Abstract
This review documents all the new homoisoflavonoids (HIFs) that have been reported since 2007, whose total number has grown from 159 in 2007 to 295 at the present time. This review contains their structures, biological sources, plant parts they are obtained from, and, if reported, their optical rotations and melting points. The same classification is followed as an earlier review to ease reference to both reviews. This review takes note of the recent revision of plant families that were known to contain HIFs that have now been merged into one big family, Asparagaceae. Homoisoflavonoids also occur in Fabaceae and others. Two taxa, Ophiopogoan japonicus (Asparagaceae) and Caesalpinia sappan (Fabaceae), have been the source of many HIFs. These are briefly summarized. The biological properties of HIFs are also reviewed under the topics such as antioxidant, anti-inflammatory, antimicrobial, antidiabetic, and cytotoxic. The review also surveys the total synthesis of natural HIFs. All new compounds are classified and tabulated following the same style as the previous review.
Dedicated to Professor Andrew Paul Krapcho on the occasion of his 87th Birthday.
Homoisoflavonoids represent a subclass of the larger family of flavonoids (

General scaffolds of a flavonoid and an HIF.
This review is a follow-up of an earlier one in 2007 3 listing the 159 compounds which were known then. The total number of naturally occurring HIFs has now reached 295 and this review documents the new metabolites belonging to this class that have been discovered since then. This review is based on a survey of publications that have appeared in Scopus and the Science Citation Index during the period 2007 to 2018.
Five important review publications relevant to the field of HIFs have appeared during this review period. The first is a review of the chemical structures, plant origins, ethnobotany, and biological activities of homoisoflavanones,
4
listing 129 compounds. This number is less than the 159 recorded in the earlier review, because the authors focused only on metabolites having a selected number of structural types under this class. The second is a review of HIFs and their pharmacological activities.
5
This lists 240 naturally occurring HIFs and contains a comprehensive listing of all natural HIFs known at the time of the review (2014). The coverage is limited to recording all the metabolites and discusses pharmacological properties. The third review is devoted to one homoisoflavonoid, namely, brazilin.
6
Brazilin (

