Abstract
Psathyrelloic acid (
Diterpenoids from fungi have structural and biological diversity, which attracted great attention of both organic and biological researchers.
1
-3
The basidiomycete Psathyrella candolleana, belonging to the family Psathyrellaceae, is commonly found on lawns or pastures in Europe and North America. It is also widely distributed in Yunnan and Sichuan provinces of China. It is used as a medicinal mushroom in Iraq, the extract of which could provide protection against DNA oxidative damage induced by doxorubicin.
4,5
So far, the chemical constituents of this species are seldom reported. Only a number of biologically active guanacastane-type diterpenoids were isolated from the fermentation broths.
6
In our continuous search for structurally interesting and biologically active natural products from higher fungi,
7
-11
a novel diterpenoid, psathyrelloic acid (

Psathyrelloic acid (
Psathyrelloic acid (1) has a molecular formula C20H30O5 as determined by positive high-resolution electrospray ionization mass spectrometry (HRESIMS), which showed a molecular ion peak at m/z 373.19843 (calcd for C20H30O5Na, 373.19909), corresponding to 6 degrees of unsaturation. The infrared (IR) spectrum indicated the presence of hydroxy (3483 cm−1), carbonyl (1708 cm−1), and double bonds (1623, 1593 cm−1). The 1D nuclear magnetic resonance (NMR) spectra (see Supplementary material), as well as the heteronuclear single-quantum coherence (HSQC) spectrum, revealed 4 methyls, 5 methylenes, 5 methines, and 6 quaternary carbons (Table 1). Three doubles (δ
C 119.2, 128.0, 132.7, 139.8, 140.5, 152.8) and 2 carbonyl carbons (δ
C 170.2, 170.9) suggested that compound
1H (600 MHz) and 13C (150 MHz) NMR Data of 1 a in Methanol-d 4 (δ in ppm).

NMR, nuclear magnetic resonance; HSQC, heteronuclear single-quantum coherence; HMBC, heteronuclear multiple bond correlation; COSY, correlation spectroscopy; ROESY, rotating frame nuclear Overhauser effect spectroscopy.
On analyzing the 1H-1H correlation spectroscopy (COSY) spectrum, 4 fragments were established as shown in Figure 2. Based on the 1H-1H COSY data, a hydroxymethylene group for CH2-14 was identified by the NMR (δ
