Abstract
This research was carried out to assess essential oils (EOs), total phenolic content (TPC), total flavonoids (TFD), total flavonols (TFL), total chlorophyll, total carotenoids, total anthocyanins, and different antioxidant activity assays in two plant samples (leaf and bud) of Iberis amara L. The gas chromatography mass spectrophotometry (GC-MS) of the EOs and the head space (HS)-GC-MS of the methanolic leaf extracts identified 34 and 6 different compounds, respectively. The major components of the leaf EOs were carvacrol (9.4%), camphene (6.2%), р-cymene (4.3%), and eugenol (3.8%) respectively, whereas cumin aldehyde (10.4%) was the main component in the bud sample. 1-Butene, 4-isothiocyanate (50%) was identified as the main component in the HS-GC-MS analysis of leaves extract. The highest content of TPC (32.8 ± 0.7 mg GAE/gDW), TFD (28.4 ± 0.7 mg QE/gDW), TFL (11.8 ± 0.06 mg QE/gFW), and anthocyanin (0.4 ±0.02 µmol/g FW) was found in the methanolic extract of leaves. The highest antioxidant activity in the phosphomolibdate assay (628.3 ± 10 µg AAE/gFW) and the least activity in the 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay (IC50 = 415 ± 3.1 µg/mL) was found in leaf samples. The high ratio of monoterpene hydrocarbons and oxygenated monoterpenes of the EOs along with the high antioxidant activity propose the application of this medicinal plant for general or specific applications in food industries as a herbal plant.
Medicinal plants are well known to produce bioactive compounds, which can be used as natural antioxidants in pharmaceutical and food applications, 1 herbal beverages, fragrances, cosmetics, and as natural flavoring agents. 2 On the other hand, natural antioxidants are suitable agents which scavenge free radicals and reactive oxygen species (ROS) that are harmful to the DNA of cells. 1
Essential oils (EOs) represent a diverse and unique source of natural products utilized due to their different pharmaceutical properties, such as bactericidal and antiviral applications. 3,4
Iberis amara L. is a genus of flowering plants belonging to the family Brassicaceae, tribe Lepidieae, and subtribe Iberidinae. 5 It comprises annuals and perennials that are native to the Old World, including Europe, Caucasus, and Algeria. 6 It is commonly called candy tuft, derived from Candia, the earlier name of Iraklion on the island of Crete. 7 In Iran, it is only grown in the central regions. From a historical perspective, the plant appears to have been employed by ancients for curing gout, rheumatism, and other diseases. 8 In large doses, I. amara L. is useful for curing cardiac hypertrophy, asthma, and bronchitis. 6,7 Currently, the plant extract is employed in German phytomedicine and is called Iberogast. 8 In pharmacological studies, the fresh plant extract of I. amara exhibited a tonicising effect on the smooth muscles of the stomach, and can be used as a suitable drug for curing stomach disorders, especially irritable bowel syndrome. 6 The aerial parts of this plant contains glucosinolates, cucurbitacins (I and E), and flavonoglycosides. 8
Despite the large amount of information regarding different species in Brassicaceae family, research concerning biochemical traits of I. amara L. is very limited. No study has been done on the evaluation of the components of EOs in I. amara. Additionally, literature review showed no study regarding the evaluation of phenolic compounds and antioxidant activity of I. amara L. originating from Iran.
This research was conducted to (1) document the EO components of I. amara L. by gas chromatography mass spectrophotometry (GC-MS) and head space (HS)-GC-MS methods; (2) report the relative content of total phenolics, total flavonoids (TFD), total flavonols (TFL), carotenoids, and anthocyanin; and (3) report different antioxidant activities of I. amara L. using 3 different assay models (ie, DPPH assay, total antioxidant capacity [TAC] assay, and β-carotene/linoleic assay), with special focus on its natural antioxidant properties.