Examples of HIFs.
Distribution of HIFs
Many of the plant families that were known to produce HIFs, such as Anthericaceae, Liliaceae, Convallariaceae, Dracaenaceae, and Hyacinthaceae, have now been grouped under one big family, Asparagaceae (Accessed September 13, 2018 [http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:481889-1]). So, all references to these families are recorded as Asparagaceae in this review. There are now 7 other families besides Asparagaceae that are known to produce HIFs: Amarillidaceae, Araliaceae, Fabaceae, Orchidaceae, Polygonaceae, Portolucaceae, Rosaceae, Meliaceae, Polypodiaceae, and Similacaceae. By far the majority of HIFs is derived from the family Asparagaceae, a much lesser number from Fabaceae, and 1 or 2 genera belonging to the other families. Many names of species have also changed. For example, C. sappan L. is considered as the synonym of Biancaea sappan (L.) Tod. and taxonomic information for this taxon from Kew’s
Advances in Extraction and Separation Methods
One of the major challenges in the study of secondary metabolites has always been their purification and isolation from complex plant extracts.
The usual methods involve silica gel chromatography, which inevitably results in the irreversible adsorption of some of the components and in some cases even a change in the integrity of the metabolites. In this regard, there have been some advances reported during the period under review of gentler methods of isolation of HIFs. Xu et al have reported the successful separation of 4 HIFs from C. sappan by high-speed counter-current chromatography (HSCCC).
12
The crude extract of C. sappan was fractionated by HSCCC using a 2-phase solvent system consisting of chloroform-methanol-water (4:3:2, v/v/v). The separation conditions were: flow rate, 1.0 mL/min; revolution speed, 900 rpm; detection wavelength, 280 nm; separation temperature, 25°C; sample size, 120 mg crude sample dissolved in a mixture of the upper and lower phases (10 mL each). The retention of the stationary phase was 83%. This method delivered 5 mg of 3′-deoxysappanol (
In another instance Ma et al
13
reported a method that combines supercritical fluid extraction and HSCCC to extract and purify HIFs from O. japonicus. Thus, in a single operation, 140 mg crude extracts were separated and yielded 15.3 mg of methylophiopogonanone A (
Uddin et al have reported a centrifugal partition chromatography (CPC)-based one-step isolation of sappanol (
Theoretical Studies on HIFs
As this class of compounds are more explored in terms of their structural diversity and biological properties, it becomes important to obtain a deeper insight into their properties through theoretical studies. We have observed such studies during the last decade that address the electronic distribution of HIFs. Discrete Fourier Transform (DFT) calculations of nuclear magnetic shielding for optimized geometries of HIFs show good correlations with experimentally measured values. 15
More effective chiroptical methods are now available16,17 to solve absolute configuration issues. These involve combinations of electronic and vibrational chiroptical spectroscopic methods and interpretations of measured spectra with the aid of density functional theory calculations.
These methods
18
were employed in determining the absolute configuration of 5,7-dihydroxy-6-methoxy-3-(9-hydroxy-phenylmethyl)-chroman-4-one (
Ophiopogon japonicus and C. sappan
Ophiopogon japonicus (Thunb.) Ker Gawl. and C. sappan (now called Biancaea sappan (L.) Tod. have been the subject of intense investigations during the last 10 years. Ophiopogon japonicus is an important medicinal plant which is native to Central and South China to Vietnam, temperate East Asia to the Philippines and has been introduced to some areas of central South America (Argentina, Uruguay, and Paraguay). Besides its use in the treatment of a variety of ailments, it is considered as a functional food in China and other East Asian countries. The main active ingredients are believed to be steroidal compounds and HIFs.
7
The Chen review
7
of this plant records 36 HIFs reported from O. japonicus. Further metabolites have been reported since then. Thus, 18 new HIF metabolites have been reported from this taxon since our last review, homoisopogon B-D (
For C. sappan, the native range is the Indian subcontinent to Indo-China. It has been introduced to Nigeria, the Congo, Mozambique, Tanzania, and Uganda in Africa (Accessed September 13, 2018 [http://www.plantsoftheworldonline.org/taxon/urn:lsid:ipni.org:names:481889-1]). The heart wood of C. sappan is the most used part and studies have shown that the main homoisoflavonoid ingredient of the heartwood of this plant is brazilin (
Novel Structures Containing the HIF Skeleton
Novel structures that have been reported during this period will be summarized hereunder. In the earlier review
3
a few HIFs with attachment of sugar units were reported. A 5-O-glucoside, 5-O-rutinoside, and a 5-O-neohesperoside were listed, which were linked to 3-hydroxy-3-benzylchroman-4-ones. These were isolated from Ornithogalum caudatum. During the current review period a few more glycosides have been reported. Simple 7-O-glucosides (
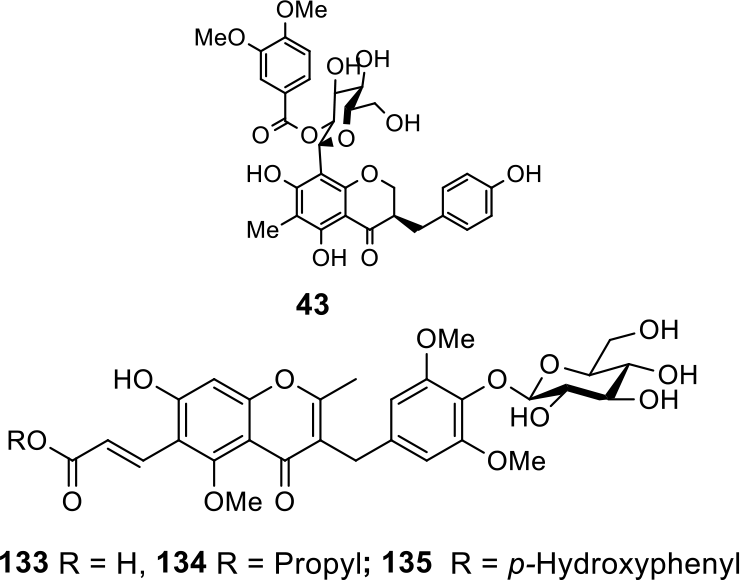
Glycosylated HIFs.
Furthermore, a biogenetically intriguing set of 3 homoisoflavonoid glycosides (
Although prenylated and geranylated flavonoids are very common in certain families of plants, compounds

Examples of unusual HIFs.
Zhao et al
37
reported the isolation of the 3-benzylchromen
In 2014 Wang and coworkers isolated caesappin A (
Classification of HIFs
This report is prepared by following the classification of HIFs proposed in the 2007 review 3 and carefully building on it to update the total number of HIFs that is known from natural sources to date. This will enable readers to consult the 2 reviews and get a full and broader coverage of the subject. Therefore, the HIFs are classified into 5 major groups A to E as restated below and depicted in Figure 5.

