Abstract
Myrtus communis L. is a shrub growing wild in most countries all around the Mediterranean Sea and myrtle leaf oil is useful in the perfumery and pharmaceutical industries. Nowadays, “chemotyped” essential oils are more and more appreciated. Myrtle oil from eastern and central-north Algeria belong to the “α-pinene-cineole” chemotype. In contrast, Moroccan myrtle oil displayed appreciable content of myrtenyl acetate. Therefore, it appeared crucial to investigate the composition of myrtle leaf oil from northwestern Algeria in order to ensure the homogeneity of the composition of Algerian myrtle leaf oil. The chemical composition of 42 oil samples of M. communis isolated from leaves collected in five locations from Tlemcen Province was investigated by gas chromatography (GC) in combination with retention indices, GC-mass spectroscopy, and 13C nuclear magnetic resonance spectroscopy. Yields ranged between 0.13% and 1.04% (w/w). The chemical composition of the oils was largely dominated by monoterpene hydrocarbons, with α-pinene (24.3-59.0%) and 1,8-cineole (13.2-49.5%) being the major compounds followed by limonene (3.2-19.8%). The 42 compositions were submitted to statistical analyses. The combination of hierarchical cluster analysis and principal component analysis allowed the distinction of two groups, the second group being subdivided into two subgroups. Groups and subgroups were differentiated with respect to their contents of α-pinene and 1,8-cineole and, to a lesser extent, of limonene. This study ensures that Algerian myrtle leaf oil belongs to the “α-pinene-cineole” chemotype which displays various biological activities. The observed homogeneity could be a positive point to join the international market of essential oils.
Keywords
Despite the importance, the abundance, and the diversity of its flora, Algeria imports most of the essential oils (EOs) and plant extracts used in the country for perfumery, food flavoring, or pharmaceutical purposes. Various projects based on the valorization of Algerian aromatic and medicinal plants at artisanal or industrial scale were born. 1,2 Nowadays, quality of commercialized EOs is a crucial parameter. “Chemotyped” EOs are more and more appreciated, particularly in the domain of aromatherapy. 3
Myrtus communis L. grows wild in most countries all around the Mediterranean Sea. EO is isolated from different organs, leaves, flowers, and berries. Leaf oil has an industrial importance, due to its pleasant odor (perfumery) and biological activities (aromatherapy). Biological aspects of Algerian myrtle oil have been recently reported 4 and reviewed. 5
Various papers reported on the chemical composition of Algerian myrtle leaf oil. Almost all the papers described the composition of a limited number (1-3) of samples and almost all the investigated oil samples contained α-pinene and 1,8-cineole as main components. 5 An overview of myrtle oil from eastern and central Algeria has been done. 6,7 Taking into account the relative contents of the two major components α-pinene and 1,8-cineole as well as those, at a lesser extent, of limonene and linalool, statistical analysis allowed the differentiation of two groups and two subgroups. 7 Similar results were reported for myrtle from Chlef Province. The composition of 11/14 oil samples was dominated by α-pinene and 1,8-cineole while linalool reached high contents for the last three samples. 8,9 Otherwise, a few atypical samples have been reported. For instance, an oil sample from Tizi Ouzou contained 1,8-cineole and cis-geraniol as main components. 10 A sample from Forest of Bissa (Chlef) contained mainly limonene besides linalool, α-pinene, and geranyl acetate. 8,9 In all the reported compositions, myrtenyl acetate was absent or present at trace level. Surprisingly, it has been reported as major component (38.7%) of a myrtle leaf oil sample from Aïn Defla Province, 150 km south-west from Algiers. 11 Such a composition that looks really unusual in Algeria is classical for Moroccan myrtle leaf oils. 12-14
As a consequence, it appeared crucial to investigate the composition of myrtle leaf oils from northwestern Algeria, in order to get an overview of the composition of wild Algerian myrtle leaf oil. Plants have been collected in the province of Tlemcen near the Moroccan frontier. Indeed, it could be useful for EO producers and eventual exporters to describe a representative chemotype of Algerian myrtle leaf oil.