Gas Chromatography Mass Spectrophotometry and Head Space GC-MS Results
Analyzing volatile compounds require analytical methods which are able not only to evaluate the composition of the plant but also to detect the trace components of the plant or the extracts. 9 For this purpose, HS-GC-MS and GC-MS methods have been proven to be powerful for the identification of chemical constituents10. The mean yield of EOs was significantly more in the leaves (0.1% v/w) than that in the terminal buds (0.07% v/w) (Table 1). The color of EOs in the leaves was pale yellow, but it changed to semiturbid yellow in the buds. Gas chromatography mass spectrophotometry analysis of the EOs led to the identification of 33 compounds representing 94.3% and 88.4% of the total composition in the leaf and bud sources, respectively (Table 1). The variation in EOs composition could be affected by different factors specially explants source, 10 sample preparation method 3 plant growth stage, 4 edaphoclimatic conditions, 11 and season of collection. 12 The major components in the EO of leaf sample were carvacrol (9.4%), camphene (6.2%), р-cymene (4.3%), and eugenol (3.8%), whereas the ones in the bud were cumin aldehyde (10.4%), α-terpineol (4.7%), and isobornyl acetate (4.6%) (Table 1). Essential oils also contained the largest proportion of monoterpene hydrocarbons (22.6% in leaf to 24.1% in bud) followed by oxygenated sesquiterpenes (12.8% in leaf to 10.8% in bud) (Table 1). Other chemical groups were aldehydes (10.1% in the leaf to 16.2% in the bud), fatty acid amides (7.9% in the leaf to 1.2% in the bud), oxygenated diterpenes (13.1% in the leaf to 5.9% in the bud), oxygenated monoterpenes (9.1% in the leaf to 10.2% in the bud), aliphatic esters (3.6% in the leaf to 9.7% in the bud), and aromatic compounds (3.9% in the leaf and bud) (Table 1).
Volatile Compounds (%) of EOs of Studied I. amara Iranian Accession.

aCompounds are listed in order of their retention index from a HP-5 column.
bRelative proportions of the EO components expressed as percentages obtained by GC-MS responses (data ± SE).
Monoterpene hydrocarbons and oxygenated monoterpene compounds in the EO of I. amara could act as radical scavenging agents with greater antioxidant capacity, similar to the major components found in the EOs of other aromatic plants such as Salvia cryptantha, 13 Salvia multicaulis, 13 and Thymus species. 12
Such components of EOs have been influential both in plant defense systems and in protection against oxidative stress. 4 The medicinal effects of I. amara are thought to be mainly due to the EO fractions, which contain the highest percentages of oxygenated monoterpenes and monoterpene hydrocarbons. The beneficial oxygenated monoterpenes (carvacrol [9.4%-4.1%], α-terpineol [1.2%-4.8%], and γ-terpineol [1.2%-4.5%]) and monoterpenes hydrocarbons (ρ-cymene, limonene, carene, and α-terpinene) were found in the mentioned groups of EOs of I. amara (Table 1), of which, antibacterial, 1 antiviral, 12 antifungai, 14 and immune system stimulant activities of these compounds 12 is reported previously in other medicinal species.
Eugenol (a hydroxyphenyl propene), an antimicrobial (antifungal and antibacterial compound), 15 was found as a major compound (3.8 ± 0.9%) in the EO of the leaf (Table 1). Notably, oleamide (4.3% in the leaf to 0.2% in the bud) has been considered a potential treatment for mood and sleep disorders, as well as cannabinoid-regulated depression by other researches. 16 Moreover, streamide (3.6% in leaf to 0.9% in bud) can be used in process industries as a release agent or an antistatic agent. As an interesting finding, cuminaldehyde (8.9%-10.4 %) was found in the EOs of the leaf and bud samples (Table 1). This natural aldehyde in Cuminum cyminum is reported to be potential therapeutic for Parkinson’s and dementia diseases. 17 The presence of trans methyl cinnamate, (4.1%) as the second main component in the bud sample, suggests a “methyl cinnamate” subtype for this sample of I. amara that could be a good source for the cosmetic and perfume industries.