The five groups (A-E) of homoisoflavoids.
The first group, A (Table 1), is based on the 3-benzylflavan skeleton, with a new subclass containing Δ3,4-unsaturation and 3,4-dioxygenation.
Group A: 3-Benzylflavan (

aA, Araliaceae; F, Fabaceae; Po, Polygonaceae; Pt, Portulacaceae; M, Meliaceae; PP, Polypodiaceae.
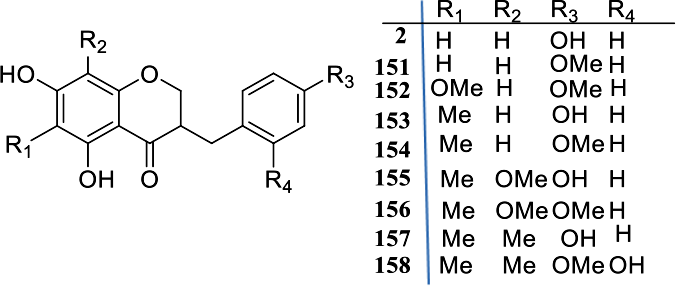
The second group, B (Table 2), consists of the 3-benzyl-chroman-4-ones. The majority of the recently discovered HIFs belongs to this class. Also included in this section are the 3-hydroxy derivatives (Table 3), which are fewer, with just 2 compounds
Group B-1: 3-Benzylchroman-4-Ones: Compounds

aA, Asparagaceae; Ar, Araliaceae; F, Fabaceae; Po, Polygonaceae; Pt, Portulacaceae.
bSubstitutent at C-8 is a 2″-veratroyl-substituted-C-glucoside.
cO-[α-rhamnopyranosyl-(1→6)-β-glucopyranosyl.
dGan Luo Xin is a traditional Chinese medicine for the treatment of hepatitis B formulated using 20 herbs including Polygonatum sibiricum.
eThis compound was isolated as 84.9% 3R and 15.1%3S.
Group B-2: 3-Hydroxy-3-Benzylchroman-4-Ones and 9-Hydroxy-3-Benzylchroman-4-Ones: Compounds
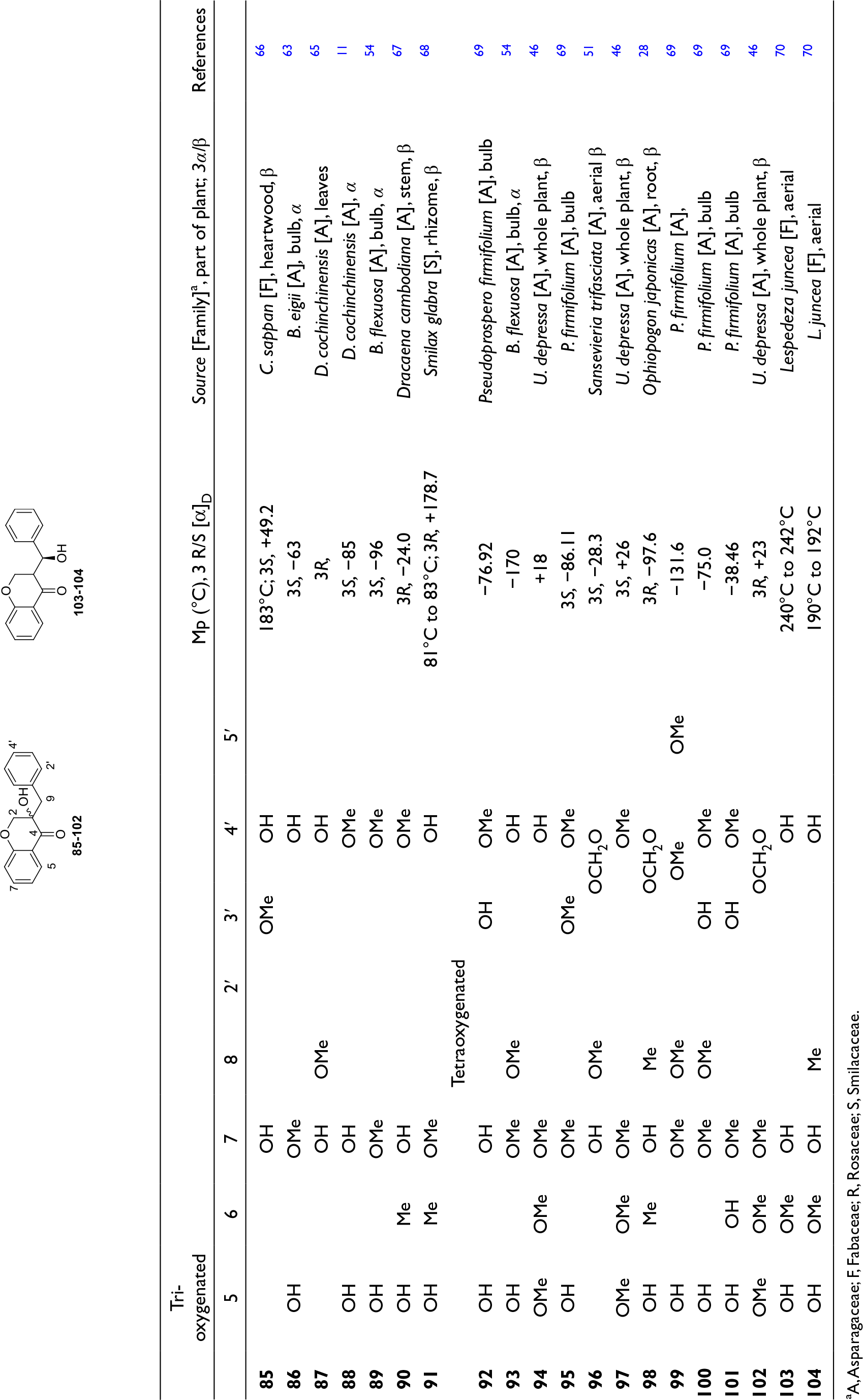
aA, Asparagaceae; F, Fabaceae; R, Rosaceae; S, Smilacaceae.
Group C compounds have the Δ3,9 with either E or Z stereochemistry at the Δ3,9-double bond and derivatives (Table 4). Many HIFs in this subgroup belong to the E series. The observation made in the previous review where the Δ2,33 unsaturated HIFs were largely restricted to the genus Ophiopogon is repeated once again.24-26,28,35
Group C: Δ3,9-Z (

