Abstract
For prosperous domestication, breeding, and cultivation of a herbal species, it is important to screen its medicinally valuable compounds as well as its referred biological activity. Salvia L. species (Lamiaceae), distributed throughout the world, contain a wide range of secondary metabolites including terpenoids and phenolic derivatives. Betulinic acid (BA), oleanolic acid (OA), and ursolic acid (UA) are highly valuable triterpenic acids (TAs) because of their wide range of biological activities. The objective of the present work was to evaluate the BA, OA, and UA contents among 22 Salvia species native to Iran. TA content in the studied Salvia species was compared with that in Salvia officinalis as a commercial species. High-performance liquid chromatography with photodiode array detector results showed that the maximum content of BA (3.12 ± 0.03 mg/g dry weight [DW]) and OA (1.96 ± 0.05 mg/g DW) was determined in Salvia multicaulis. The highest content of UA (4.34 ± 0.1 mg/g DW) was quantified in S. officinalis L. followed by S. multicaulis (3.71 ± 0.08 mg/g DW). Salvia multicaulis exhibited significantly higher agro-morphological values than S. officinalis in traits related to plant width, leaf length, internode length, and inflorescence length. The cytotoxicities of both species were determined against human cancer cell lines using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. The methanolic extract of S. multicaulis and S. officinalis showed cytotoxic effects against SH-SY5Y and MCF-7 cell lines, respectively. Both species were equally cytotoxic against the HL-60 cell line. This study provides scope for the selection of high-yielding species and genetic improvement through breeding and biotechnological programs in the future.
Medicinal plants are among the chemical reserves and natural riches of the world’s regions and territories. Many countries are considering their plant species diversity as a natural chemical wealth.1,2 Unfortunately, increasing attention to the marketing of medicinal plants has created problems for these resources and has left many medicinal species at risk, with destruction. According to the available reports, about 8% of the total plant species in the world (over 34 000 species, with their medication being considered) are at risk of erosion and degradation. Therefore, in order to exploit and protect the genetic reserves of medicinal species, this necessity, especially due to the cultivation and industry of medicinal plants, seems to be in favor of domestication and introduction of these species into agricultural systems. Medicinal species are morphologically and phytochemically heterogeneous in different climates and natural habitats.3-5 Therefore, for the principal and industrial use of these plants, it is necessary to evaluate their identity and nature from a variety of genetic, chemical, and manufacturing perspectives.1,6 If a medicinal species is required, due to the economic importance and, in particular, the risk of the destruction of such populations, into imported and domestic agricultural systems, it should be noted that the first and foremost important domestication strategy includes the careful evaluation of the chemical, morphological, and genetic aspects as well as the medicinal properties of species populations. The first step in achieving breeding goals is to be aware of the diversity of wild populations and the selection of superior individuals in terms of growth and phytochemical characteristics. Due to its climate diversity, Iran has a vast and unique biodiversity.
Cancer is a major cause of mortality in the world and occurs as a result of various factors, including chemical mutagens and cancer-causing chemicals. 7 In the past decades, extensive studies have been carried out in order to identify various molecular targets that can potentially be used for both cancer prevention and treatment. Natural products, especially those derived from the plants, have been used in the treatment of various diseases for thousands of years. The plants contain phytochemicals such as vitamins, carotenoids, terpenoids, flavonoids, alkaloids, tannins, saponins, enzymes, and minerals. These compounds have antioxidant activities that can be used to prevent or treat many diseases including cancer. 8 Terpenoids, widely distributed in medicinal plants, fruits, and vegetables, are derived from five carbon isoprene units and are classified into four structural categories, namely mono-, sesqui-, di-, and triterpenoids. Triterpenoids (TTs, C30H48) are organic compounds with diverse structures and to date more than 15 000 types have a range of unique and potentially usable biological properties.9-11 Three well-known triterpenic acids (TAs), namely betulinic acid (3β-hydroxylup-20(29)-en-28-oic acid, BA), oleanolic acid (3β-hydroxyolean-12-en-28-oic acid, OA), and ursolic acid (3β-hydroxyurs-12-en-28-oic acid, UA) (Figure 1), have frequently been reported from different plant organs such as berries, leaves, flowers, and fruits.12-14 Various biological effects of TAs including anti-inflammatory and antioxidant, 15 anti-HIV, 16 anti-fungal, and immunomodulatory activities 17 have been characterized. Recently, potential effects of BA, OA, and UA for the treatment of type 2 diabetes have also been highlighted. 18 Recently, bioassay of TAs against different human cancer cell lines has been extensively described.10,19,20 Based on their considerable anticancer activity and probably the high demand for the drugs, there is an urgent need to find plant-rich sources of TAs for their production. So far, many plant families including Dilleniaceae, 21 Ericaceae, 22 Euphorbiaceae, 23 Rosaceae, 24 and Myrtaceae 25 have been studied for their TA content.