Six different volatile compounds were identified in the leaves of I. amara by HS-GC-MS (Table 2) in which the highest (50.1%) and lowest (4.1%) amounts being related to butene, 4-isothiocyanate and (E)-2-butenal, respectively (Table 2). Glugosinolates are the key components in the Brassicaceae family which play an important role as feeding inhibitors of flowering plants in this family such as I. amara. 17 The content of isothiocyanates, hydrolytic compounds produced by degrading glucosinolates, depends on the source organ, the developmental stage of the plant and the effect of exogenous factors. 18 Isothiocyanates, could also lower the risk of different cancers, especially gastrointestinal. 19 Thus, it may be concluded that Iranian accession of I. amara may have potential as an anticancer agent.
Volatile Compounds (%) of EOs of I. amara Obtained by Leaf Sample Through Head Space GC-MS Analysis.

RI, retention index; RT, retention time.
Total Phenolics, Flavonoids, and Flavonols Content
The results of the analysis of the variance indicated that there was significantly difference between two samples (leaf and bud) for all the biochemical traits. This result implied at the effect of extraction source on the content of different secondary metabolites in plants. 10 The content of phenolics, as the strong free radical scavengers, in plants depends on different factors such as plant species, 11 extraction source, 20 environmental factors, 11 extracting solvents, 21 ecological and storage conditions of a plant and/or interactions between an extraction agent (eg, methanol), and polyphenolic compounds. 11 The total phenolic contents (TPC) were 32.9 ± 0.7 (mg/g GAE/g DW) and 28.3 ± 1.7 (mg/g GAE/g DW) in leaf and bud explants, respectively (Table 3). This finding was similar to that reported by Cartea et al, 20 demonstrating high amounts of phenolic constituents in Brassicaceae family leaves.
Contents of Total Phenolic Compounds, TFD, TFL, Carotenoids, Total Chlorophyll and Anthocyanin in Different Samples of I. amara.

Ant, anthocyanin; ChlT, total chlorophyll; Car, carotenoids; TFD, total flavonoids; TFL, total flavonols; TPC, total phenolics content.
Means for each trait with the same letter are not significantly different from each other at P > 0.05 .
The TPC in I. amara was higher than that found in several species of the same family cited in the literature, including broccoli (23.6 mg/g GAE/g DW) and white cabbage (18.4 mg/g GAE/g DW), 22 and lower than that in other species such as York cabbage (33.5 mg/g GAE/g DW, 22 water cress (262.1 mg/g GAE/g FW), wild rocket (139.4 mg/g GAE/g FW), and salad rocket (132.3 mg/g GAE/g FW). 23 However, the biochemical pathways and mechanisms which are responsible for the presence of phenolic compounds in I. amara L. have not been identified.
Flavonoids, as key components of the antioxidant system, 24 belong to the polyphenolic compounds which have a wide range of functions in plant systems such as acting as chemical messengers and defense factors against UV-B radiation. 24,25 The TFD content varied from 28.4 ± 0.7 (QE/g DW) in the leaves to 22.9 ± 2.3 (QE/g DW) in the buds (Table 3). The most diverse group of polyphenols in Brassicaceae species consists of flavonoids (mainly flavonols and anthocyanins), 20,26,27 the variation of which is dependent on different extraction methods, plant tissue and inter cultivar variability. 27 Wegener and Wagner 8 reported that flavonoids (such as kaempferol) are the main constituents of secondary metabolites in I. amara.
Flavonols are the most important flavonoids participating in plant stress responses. 28 As shown in Table 3, TFL varied from 11.86 ± 0.6 (mg QE/g FW) in the leaves to 9.1 ± 0.9 (mg QE/g FW) in the buds (Table 3). The following order was observed for phenolics as TPC >TFD > TFL in both extracts of I. amara (Table 3).
Anthocyanins Content
Anthocyanins as a subgroup of flavonoids contain antiviral, antibacterial, anti-cancer and anti-inflammatory agents, and improve visual acuity properties. 29,30 Brassicaceae also synthesize anthocyanins, which are especially abundant in pigmented brassica crops. 26 The anthocyanin content ranged from 0.43 ± 0.02 µg/g DW in the leaf samples to 0.28 ± 0.03 µg/g DW in the buds (Table 3). This value demonstrated the potential health promoting properties of I. amara leaves.