aA, Asparagaceae; F, Fabaceae; Po, Polygonaceae; R, Rosaceae.
Group D is the scillascillins (Table 5). Only 6 HIFs in this class (
Group D: Scillascillins: Compounds

aA = Asparagaceae.
Group E contains the rearranged polycyclic HIFs (Figure 6). Ten HIFs (

Group E: Rearranged HIFs: Compounds
Biological Activities of HIFs
Pharmacological Properties of Brazilin
A recent review 6 has dealt with the pharmacological activities of the homoisoflavonoid brazilin. This review states: Brazilin is the safe natural compound having potential to develop as a medicinal compound with application in food, beverage, cosmetics and pharmaceutical industries to screen its clinical use in modern medicine but cautions that more studies are needed to evaluate the potential application of brazilin as preservative and coloring agent in food processing industries. Brazilin is reported to have a high antioxidant property (IC508.8 µM) comparable with or better than (+)-catechin (10.2 µM). It has also been found to be effective against drug-resistant Gram-positive bacteria including MRSA, vancomycin-resistant enterococci, and multi-drug-resistant Burkholderia cepacian. Brazilin showed antibacterial activity with a Minimum inhibitory concentration of 0.50 mg/mL. The 50% inhibitory concentration (IC50) for lipase inhibition was lowest for brazilin (6 µM), which showed strong inhibition compared with chloramphenicol (677 µM, positive control). 80 Experimental evidence is outlined in the Nirmal review supporting the beneficial hypoglycemic role of brazilin by enhancing glucose metabolism in adipose tissue, by inhibiting protein kinase C and insulin receptors serine kinase, and by inducing glucose transport in isolated rat epididymal adipocytes, including details on its mechanism of action. 6 The vasorelaxing property of brazilin has also been documented. Brazilin relaxed phenylephirine-induced vasoconstriction and increased cGMP in isolated rat aorta. More recently, brazilin is reported to increase the life span of the nematode worm Caenorhabditis elegans. The mean life of 12.3 ± 0.4 days was increased to 14.5 ± 0.4 days when the worms were fed 100 µM brazilin. 81
Antioxidant and Anti-Inflammatory Properties
The antioxidant and anti-inflammatory properties of several HIFs have been reported using the simple 2,2-diphenyl-1-picrylhydrazyl (DPPH) method, the inhibition of nitric oxide (NO) or the suppression of inducible nitric oxide (iNOS) synthase, inhibiting prostaglandin (PGE2), interleukin (IL)−1b production, etc. Ledebouria floribunda has been identified as a source of HIFs, some with prenyl and geranyl groups and others with glycosidic linkages.33,36 The 3 prenylated and geranylated derivatives (
Antioxidant Data of HIFs Using DPPH Radical Scavenging (System A), and B-Carotene/Linoleic Acid (System B) Assay Systems.
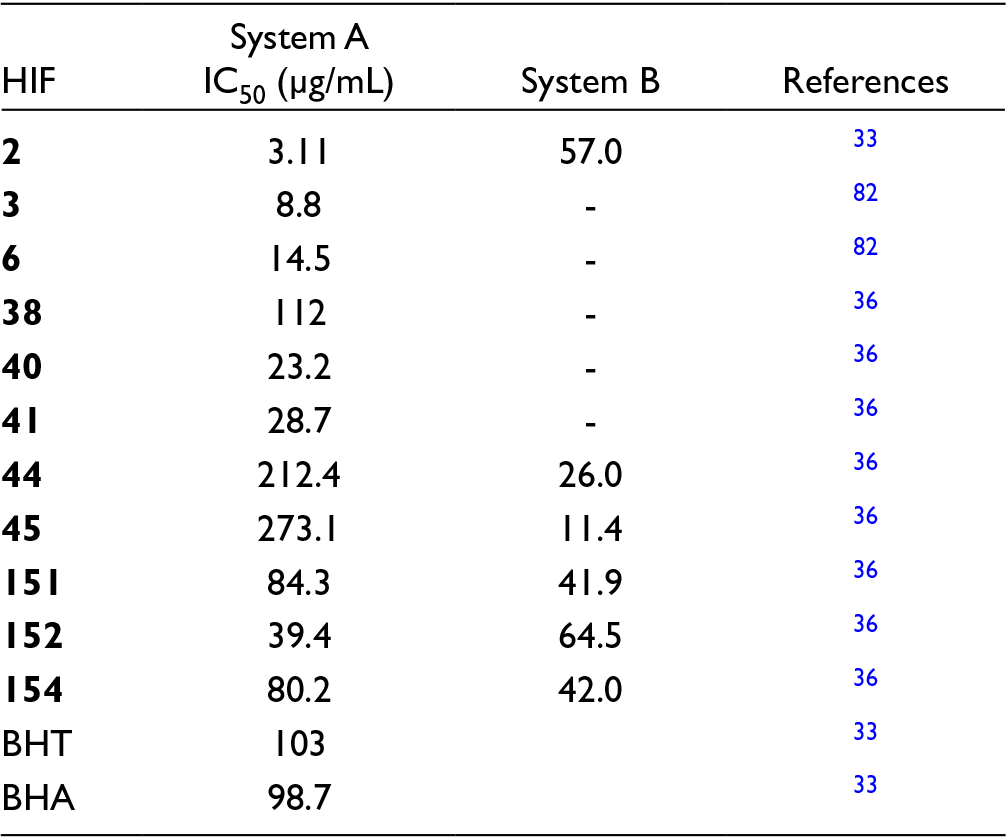
Calvo et al 36 also reported HIFs with sugar groups attached to the C-7 phenolic group of ring A. Their antioxidant properties have been assessed using both the DPPH scavenging as well as the β-carotene/linoleic acid system. The results showing strong antioxidant properties are shown in Table 6.
Hung et al
26
investigated the effect of HIFs on the release of the inflammatory chemokine eotaxin, stimulated by IL-4 and in combination with TNF-α in BEAS-2B cells, which mimics the in vivo conditions in bronchial allergic asthma. They investigated the ability of HIF ophiopogoside A (
The anti-inflammatory properties of sappanone A (
Antibacterial, Antiviral, and Antifungal Activities
An interesting study by Jeong et al
45
presents the antiviral activities of closely related HIFs assessed against neuraminidases (NAs) on the surface of influenza viruses (A/PR/8/34 [H1N1], A/Hong Kong/8/68 [H3N2], and A/Chicken/Korea/MS96/96 [H9N2]) and confirmed by the positive control with oseltamivir (IC50 = 5.8 nM [H1N1], 5.6 nM [H3N2], and 1.2 nM [H9N2]). The HIFs included in these studies were brazilin (
Example of antiviral HIF.
Antihyperglycemic Properties