Chemical structures of the most important TAs.
Members of the mint family (Lamiaceae) have been potentially reported as rich sources of free TAs besides other compounds.13,26
Salvia L. is one of the most important genera in Lamiaceae and includes over 900 species that grow worldwide. 27 Salvia species contain a wide range of secondary metabolites including terpenoids and phenolic derivatives. 28 Literature survey revealed that Salvia species have been interestingly considered for the isolation and production of medicinally valuable TAs.29,30-33 Fifty-eight Salvia species have been documented in the flora of Iran, of which 17 are endemic.34,35 Many Salvia species are used as herbal tea and in food flavoring, as well as in the cosmetics, perfumery, and pharmaceutical industries. 36 As far as the literature data could ascertain, there is a significant variation between the different species of Salvia based on their medicinal compounds. Therefore, a comprehensive study to investigate the Iranian germplasm with the aim of identifying and introducing superior species in terms of TA content and other desirable morphological traits for breeding, domestication, and production in agronomical systems is needed. In the present study, 22 wild-growing Salvia species collected throughout Iran based on their TA content were assessed using high-performance liquid chromatography (HPLC). Agro-morphological traits of the high productive species were then evaluated. The cytotoxic effect of the methanolic extracts of the superior species on three human cancer cell lines MCF-7, SH-SY5Y, and HL-60 by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method was also studied. Our findings could lead to the introduction of a pharmaceutical index species of Salvia for use in breeding and biotechnology programs to produce TAs as valuable pharmaceutical compounds.
In the present work, the contents of TAs, that is, BA, OA, and UA, from the aerial parts of 22 Salvia species native to Iran have been reported here for the first time (Table 1). HPLC results revealed that the amount of TAs studied in Salvia species ranged from 0.17 ± 0.02 to 3.12 ± 0.03 mg/g dry weight (DW) for BA, 0.06 ± 0.01 to 1.96 ± 0.05 mg/g DW for OA, and 0.08 ± 0.07 to 4.34 ± 0.1 mg/g DW for UA. The maximum content of BA (3.12 ± 0.03 mg/g DW) and OA (1.96 ± 0.05 mg/g DW) was observed in the aerial parts of Salvia multicaulis. The aerial parts of cultivated Salvia officinalis contained the maximum content of UA (4.34 ± 0.1 mg/g DW). Salvia multicaulis was the second rich species for UA (3.71 ± 0.08 mg/g DW). Our results showed that TA contents in all studied Salvia species were significantly different (P ≤ 0.05). No traces of BA were detected in 9 species (viz. Salvia aristata Aucher ex Benth., Salvia atropatana Bunge, Salvia candidissima Vahl, Salvia hydrangea DC. ex Benth., Salvia limbata C.A.Mey., Salvia mirzayanii Rech.f. & Esfand., Salvia syriaca L., Salvia urmiensis Bunge, and Salvia xanthocheila Boiss. ex Benth.). In addition, the lowest amount of OA was observed in the three species (namely S. atropatana, S. mirzayanii, and S. xanthocheila). UA was also not detected in Salvia urumiensis (Table 1).
The Content of BA, OA, and UA in Each Salvia Species Studied.