Photosynthetic Pigments
Photosynthetic potential of each plant is directly determined by the chlorophyll content. 31 The highest (2.5 ± 0.1 mg/g DW) and the lowest (1.4 ± 0.05 mg/g DW) values for total chlorophyll were observed in leaf and bud samples, respectively (Table 3).
Carotenoids play a major role in protecting plant cells against photooxidative processes by scavenging ROS. 1,30 Chemical investigation of the Brassicaceae family has also revealed the presence of phytonutrients such as carotenoids as the most relevant types. 27 Carotenoid content showed no significant difference between leaf (0.21 ± 0.05 mg/g DW) and bud (0.1 ± 0.04 mg/g DW) extract sources (Table 3).
Antioxidant Assay
The TAC of plant extracts cannot be measured accurately by any single method due to the complex nature of phytochemicals and involvement of multiple reaction characteristics. 30 For example, the antioxidant activity evaluated by different methods as ABTS ([2,2'-azinobis-(3-ethylbenzothiazoline-6-sulfonate)]), DPPH and FRAP (Ferric Reducing Anti-oxidant Power in 4 different species of the Brassicaceae family was highly correlated with the content of polyphenols. 23 Thus, in this experiment, the antioxidant activity of I. amara was evaluated by 3 different methods.
DPPH Assay
DPPH free radical scavenging is generally accepted as a main mechanism against lipid oxidation. 30,32 In the present study, the free radical scavenging potentials of the extracts at 6 different concentrations (6.25, 25, 50, 100, 200, 400, 800, and 1000 µg/mL) were tested to obtain IC50 values. According to this figure, the IC50 values of the bud extracts were higher than those of leaf extracts, demonstrating the higher antioxidant activity of the leaf samples (data not shown). Considering the fact that lower IC50 values indicated higher antioxidant activity, radical scavenging activity showed an increasing trend from bud to leaf samples, ranging from 458.6 ± 5.6 µg/mL in buds to 415.13 ± 3.16 µg/mL in the leaves (Table 4). The findings showed weak antioxidant activities in methanolic extracts compared to the antioxidant ascorbic acid with an IC50 = 152 µg/mL. In line with the present findings, Loganayaki et al. 33 reported lower antioxidant activity in the methanolic extract of Helicteres isora L. (IC50 = 428.1 µg/mL) compared to ascorbic acid as the control (IC50 = 30 µg/mL). The DPPH radical scavenging activity is known to be related to the difference in phenolic compounds and the extraction solvent used. 21,28 For example, ethanol and acetone extracts showed the highest scavenging activities in the Teucrium polium 25 and Piper methysticum 21 extracts, respectively. The nature of phenolic compounds such as α-pinene and p-cymene play a significant role in electron transfer/hydrogen donating ability, 34 of which p-cymene was in the EOs of I. amara.
Evaluation of Different Antioxidative Assay Models in Different Samples of I. amara.

β car, β-carotene/linoleic acid; DPPH, 2,2-diphenyl-1-picrylhydrazyl; PM, phosphomolibdate assay.
Means for each trait with the same letter are not significantly different from each other at P > 0.05 .
β-Carotene/Linoleic Acid
The free radical linoleic acid attacks the highly unsaturated β-carotene, and the presence of different antioxidants can reduce the extent of β-carotene bleaching by neutralizing the linoleate free radical and other free radicals formed in the system. 32 The antioxidant activity with β-carotene assay varied from (IC50 = 1676.56 ± 6.35 µg/mL) in the leaves to (IC50 = 1264.33 ± 105.63 µg/mL) in the buds, and was lower than that of synthetic antioxidant butylated hydroxy toluene (BHT) (IC50 = 1057.73 µg/mL) (Table 4). These results imply that different explant sources of I. amara showed lower antioxidant activity than BHT.