The hypoglycemic properties of HIFs have been assessed in terms of their ability to modulate the insulin receptor, lower blood glucose, and regulate the levels of various kinases (protein kinase C, phosphotidylinositol 3-kinase, pyruvate kinase, and 6-phosphofructo-2 kinase), which are involved in the gluconeogenesis and glycolytic pathways and glucose transport. 60
The 2 protein components of the glucose absorption system of the intestine are the sodium-dependent glucose transporter-1 (SGLT1), and glucose transporter (GLUT). Several HIFs have been evaluated for their ability to either activate adenosine monophosphate-activated protein kinase (AMPK) or to inhibit the activity of the GLUT transmembrane carriers.85,86 Adenosine monophosphate protein kinase activators are potential therapeutic candidates for the treatment of diabetes as they can increase the uptake of glucose via membrane translocation of GLUT 4. Six previously known HIFs from the rhizomes of P. odoratum were evaluated for their ability to activate AMPK, and 3 of them (
Examples of antihyperglycemic HIFs.
In another earlier study
87
2 of the HIFs,
The 3 esters, prunuside A to C, with a rather unusual substitution and carbon framework, were obtained from the shoots of the Rosaceae plant, P. domestica. The source of these prunusides is a tree whose fruits are edible and used to lower blood sugar in India and Pakistan. The HIFs (
Adenosine monophosphate protein kinase activation is known to affect glucose and lipid metabolism, gene expression, and protein synthesis. Two out of 6 HIFs isolated from the rhizomes of P. odoratum, (3R)-5,7-dihydroxyl-6-methyl-8-methoxyl-3-(4ʹ-hydroxybenzyl)-chroman-4-one and (3R)-5,7-dihydroxyl-6,8-dimethyl-3-(4ʹ-hydroxybenzyl)-chroman-4-one were found to have significant activation effects. 86
Cytotoxic Activities
The cytotoxicity of HIFs on various cell lines, including tumor cells has been documented.
Uesawa et al
88
undertook a SUGITA3 quantitative structure-activity relationships of 16 HIFs (3-benzylidinechromanones) and concluded that those 3-benzylidenechromanone derivatives that have a methoxy group at position C-7 of the chromanone ring and either hydroxyl or methoxy group at C-4ʹ of the B ring showed relatively higher tumor-specificity values, exceeding those of doxorubicin and 5-fluorouracil. The paper concluded by pointing out that molecular shape, size and polarization are useful indicators for the evaluation of tumor specificity of this class of compounds. Compound
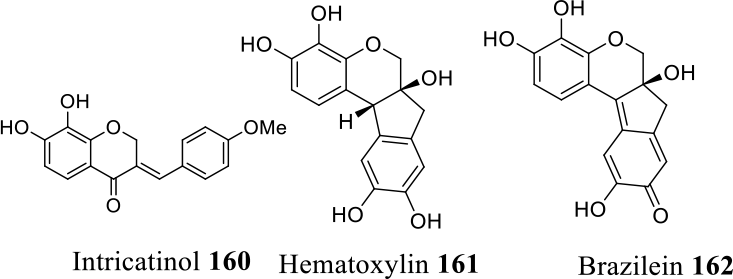
The anticancer potential of intricatinol (
Cytotoxic HIFs.
In studying the roles of protein tyrosine kinase (PTK) inhibitors for cancer chemoprevention, Lin et al90,91,108 found that hematoxylin (
In a recent study Yang et al
93
investigated the antitumor properties of protosappanin B (
Antiproliferative activity guided investigation of the South African plant U. depressa led to the isolation of 6 new HIFs
The finding that 5,7-dihydroxy-3-(4ʹ-hydroxy-3ʹ-methoxybenzyl)-6-methoxyhomoisoflavanone (cremastrone) possesses antiangiogenic activity both in vitro and in vivo and was a potent inhibitor of the proliferation of human umbilical vein endothelial cells,
94
prompted further work to identify other natural HIFs and their derivatives which may be developed as small molecule therapeutic agents.95,96 Among natural HIFs methylophiopogonanone A (
Synthesis of Naturally Occurring HIFs
The commonly employed methodologies for the construction of the homoisoflavonoid scaffolds can be categorized into 2 general groups:
Aldol condensation of pre-prepared chroman-4-ones with aryl aldehydes (Scheme 1a) provides HIFs ( Acid catalyzed cyclization of dihydrochalcones
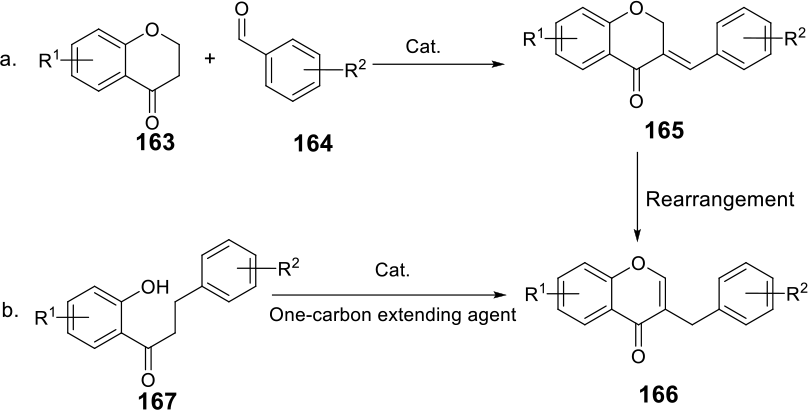
General protocols for the synthesis of HIFs.
Since our last review, few novel synthetic methodologies have been developed for the construction of the homoisoflavone scaffolds. The cyclobenzylation reaction of (E)-3-(dimethylamino)-1-(2-hydroxy-4,5-methylenedioxy-phenyl)prop-2-en-1-one