Leaves from 42 individual plants have been harvested in the following locations: Aïn Kebira, Beni Khellad, Beni Ouarsous, Honaïne, and Nedroma. Essential oil was isolated using a Clevenger-type apparatus. Yields varied substantially, between 0.13% and 1.04%. Highest yields (0.71%-1.04%) were observed at Beni Ouarsous, and lowest yields (0.13%-0.50%) at Beni Khellad. The 42 oil samples were submitted to gas chromatography with flame ionization detector (GC(FID)) analysis on two columns of different polarity, in combination with retention indices; 8 samples were also subjected to GC-mass spectrometry (MS) analysis and 7 samples to 13C nuclear magnetic resonance (NMR) analysis following a method developed at the University of Corsica. 15-17
In total, 30 individual components have been identified. In short, α-pinene (35.5%-59.0%) was the major component of 50% of the samples; 1,8-cineole (32.9%-46.5%) was the main component of 30% of the samples, the last 20% displayed similar contents of both compounds. The content of limonene varied drastically from sample to sample (3.2%-19.8%). Linalool ranged between 0.2% and 3.7% with an atypical sample having 12.7%.
Other components present at appreciable contents were α-terpineol (1.3%-4.3%), linalyl acetate (tr-2.8%), α-terpinyl acetate (0.0%-2.0%), geranyl acetate (1.1%-4.8%), and methyl eugenol (1.0%-7.3%). The composition of a representative oil sample, analyzed by GC with retention indices (GC(RI)), GC-MS, and 13C NMR, is reported in Table 1.
Chemical Composition of a Myrtle Leaf Oil Sample From Tlemcen Province.

aComponents are listed following their order of elution on apolar chromatographic column, (BP-1), percentages on apolar column except for compounds with * (BP-20).
bRIa, retention indices on apolar column.
cRIp, retention indices on polar column.
dTr, trace.
e NMR (italic), compound identified by NMR in another oil sample.
fDione, 3,3,5,5,8,8-hexamethyl-7-oxabicyclo[4.3.0]non-1(6)-ene-2,4-dione.
The 42 compositions were subjected to statistical analysis in order to distinguish clusters. The combination of hierarchical cluster analysis (HCA; dendrogram shown in Figure 1) and principal component analysis (PCA; plot shown in Figure 2), for which the first two axes accounted for 70.4% and 20.3% of the total variance of the population, respectively, suggested the existence of two principal groups, group II being subdivided into two subgroups IIA and IIB. Groups and subgroups were differentiated by their content of α-pinene and 1,8-cineole; however, limonene and methyl eugenol contributed to the differentiation (Table 2).