Phosphomolybdenum Assay
The evaluation of the TAC, as a novel technique, may be an appropriate and complementary method for determining the additive antioxidant properties of plant foods. 30,35 The antioxidant capacity of I. amara showed variation from 628.3 ± 10.1 µg AAE/g FW in the leaf to 475.5 ± 10.9 µg AAE/g FW in the bud (Table 4). In addition, the different antioxidant activities obtained by these different assays suggested that various extracts might act via different mechanisms, including prevention of radical chain initiation, decomposition of peroxides and prevention of hydrogen abstraction. However, endogenous parameters such as the genotype or environmental conditions may also affect the antioxidant potential of Brassicaceae families27 including I. amara.
Correlation of Bioactive Compounds
Correlation coefficients between different studied traits were calculated (Table 5). As expected, the positive correlation (r = 0.9**) was obtained between TPC and TFL, which might be attributed to their similar biosynthetic pathways. Total phenolic content showed positive correlations with two model systems (PM and β-carotene) while high negative correlation was gained for the DPPH model system (Table 5). Phenolics, due to their chemical capacity in transfer of hydrogen atoms to free radicals, were introduced as the major factor contributing to the antioxidant activity of the species. 36 There was a negative and significant correlation between DPPH assay with different phenolic components including TPC (−0.8**), TFD (0.7**), and TFL (−0.8**) (Table 5). So, this negative correlation could be described by the important role of antioxidant activity of phenolics compounds including TPC, TFD, and TFL.
Correlation Coefficients Between Bioactive Components on Studied I. amara Accession.
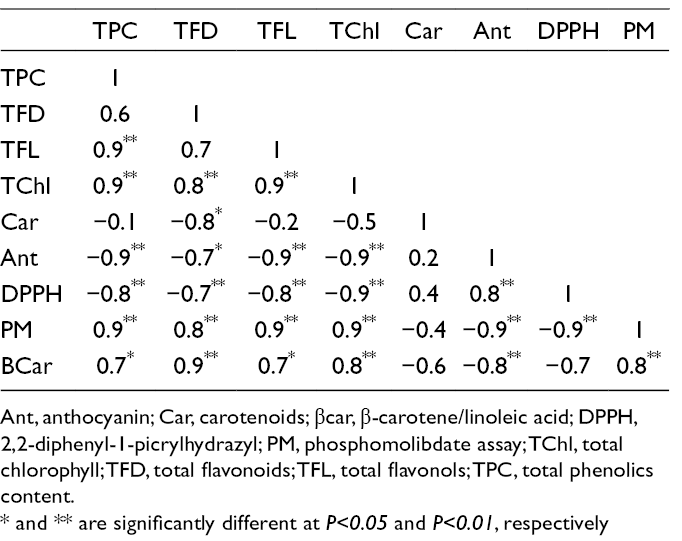
Ant, anthocyanin; Car, carotenoids; βcar, β-carotene/linoleic acid; DPPH, 2,2-diphenyl-1-picrylhydrazyl; PM, phosphomolibdate assay; TChl, total chlorophyll; TFD, total flavonoids; TFL, total flavonols; TPC, total phenolics content.
* and ** are significantly different at P<0.05 and P<0.01, respectively
Moreover, EOs consisting of phenolic monoterpenes and/or sesquiterpenes has been recognized for their higher antioxidativecapacity. 12 Based on these observations, the antioxidant capacity of the I. amara L. extract recorded in the present study might be due to its high amounts of phenolic constituents such as carvacrol and ρ-cymene.
Conclusion
The identification of bioactive components in the EOs and evaluation of antioxidant activity in I. amara L. from Iran is reported. The findings of the present study strongly suggest that I. amara L. contains a valuable source of bioactive compounds with pharmaceutical activity which could have physiological actions improving human nutrition as food. On the other hand, the antioxidant activity of the leaf explants of I. amara can potentially increase the benefits of the plant as a supplementary food due to its natural antioxidant properties. However, further investigations are required to evaluate the safety of the flavonoid compounds of the leaf extract and the effect of different extraction methods on EOs variation in I. amara. It seems that it is crucial to evaluate the genetic diversity of this species for different total phenolic compounds and their antioxidant activity in different geographical regions of the world.
