Epicatechin dimers, 7 guibourtidinol-(Cat)Epicatechin dimers, one of which we have previously definitively identified as (2R*, 3S*, 4S*, 2'' R*, 3'' S*-guibourtinidol-(4α-8)-catechin 8 and 4 cassiaflavan-(Cat)Epicatechin dimers in methanol extracts of Senna macranthera pudibunda. Of relevance to this study, Sobeh et al, 9 on the other hand, identified 1 apfelechin-(Cat)Epicatechin dimer, 4 guibourtidinol-(Cat)Epicatechin dimers, and 2 cassiaflavan-(Cat)Epicatechin dimers) along with 7 gallo-(Cat)Epicatechin dimers in Senna singueana Delile extracts, the latter of which we did not detect in the botanical parts of Senna macranthera pudibunda. Furthermore, a series of procyanidin trimers were also identified in methanol extracts of Senna singueana Delile 9 but again were not detected in Senna macranthera pudibunda.
In conclusion, this is A very comprehensive report on the identification and quantitation of polyphenolic compounds in a member of the Senna genus, namely Senna macranthera pudibunda. As far as we are aware, there are no previous substantive reports on the identification of the polyphenolic compounds present in this species and, certainly no quantitative data. Botanical parts of Senna macranthera pudibunda are shown to contain predominantly a complex profile of flavan-3-ol dimers of the afzelechin, guibourtinidol, and procassinidin types, representing a loss of 1, 2, and 3 hydroxyl groups from the apex unit of the generally far more common procyanidins B1 and B2. This is in complete contrast to the polyphenolic profiles described in Senna splendida (Vogel) Irwin & Barneby, 4 Senna gardneri Irwin & Barneby and Senna georgica Irwin & Barneby 5 which were dominated by flavonoids in the flowers, leaves and bark and naphthopyrones and stilbenes in the root extracts.
Therefore, it would be of interest to establish, why the profiles of polyphenolic compounds in the botanical parts of Senna macranthera pudibunda are so completely different from those of Senna splendida, 4 Senna gardneri and Senna georgica 5 given that they were harvested from the same county namely Ceara, in northeast Brazil.
Experimental
Chemicals and Reagents
(+)-Catechin, (−)-epicatechin, (−)-epigallocatechin, (+)-gallocatechin, procyanidin B1, procyanidin B2, quercetin-3-O-glucoside (isoquercitrin), kaempferol-3-O-rhamnoglucoside, and rutin were obtained from Extrasynthese, (Lyon Nord, Genay, France); quercetin-3-O-galactoside from Sigma Aldrich (Diesenhofen Germany); n-hexane, acetic acid, and acetonitrile from Fluka/Riedel de Haen (Seelze, Germany); methanol from Karl Roth (Karlsruhe, Germany); BSTFA and DMSO from Merck (Darmstadt, Germany) and Sep-Pak C18 cartridges for solid-phase extraction (Supelclean™ LC-18 SPE, 5000 mg) from Supelco (Bellefonte, PA, USA). All solutions were made up in methanol or doubly distilled water.
Plant Material
The botanical parts of Senna macranthera pudibunda plants were collected in the Tiangua region of Ceara, Brazil and identified by L.W. Lima Verde at the Biology Department, Universidade Federal do Ceará (UFC). Voucher specimens are deposited in the Prisco Bezerra Herbarium, UFC, Brazil with the number 54 170.
Soxhlet Extraction of Senna Macranthera Pudibunda Botanical Parts
The leaves, roots, bark, and fruits of Senna macranthera pudibunda were freeze-dried to constant weight and pulverized to a fine homogeneous powder. Dried material (10 g × 2) was extracted with n-hexane in a Soxhlet apparatus (3 hours) to remove lipids. After drying under a stream of nitrogen, the solids were further extracted with methanol (3 × 3 hours) as described by Owen et al 12 Organic solvent was removed by rotary evaporation at 35°C in vacuo.
Column Chromatography on C18
Column chromatography on C18 was conducted as described by Pfundstein et al. 13
High Performance Liquid Chromatography-Electrospray-Ionization Mass Spectrometry
HPLC-ESI-MS was conducted as described by Maia et al. 4
Gas Chromatography Coupled with Electron Ionization Mass Spectrometry
Analyses in the scan mode were performed as described by Maia et al. 4
SemiPreparative HPLC
Semipreparative HPLC was conducted on a HP 1100 liquid chromatograph (Agilent Technologies, Waldbronn, Germany) as described by Maia et al. 4
Supplemental Material
Supplementary data - Supplemental material for Characterization and Quantitation of Polyphenolic Compounds in Senna macranthera var pudibunda From the Northeast of Brazil
Supplemental material, Supplementary data, for Characterization and Quantitation of Polyphenolic Compounds in Senna macranthera var pudibunda From the Northeast of Brazil by Irvila Ricarte de O. Maia, Maria Teresa Salles Trevisan, Maria Goretti de V. Silva, Andrea Breuer, and Robert W. Owen in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to the Federal University of Ceara (UFC) and German Cancer Research Center (DKFZ) for their support with analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by CAPES/PDSE and CNPq.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