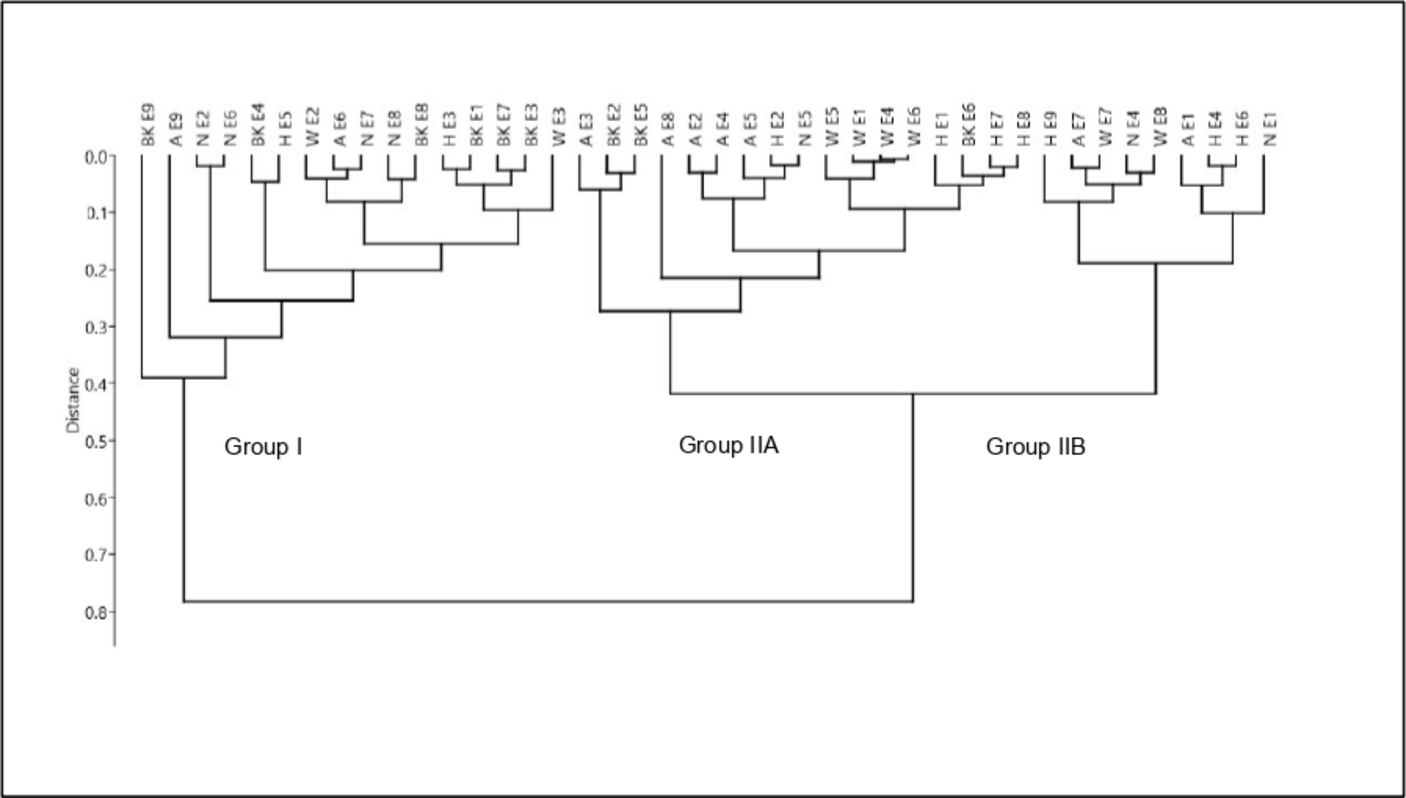
Dendrogram obtained by hierarchical cluster analysis of the 42 samples of myrtle essential oil from northwestern Algeria.
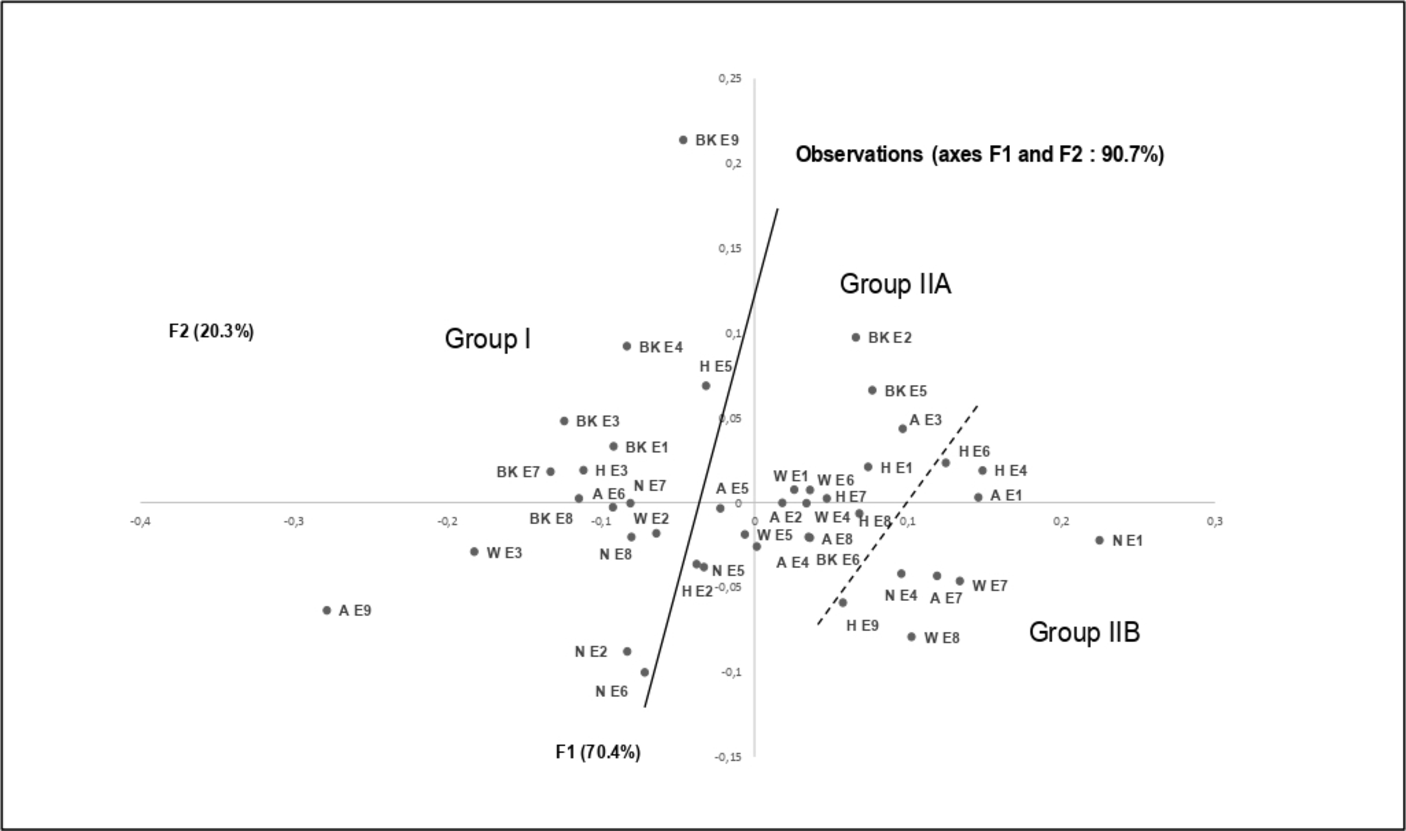
Principal component analysis of the 42 samples of Myrtus communis essential oil from northwestern Algeria.
Main Components of 42 Samples of Myrtle Essential Oil From Northwestern Algeria.