Experimental
The plant materials (leaves and terminal buds) of I. amara were collected in September of 2016 from the Research Field of Goldaru Pharmaceutical Com, located at Falavarjan (32.46 88° N, 51.55 77° E, 1600 m altitude), southwest of Isfahan, Iran. The samples were identified by Prof Fariborz Moattar and the Voucher specimens (GLIR-IA.01/2016) deposited in Goldaru-com, Pharmaceutical Laboratory, Iran.
Gas Chromatography Mass Spectrophotometry and Head Space GC-MS Analysis
Dried leaves and terminal buds (15-20 g) of each sample were subjected to hydro-distillation for 5 hours using a Clevenger-type apparatus to extract the EOs. The EOs were kept in dark vials at 4°C prior to analysis. The yield of EOs was calculated according to the following formula: EOs (%): Content of EO (g)/Content of dry matter (g) (%). 11 Identification of bioactive compounds of I. amara L. was performed on an Agilent 7890A/MS-5975C model (Agilent Technologies, Santa Clara, CA, USA). Agilent Technologies GC systems are equipped with HP-5MS column (30 m in length × 0.25 mm in diameter, film thickness 0.25 µm). Helium gas was employed as the carrier gas with a flow rate of 1 mL/min. The oven temperature was set at 50°C for 3 minutes initially. Then, it was increased to 289°C at 5°C/minute. Finally, the temperature was held at 280°C for 15 minutes. One µL of the prepared 1% (1/100, v/v in methanol) of the extracts was injected in a splitless mode. Relative quantities of the identified chemical compounds in the extracts of I. Amara are expressed as percentages. Identification of chemical constituents was based on comparing their retention indices to n-alkanes (C9–C25) and their mass spectra with those recorded in the National institute of standard and technology (NIST) 37 and those reported in the literature. 38
Plant Extraction Procedure
For achievement of leaf and bud extract, 10 mL of 80% methanol was added to 0.25 g of the dried-milled leaf and bud accessions and shaken slowly. The solution thus obtained was filtered through a 0.45-µm size pore into clean vials and stored at 4°C in the dark for further experiments. 32
Total Phenolic Content
Total phenolic content was calculated using the Folin-Ciocalteau method. 32 In brief, 500 µL aliquot of methanolic extract was mixed with 2.5 mL of 0.2 N Folin-Ciocalteau reagent. The mixture was kept for 10 minutes, followed by the addition of 2.0 mL of 15% sodium carbonate solution. After 2 hours of incubation at room temperature, the mixture was centrifuged for 20 minutes (4500 rpm). The absorbance of the supernatant was measured at 760 nm by a spectrophotometer. Total phenolic content was then quantified by a calibration curve method using gallic acid as standard. The concentration of total phenolic compounds present in the leaf and bud extract is shown as milligrams of gallic acid equivalent per mg of extract dry weight.
Total Flavonoids Content
Leaf extract (0.5 mL), ethanol 95% (2.5 mL), aluminum chloride 10% (0.2 mL), 1 M potassium acetate (0.2 mL), and distilled water (5.6 mL) were mixture until total volume up to 9 mL. This mixture was incubated at room temperature (30 minutes) and spectrophotometer was applied in order to measure this reaction mixture absorbance at 415 nm. Then, 0.2 mL of distilled water was substituted for 0.2 mL AlCl3 10% in blank. By employing a standard curve developed from quercetin equivalence (QE), each sample final absorbance was compared. The content of TFD was stated in terms of quercetin equivalents milligram per gram of dried extract (mg QE/g DW).
Total Flavonols Content
Total flavonols content was estimated, following what proposed by Akkol et al 25 with minor modifications. A mixture was prepared for TFL assay containing methanolic extract (1 mL), aluminum chloride solution (1 mL, 2% v/v), and sodium acetate (3 mL). Then, the absorbance was measured at 445 nm. The standard curve of quercetin was used for calibration. Total flavonol values are shown as mg quercetin equivalence (QE) per gram of fresh extract.