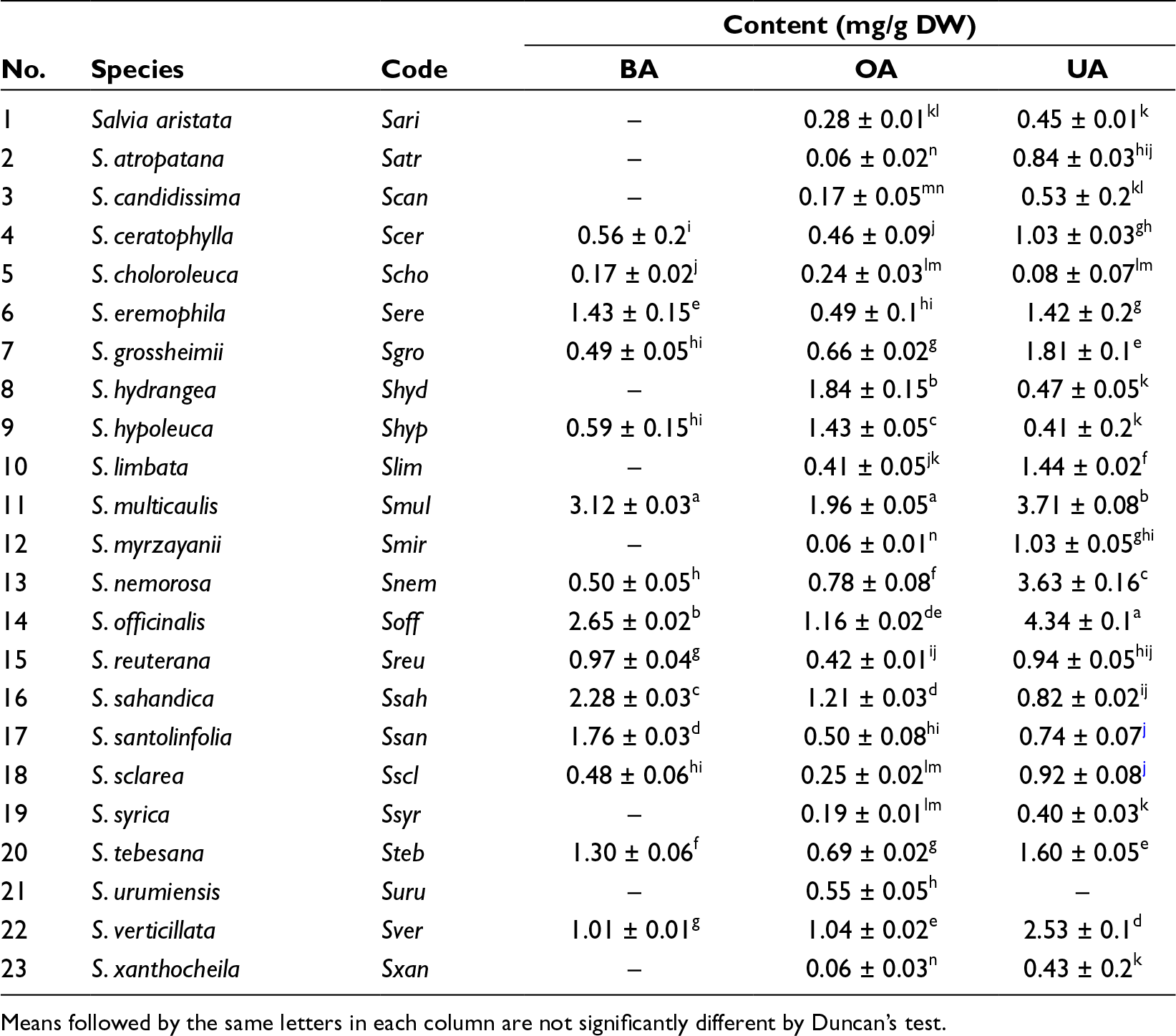
Means followed by the same letters in each column are not significantly different by Duncan’s test.