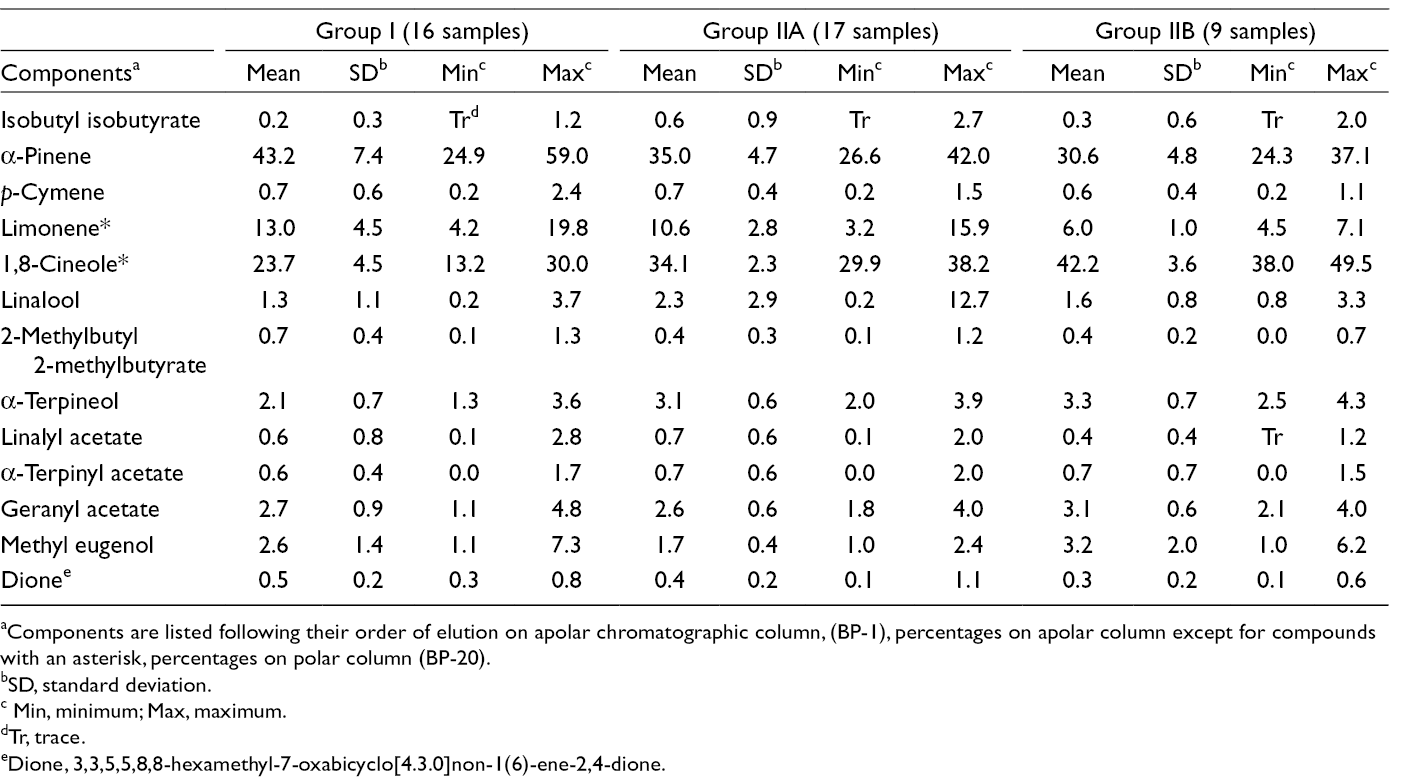
aComponents are listed following their order of elution on apolar chromatographic column, (BP-1), percentages on apolar column except for compounds with an asterisk, percentages on polar column (BP-20).
bSD, standard deviation.
c Min, minimum; Max, maximum.
dTr, trace.
eDione, 3,3,5,5,8,8-hexamethyl-7-oxabicyclo[4.3.0]non-1(6)-ene-2,4-dione.
The composition of samples of Group I (16/42 samples) was dominated by α-pinene (mean M = 43.2%, standard deviation SD = 7.4); the content of that compound reached 59.0% in one sample. Other components present at appreciable contents were 1,8-cineole (M = 23.7%, SD = 4.5) and limonene (M = 13.0%, SD = 4.5). Samples of subgroups IIA (17/42 samples) and IIB (9/42 samples) contained similar amounts of α-pinene (M = 35.0%, SD = 4.7 and M = 30.6%, SD = 4.8, respectively). They were differentiated by their contents of 1,8-cineole, more abundant in subgroup IIB (M = 42.2%, SD = 3.6 vs M = 34.1%, SD = 2.3) and limonene more abundant in subgroup IIA (M = 10.6%, SD = 2.8 vs M = 6.0%, SD = 1.0).