H 4.25; δ
C 58.4) and mass spectroscopy (MS) data. Two downfield shifts for 2 olefinic protons at δ
H 6.74 and 6.75, as well as the corresponding carbon resonances at δ
C 139.8 (d, C-3) and 140.5 (d, C-13), together with 2 carbonyl carbons at δ
C 170.9 (s, C-1) and 170.2 (s, C-19), suggested the existence of 2 α,β-unsaturated carbonyl moieties. They were further identified as 2-methyl-2,3-unsaturated-1-carbonyl and 12,13-unsaturated-19-carbonyl, respectively, by the heteronuclear multiple bond correlation (HMBC) correlations from H-15 to C-1, C-2, and C-3, as well as from H-13 to C-19. According to the MS data, as well as the NMR data at δ
C 170.9 and 170.2, 2 carboxylic acid groups were identified for C-1 and C-19, respectively. In addition, a singlet for protons of a methyl at δ
H 1.08 (3H, s, H-20) showed key HMBC correlations to C-5, C-6, C-9, and C-4. This information, together with an HMBC correlation from H-8 to C-9, established a 5-membered carbon ring. This data, along with the long fragment as revealed by the 1H-1H COSY data, established an isopropyl connected to C-8. The third double bond between C-9 and C-10 determined the same. From HMBC correlations from H-11 to C-12 and C-19, the connection between C-11 and C-12 was established. Similarly, the connection between C-4 and C-5 was indicated by the HMBC correlations from H-4 to C-5 and from H-20 to C-4. So far, the planar structure of
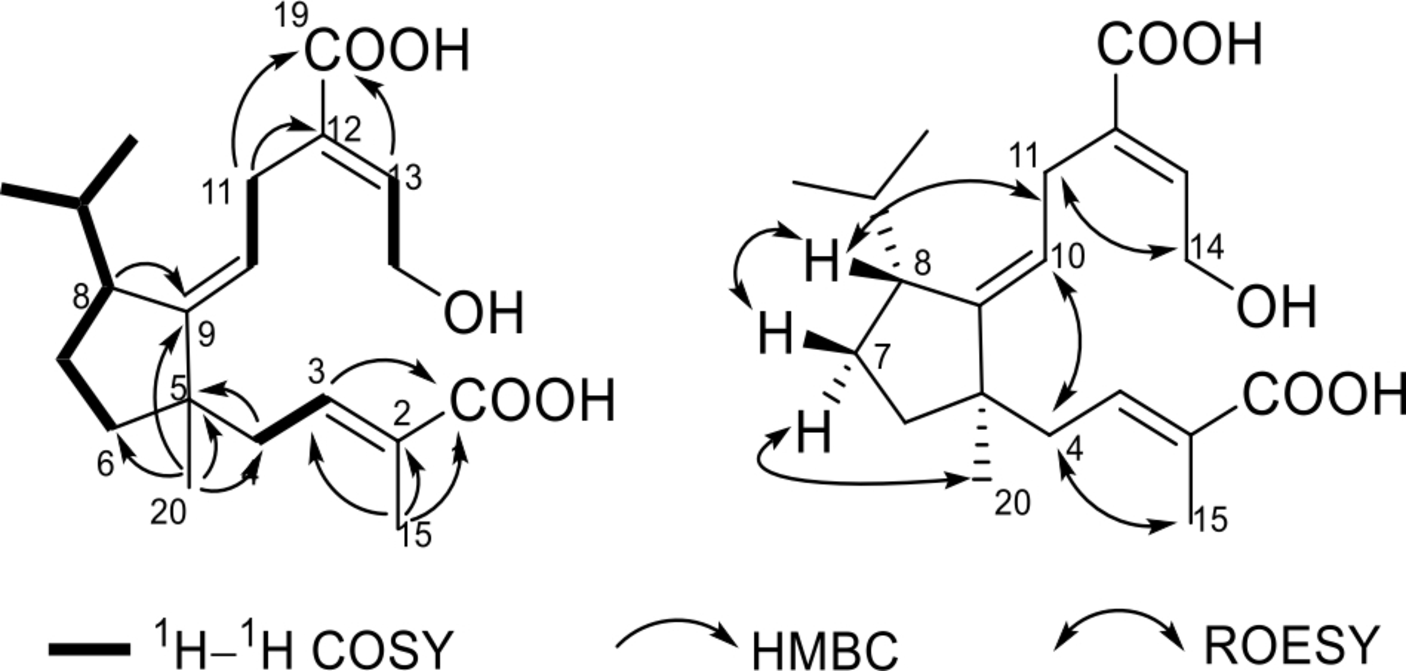
Key 2D NMR correlations of 1. 2D NMR, two-dimensional nuclear magnetic resonance.
A ROESY experiment was used for the determination of relative configuration. At first, 3 double bonds were all elucidated as E form by the ROESY correlations of H-15/H-4, H-8/H-11, and H-11/H-14. In addition, a correlation of H-8 with H-7b, not observed for H-8 with H-7a, together with a correlation of H-20 with H-7a indicated that Me-20 and H-8 are in the opposite side. The structure of
Structurally, compound
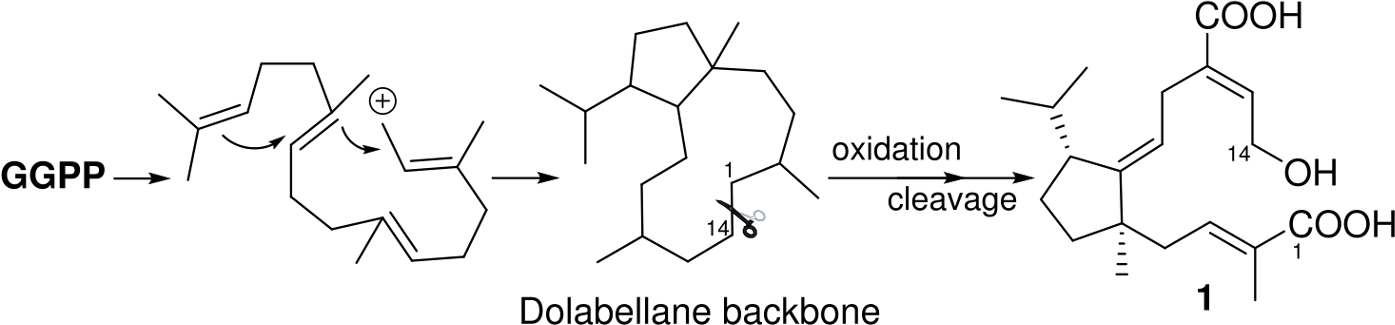
Proposed biosynthesis pathway for 1.