BnBr, NaI, acetone, 80°C, 5 hours, 80%.
In a serendipitous discovery, Lee and co-workers developed an interesting ruthenium-catalyzed synthesis of 2-arylhomo-isoflavonoid
Aldol condensation between the flavonoid
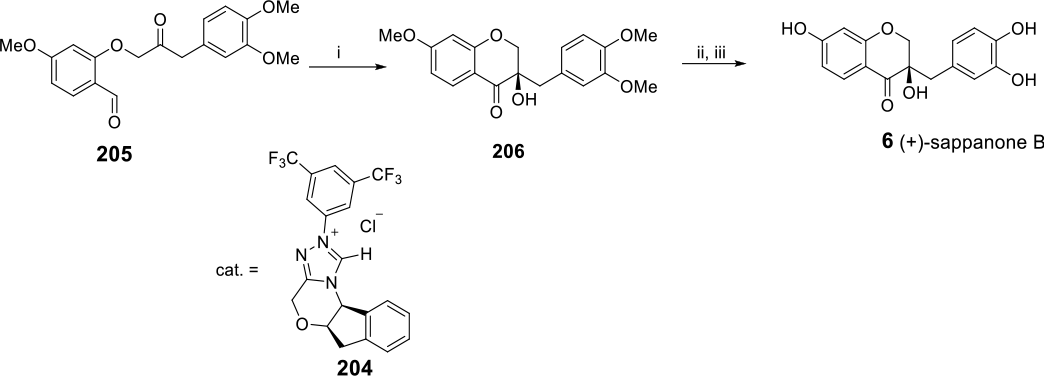
The major advance made in the synthesis of HIFs post 2007 is the focus on asymmetric synthesis. Yu et al, for instance, reported the asymmetric synthesis of several sappanin-type natural HIFs (