The compositions of α-pinene-rich oil samples of Group I isolated from plants harvested in northwestern Algeria resembled to those of samples of subroups IA and IB coming from plants growing wild in northeastern and central-north Algeria. 7 Samples of subgroup IIA containing about one-third of α-pinene as previously observed for some samples from eastern and central Algeria differed however by higher contents of 1,8-cineole or limonene. Finally, samples of subgroup IIB (9/42 samples) displayed a high percentage of 1,8-cineole that brought a slight difference with all other Algerian myrtle oil samples.
In conclusion, the composition of essential oils isolated from myrtle leaves harvested in northwestern Algeria completes previous results relative to the same plant growing wild in north-eastern and central-north Algeria and provides an overview of Algerian myrtle leaf oil. Although quantitative differences have been observed in the content of the main components, it could be assumed that Algerian myrtle leaf oil belongs to the “α-pinene-1,8-cineole” chemotype, also found in Tunisia, 13,18,19 Sardinia, 20 and Corsica. 21,22 In contrary, Algerian myrtle leaf oil differs drastically from oils containing myrtenyl acetate at appreciable content isolated from leaves harvested in Morocco, 12-14 Spain, 23 Portugal, 24 Italy, 25 ex-Yugoslavia, 13 Croatia, 26 Albania, 27 and Turkey. 28
The occurrence of the “α-pinene-1,8-cineole” chemotype of myrtle leaf oil from eastern to northwestern Algeria could be advantageous for its production, commercialization, and eventual exportation. Indeed, it has been demonstrated in vitro that Algerian myrtle leaf oil (“α-pinene-1,8-cineole” chemotype) displays various biological activities: antibacterial activity, 8,10,29,30 antifungal effect, anti-inflammatory potential (myrtle leaf oil was able to significantly inhibit nitric oxide production, without affecting cell viability), 4 antioxidant activity, 31 antimotility, and antidiarrheal activity. 32 Biological activity was also assessed on a pest destructive of stored products, Callosobruchus maculatus. 33 Therefore, a homogenous oil composition is obviously appreciated.
Experimental
Plant Material
Leaves of M. communis were collected in February-March 2018 from northwestern Algeria. Forty-two samples of individual plants were harvested in 5 stations at Tlemcen Province (Figure 3): Beni Khellad (BK1-9), Honaïne (H1-9), Beni Ouarsous (BO1-8), Nedroma (N1-7), and Aïn Kebira (AK1-9).

Harvesting locations of the leaves of Myrtus communis.
Identification of the plants was performed by Pr N. Benabadji (Laboratory of Ecology and Management of Natural Ecosystems, Tlemcen University). A voucher specimen has been deposited at the Laboratory of Natural Products, (Tlemcen University), under the accession number M. 1856.
Isolation of Essential Oils
The EOs were obtained from each sample by hydrodistillation of dried leaves (160-180 g) in a Clevenger-type apparatus for 2.5 hours. Essential oil yields were estimated on the basis of the dry weight of plant material.
GC (FID) and 13C-NMR Analyses
GC-MS Analysis
The EOs were analyzed with a Perkin-Elmer TurboMass detector (quadrupole), directly coupled to a Perkin-Elmer Autosystem equipped with a fused-silica capillary column (50 m × 0.22 mm i.d., film thickness 0.25 µm), BP-1 (dimethylpolysiloxane). Carrier gas, helium at 0.8 mL/min; split: 1/60; injection volume: 0.5 µL; injector temperature: 250°C; oven temperature programmed from 60 °C to 220 °C at 2 °C/min and then held isothermal (20 min); ion source temperature, 250°C; energy ionization, 70 eV; electron ionization mass spectra were acquired over the mass range 40 to 400 Da.
Identification of Components
Data Analysis
PCA and HCA (Ward’s method) were performed with Xlstat (Adinsoft, France). 35
Footnotes
Acknowledgments
This work was conducted within the cooperative program between the Universities of Abou Bekr Belkaïd (Tlemcen) and Pascal Paoli (Corsica).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: C. Bekhechi thanks University of Corsica for a research grant as associate professor (September 2018).