Compound
In summary, the chemical investigation on the cultures of the edible mushroom P. candolleana resulted in the isolation of a novel monocyclic diterpenoid, psathyrelloic acid (
Experimental
General
Optical rotations were measured on a Rudolph Autopol IV polarimeter (Rudolph Technologies, Wilmington, USA). UV spectra were obtained on a UH5300 UV-VIS Double Beam Spectrophotometer (Hitachi High-Technologies (Shanghai) Co., Ltd., Shanghai, China). IR spectra were obtained by using a Shimadzu IRTracer-100 Fourier Transform Infrared spectrometer (Shimadzu, Kyoto, Japan) with KBr pellets. NMR spectra were acquired with a Bruker Avance III 600 instrument (Bruker, Germany). HSESIMS was measured on a Thermo Scientific Q Exactive Orbitrap MS system (Thermo Fisher, MA, USA). Silica gel (200-300 mesh and 80-100 mesh, Qingdao Marine Chemical Inc., Qingdao, China), RP-18 gel (40-75 µm, Fuji Silysia Chemical Ltd., Kasugai, Japan), and Sephadex LH-20 (Amersham Biosciences, Uppsala, Sweden) were used for column chromatography (CC). Fractions were monitored by thin-layer chromatography (Qingdao Marine Chemical Inc., China) and spots were visualized by 10% H2SO4 in methanol, in combination with Agilent 1260 series HPLC system (Zorbax SB-aq-C18 column, 5 µm, 6.4 × 150 mm). Preparative HPLC was performed on an Agilent 1260 series with a Zorbax SB-C18 (5 µm, 9.4 × 150 mm) column.
Fungal Material and Cultivation Conditions
Fruiting bodies of P. candolleana were collected at Jingdong, Yunnan Province, China, in 2003 and identified by Prof Zhu-Liang Yang (Kunming Institute of Botany). The voucher specimen (No. CGBWSHF00118) was deposited at the herbarium of Kunming Institute of Botany. 6 Culture medium was composed of glucose (5%), pork pepton (0.15%), yeast (0.5%), KH2PO4 (0.05%), and MgSO4 (0.05%). Initial pH was adjusted to 6.0; the fermentation was first carried out on an Erlenmeyer flask for 6 days till the mycelium biomass reached to the maximum. Then it was transferred to rice medium at 24°C in dark culture for 40 days. Rice medium: 50 g of rice, 50 mL of water, placed in a 250 mL Erlenmeyer flask, sterilized at 121°C for 15 minutes, a total of 180 bottles.
Extraction and Isolation
The rice culture (9 kg) was extracted 4 times with EtOAc. The organic layer was evaporated to give a crude extract (90 g). Then it was subjected to silica gel CC (200-300 mesh) eluted with petroleum ether (PE)-Me2CO gradient system to afford 8 fractions A to H. Fraction G (8 g) was first isolated by silica gel CC (200-300 mesh) eluted with PE-Me2CO (2/1) to give subfractions G1 to G7. Fraction G5 (1.2 g) was separated by preparative MPLC with a reversed-phased C18 column (MeOH-H2O, 10%-100%) to give fractions G5a to G5e; then fraction G5e (200 mg) was applied to Sephadex LH-20 CC (Me2CO) and purified by HPLC (MeCN-H2O, 20%-40%, 3 mL/min) to give
Antibacterial Assay
The tested bacteria strains E. coli ATCC25922, S. aureus subsp. aureus ATCC29213, S. enterica subsp. enterica ATCC14028, and P. aeruginosa ATCC27853 were purchased from China General Microbiological Culture Collection Center (CGMCC). All these strains were cultured in Mueller Hinton broth (MHB) (Guangdong Huankai Microbial Sci. &Tech. Co., Ltd, Guangzhou, China) at 37°C overnight with shaking (200 rpm). A sample of each culture was then diluted 40-fold in fresh MHB broth and incubated with shaking (200 rpm) at 37°C for 2 to 3 hours. The resultant mid-log phase cultures were diluted to a concentration of 5 × 105 CFU/mL, then 50 mL was added to each well of the compound-containing plates, giving a final compound concentration range of 128 or 50 mg/mL. Plates were covered and incubated at 37°C for 24 hours. Inhibition rates were determined using photometry at OD625 nm. Penicillin G sodium salt (Biosharp, Shanghai, China) and ceftazidime (Shanghai Yuanye Biotechnologies, Shanghai, China) were used as positive inhibitor controls.
Psathyrelloic Acid (1)
Colorless oil, purity >95% (HPLC).
[α]20 D: +101.3 (c 0.5 MeOH).
IR (KBr) v max: 3483, 2972, 2932, 1708, 1623, 1593, 1474, 1381, 1298, 1090, 1025, 960 cm−1.
UV (MeOH) λ max (log ε): 202 (3.61), 218 (3.70), 262 (3.16) nm.
1H and 13C NMR data: see Table 1.
HRESIMS (pos.): m/z 373.19843 (calcd for C20H30O5Na, 373.19855).
Footnotes
Acknowledgments
The authors thank the Analytical & Measuring Center, School of Pharmaceutical Sciences, South-Central University for Nationalities for the spectra test.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (81872762, 81773590, 31870513, 81561148013), the National Key Research and Development Program of China (2017YFC1704007), the Thailand Research Fund (grant No. DBG5980002), the Key Projects of Technological Innovation of Hubei Province (No. 2016ACA138), and the Fundamental Research Funds for the Central University, South-Central University for Nationalities (CZP18005, CZQ17010, CZQ17008, CZT18014, CZT18013).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