Anthocyanin Assay
Hundred mg of frozen tissues were soaked in 2 mL of 0.1 N hydrogen chloride at once. A glass pestle was utilized to crush these tissues. Then, the tissues were kept at room temperature for 3 hours. The centrifuging process was performed for 10 000 g of the extract at room temperature for 5 minutes. Then, a spectrophotometer was used for analyzing absorption at 511 nm of the supernatant. Finally, Raphanusins coefficient of molar extinction was employed for calculating anthocyanin content. 29
Photosynthetic Pigments
A UV-Vis Spectrophotometer (Unico- UV 2100) was applied for analytical determination of chlorophyll content (total chlorophyll) and carotenoids according to Gitelson et al. 31
Antioxidant Assay
DPPH Assay
According to what proposed by Salem et al 32 after minor modifications, 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) was employed for determining the activities of I. amara for scavenging free radicals. DPPH was purchased from Sigma-Aldrich (USA). One milliliter of DPPH solution in methanol (50 µM) was mixed with 20 µL of the extract from both the leaf and bud samples. DPPH absorption reduction was measured at 515 nm. As a positive control, ascorbic acid (Sigma, Inc.) was chosen to be employed. All the required measures were determined in triplicate. The regression equation containing extract concentrations and inhibition percentages was used for calculating extract concentrations providing 50% inhibition (µg/mL). The IP50 value is defined as plant extract concentration required for scavenging half of the total DPPH radicals (50%).
β-Carotene/Linoleic Assay
The β-carotene/linoleic assay was carried out according to Salem et al 32 with minor modifications. To begin, 2 mg of β-carotene (Sigma, Inc.) was dissolved in chloroform (1.0 mL). Then, a mixture was made from combining linoleic acid (Sigma, 200 µL) and Tween-80 (0.2 mL). This mixture was allowed to stay at room temperature for 15 minutes. Subsequently, oxygenated distilled water (50 mL) was added to this mixture after chloroform evaporation, and the mixture was shaken until linoleic-β-carotene emulsion was produced. Test tubes containing different concentrations of the extract (0.2 mL from each) were employed to test this emulsion. The linoleic-β-carotene emulsion (3 mL) was transferred into these tubes. These tubes were shaken and incubated in a water bath at 50°C. The zero time absorbance (A˳) at 470 nm was measured by a spectrophotometer immediately after adding the emulsion to each tube. Another absorbance (A1) was calculated after 120 minutes. As the background subtraction, a blank, without β-carotene, was employed. The following equation was utilized to calculate lipid peroxidation inhibition (%): LPO = 1 − A1/A0 × 100. Finally, the graph indicating antioxidant activity percentage against extract concentration was used in order to calculate the concentration that provides 50% antioxidant activity (IP50). The standard was set to be BHT as a synthetic antioxidant (R 2 = 0.96).
Phosphomolybdenum Assay
Total antioxidant activity of the leaf extract was determined by the phosphomolybdenum assay. 35 First, the reagent solution composed of sulfuric acid (0.6 M), sodium phosphate (28 mM), and ammonium molybdate (4 mM) was prepared. Then, 0.1 mL of the extract was mixed with 3 mL of reagent solution. The tubes were capped and incubated in a water bath at 95°C for 90 minutes, then, the samples were cooled until room temperature. Subsequently, the solution absorbance was calculated at 695 nm against blank. Methanol (0.1 mL) was replaced for the extract as the blank. The following formula was used to measure antioxidant capacity: Antioxidant effect (%): (control absorbance − sample absorbance)/control absorbance (%). The values were expressed in terms of ascorbic acid equivalent per mg of extract.
Statistical Analysis
Data were subjected to analysis of variance by SAS software version 9.3. 39 The experiment was carried out as completely randomized design with three replications for studied traits. The means ± SD (standard deviation) were calculated for each trait. The correlation coefficients between the bioactive components were calculated using the proc CORR of SAS software. 39 The means were compared with least significant difference test at 5% of probability.
Footnotes
Acknowledgments
The authors would like to thank Research Institute for Biotechnology and Bioengineering, Isfahan University of Technology, Isfahan, Iran.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Goldaru, Pharmaceutical Institute, Iran.