The analysis of variance (ANOVA, Table 2) revealed that the variation between S. multicaulis and S. officinalis was statistically significant (P ≤ 0.05) for some morphological traits including plant height, plant width, fresh weight, and dry weight. The average of the different measured morphological traits for the two superior Salvia species, that is, S. multicaulis and S. officinalis is presented in Table 3. The analysis of these data shows that the coefficient of variation varies between 3.3% and 29.1%. The results reveal that the variables such as leaf width, internode length, node number, and inflorescence length have a high variation (>10%), whereas plant height, plant width, leaf length, fresh weight, and dry weight present low variation (<10%). S. multicaulis exhibited significantly higher values than S. officinalis in traits related to plant width, leaf length, internode length, and inflorescence length, whereas S. officinalis has significantly higher values in traits related to plant height, leaf width, and node number, as well as fresh weight and dry weight.
ANOVA of Agro-Morphological Characteristics of Salvia Species Studied.

Abbreviations: CV, coefficient of variance; df, degrees of freedom; ns, not significant.
*Significant at 5% level of probability.
Mean Comparison of the Agro-Morphological Characteristics of Salvia Species Studied.

Means followed by the same letters in each column are not significantly different by Duncan’s test.
Understanding and managing the natural diversity among wild species of plants and their affinity with probably domesticated specimens have a very important role in targeted planning for their conservation and breeding. Species diversity in an environment depends on the ability to produce and sustain the ecosystem. 37 Depletion in biodiversity may be due to the environmental effects and due to the reduction in the fertility of the plant community. Determination of genetic and morphological diversity as well as phytochemical variability among wild medicinal plants is of great importance and is a basic step for identifying and preserving heritable reserves of an ecosystem, which forms the basis for genetic research and breeding programs. Due to ecological behaviors, indigenous medicinal plant species, especially their wild populations, are morphologically and phytochemically heterogeneous.
Nowadays, in the cultivation of medicinal plants, the provision of plant materials is considered to be of high security, sustainability, and efficiency. Therefore, in the case of exploitation and introduction of a medicinal species into the mass cultivation system, the study of genetic, morphological characters, and phytochemical diversity is very necessary. Following these principles, 22 indigenous Salvia species of Iran have been screened based on their TA content, and the elite species have been morphologically studied for further breeding and cultivation programs.
So far, many studies on the phytochemical variation of essential oils and extracts of various Salvia species have been carried out. During these researches, some new compounds in the oils and extracts have been identified, or the amount of valuable compounds has been evaluated. The presence of TTs in Salvia species has been previously reported.38-40 The extract of these plants consists mainly of di- and triterpenoids, flavonoids, and sometimes sesquiterpenes. 28 By the beginning of 2011, about 74 TTs and steroids have been isolated from different Salvia species. In a study, 9 new chemical structures including 3 sesterterpenoids, 1 triterpene, and 5 diterpenes were extracted from Salvia palaestina. 41 TTs, especially TAs, are very important plant compounds that are known for their anticancer property. Therefore, the evaluation of the amount of OA, UA, and BA compounds as well-known TTs in various plant species has been considered the goal of numerous researches 26 which have examined the content of these compounds in different species of the Lamiaceae family, including Rosmarinus officinalis L., S. officinalis L., Satureja montana L., S. sclarea L., and Salvia glutinosa L. Quantitative analyses of these species showed that the OA content ranged from 0.09% to 0.9% DW, content of BA ranged from traces to 0.6%, and that of UA varied from 0.09% to 1.6% DW. Quantitative variability of TAs among Salvia species studied could be attributed to the genetic variation as well as definite environmental and climatic conditions. Our results are accordingly in agreement with data reported in the literature obtained in other plants.5,42-45 Comparison of climatic variations of plant species with their phytochemical analyses revealed that the amount of TAs is significantly affected by these climatic changes. The results of the total amount of TAs measured in the studied Salvia species indicated that S. multicaulis can be introduced into agricultural systems as a superior species.
In the present study, the cytotoxic effect of 5 methanolic plant extracts on 3 cancer cell lines (MCF-7, SH-SY5Y, and HL-60) was determined using the MTT assay at a range 0 to 250 μg/mL after 24 hours of treatment. Salvia officinalis extract suppressed the MCF-7 cell line proliferation with the lowest IC50 (41.5 ± 2.0 µg/mL) (Figure 2). Proliferation of SH-SY5Y cells was most significantly reduced by S. multicaulis extract at the lower IC50 (45.9 ± 3.5 µg/mL). Similar inhibitory effects were found in HL-60 cells after incubation with S. multicaulis and S. officinalis L. extract, resulting in a lower IC50 value.