(i) TiCl4, (+)-sparteine, DCM, 0°C, 2 hours, 83%; (ii) Pd/C, EtOAc, rt, 5 hours, 95%; (iii) TiCl4, TMEDA, DCM, −20°C, 2 hours, 62%; (iv) NaBH4, THF/H2O, 5 hours, 86%; (v) p-TsCl, n-Bu2SnO, DMAP, Et3N, MeCN, rt, 3 hours, 84%; (vi) n-Bu4NF, THF, rt, 30 minutes, 92%; (vii) Ac2O, Et3N, DMAP, DCM, rt, 30 minutes, 94%; (viii) Et3SiH, CF3CO2H, DCM, 0°C, 30 minutes, 88%; (ix) K2CO3, MeOH, rt, 30 minutes, 92%; (x) 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ), dioxane, H2O, rt, 30 minutes, 74%; (xi) BBr3, CH2Cl2, rt, 1 hour, 89%; (xii) DDQ, dioxane, H2O, rt, 30 minutes, 57%.
By exploiting the versatility of Sharpless dihydroxylation and epoxidation reactions, a number of sappanone-type natural HIFs have been prepared. This is demonstrated by Kim and co-workers who successfully synthesized (+)-urgineanin A-D and (−)-urgineanin A (
Similarly, by changing the substituents at the phosphorus atom of enol phosphates
In addition to asymmetric dihydroxylation strategies for the synthesis of enantiopure sappanones, an enantioselective cyclization protocol has also been reported. Takikawa and Suzuki designed and synthesized a modified Rovis catalyst
(i) cat. (15 mol %), Et3N, toluene, rt, 12 hours, 92%, 95% ee; (ii) NaSC12H25, DMF, 80°C, 5 hours, 92%; (iii) BBr3, DCM, 0°C, 0.5 hour, 85%.

Biogenetic scheme to caesappin A (
The epoxidation and dihydroxylation reactions have been also instrumental in the total synthesis of (±)-brazilein, (±)-brazilin, and (±)-brazilane.108,109 Zhang and co-workers utilized asymmetric Sharpless dihydroxylation reaction for the enantioselective synthesis of (+)-brazilin (
In conclusion, HIFs are now recognized as an important subclass of flavonoids. They display broad structural diversity and biological properties. They occur principally in 2 plant families, Asparagaceae and Fabaceae, and several genera including Ophiopogon, Caesalpinia, Polygonatum, Ledebouria, Belevalia, Dracaena, Hematoxylon, Liriope, Portulaca, Scilla, Pseudoprospera, and Urginea. Many species belonging to these genera are important ingredients in many Chinese, Indian, Japanese, and other countries’ traditional medicine. HIFs are the major bioactive constituents of the popular medicinal plants O. japonicus,
7
C. sappan,
6
and of Dragon’s blood (Dracanea cinnabari).
8
Ophiopogon japonicus alone has yielded more than 38 HIFs,
7
more than any other species, followed by Caesalpinia, Polygonatum, and Ledebouria species. Homoisoflavonoids have been shown to possess a wide range of pharmacological properties including antimicrobial, antimutagenic, antioxidant, immunomodulatory, cytotoxic, antiangiogenic, anti-inflammatory, antiphotoaging, hypoglycemic, vasorelaxant, hepatoprotective, and antiacne activities. Brazilin (

[Ru(p-cymene)Cl2]2 (2.5 mol %), CsOAc, DMSO, 120°C, 12 hours, 64% to 82%.

Ph2O, Cs2CO3, reflux, 65% to 84%.
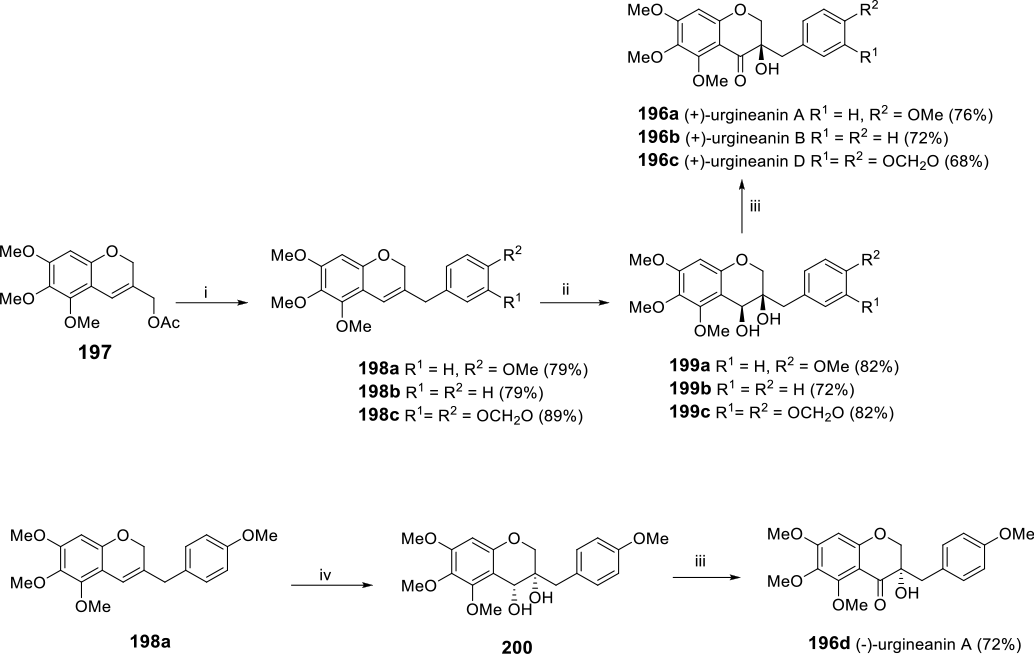
(i) ArB(OH)2, Pd(PPh3)4, K3PO4, THF, 150°C; (ii) OsO4, (DHQ)2PHAL, K3Fe(CN)6, K2CO3, MeSO2NH2, 0°C; (iii) IBX; (iv) OsO4, (DHQD)2PHAL, K3Fe(CN)6, K2CO3, MeSO2NH2, 0°C.
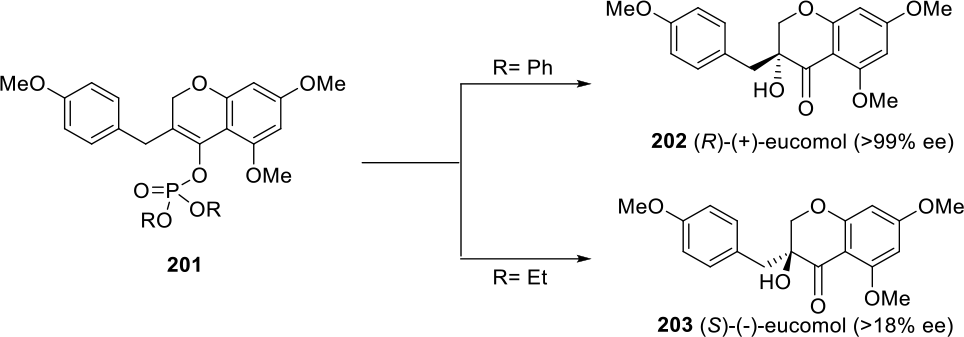
(DHQD)2PHAL, OsO4, CH3SO2NH2, K3Fe(CN)6, K2CO3, t BuOH/H2O, 0°C.

(i) DEAD, Ph3P, THF, −78°C, 60%; (ii) AD-mix-b, OsO4, MeSO2NH2, t -BuOH/H2O/DCM, 3 days, 85%, 81% ee; (iii) PPTS, toluene, 90°C, 80%; (iv) LiOH, THF/MeOH, 67%, 99.8% ee; (v) BBr3, DCM, 81%; (vi) PhI(OAc)2, THF, 0°C, 76%; (vii) m-CPBA, 32°C, 3 days, DCM, 22%; (viii) a. BF3·Et2O, DCM; b. 65% H2SO4, AcOH, 110°C, 82%.
Footnotes
Acknowledgments
This paper was written during B.M.A.’s 1-month stay (September 2018) at the Stellenbosch Institute for Advanced Study (STIAS). B.M.A. is grateful to the leaders of this enabling institution and the staff who have been very gracious and helpful. Research Centre for Synthesis and Catalysis of the Department of Chemistry at UJ and the National Research Foundation (NRF)-DST are also acknowledged for financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was financially supported by the Research Centre for Synthesis and Catalysis of the Department of Chemistry at UJ and the National Research Foundation (NRF)-DST.