Growth inhibition (IC50) of the 5 Salvia species on the 3 human cancer cell lines.
The methanolic extract of S. multicaulis and S. officinalis showed cytotoxic effects against SH-SY5Y and MCF-7 cell lines, respectively. Methanolic extract of both species showed similar cytotoxic effects against HL-60 cell line (Table 4). The current study has demonstrated that methanolic extract of S. multicaulis and S. officinalis could significantly suppress the proliferation of MCF-7, SH-SY5Y, and HL-60 cell lines. Various biological and pharmacological effects of these compounds including anti-inflammatory, antioxidant, anticancer and antitumor, anti-HIV, antifungal, and immunomodulatory activities have been previously reported. According to the results, cytotoxic effects observed can be attributed to the available TAs in extracts.
In Vitro Growth Inhibitory Activity (IC50 µg/mL) of Some Superior Salvia Species Extracts on Human Cancer Cell Lines.

Owing to the high capacity for the production and accumulation of the mentioned anticancer TAs and their exclusive cytotoxic activity, S. multicaulis and S. officinalis can be considered in further breeding and biotechnological programs for the production of TAs. Based on the results, it can be concluded that (i) Salvia species studied are growing at a latitude ranging from 28°21′ to 38°50′, a longitude ranging from 44°38′ to 55°54′, and an altitude ranging from 1000 to 2760 m in Iran. (ii) The maximum content of BA and OA was observed in the aerial parts of S. multicaulis, whereas the highest content of UA was found in the aerial parts of cultivated S. officinalis followed by S. multicaulis. (iii) Methanolic extract of S. multicaulis and S. officinalis significantly suppressed the proliferation of MCF-7, SH-SY5Y, and HL-60 cell lines. (iv) Salvia multicaulis is a suitable species for domestication and breeding programs to introduce into agronomical systems.
Experimental
Plant Material
The aerial parts of 22 Salvia species including 14 native and 8 endemic species were collected at full flowering stage from their geographical habitat throughout Iran (Figure 3). Salvia officinalis (sage) collected from the botanical garden of Medicinal Plants and Drugs Research Institute, Shahid Beheshti University (35°48′N, 51°23′E at an altitude of 1800 m) was considered a well-known cultivated commercial species to compare with the other indigenous species. The geographical characteristics of each locality are presented in Table 5. The plants’ aerial parts were dried in shade and then transferred to the laboratory to measure the TA content of each species, and a voucher specimen (Table 5) was deposited at the herbarium of Medicinal Plants and Drugs Research Institute (MPH) of Shahid Beheshti University, Tehran, Iran.

Representative species of Salvia studied in their natural habitat and cultivation site: S. aristata (a), Salvia sahendica (b), S. limbata (c), S. ceratophylla (d), S. urmiensis (e), S. hydrangea (f), S. verticillata (g), S. xanthocheila (h), S. reuterana (i), S. atropatana (j), S. multicaulis (k), and S. officinalis (l).
The Vouchers and Geographical Characteristics of Salvia Species Studied.

Extraction, Isolation, and Quantification of TAs
Dried aerial parts (1 g) of each species were extracted by MeOH for the HPLC analyses as described before. 13 The extracts were filtered by using a 2.0-µm filter and were stored in the refrigerator until analysis. A KNAUER liquid chromatography apparatus consisting of a 1000 Smartline Pump, a 5000 Smartline Manager Solvent Organizer, and a 2800 Smartline Photodiode Array Detector was used for the HPLC analysis. Data acquisition and integration were performed with EZChrom Elite software. Separation was achieved on SunFire® C18 column (15.0 × 3.0 mm, 3.5 µm) with a pre-column provided by Waters (Ireland). The isocratic method was employed for separation with a composition of the mobile phase of 87 mL methanol–50 µL phosphoric acid–12 mL + 95 µL water with a flow rate of 0.4 mL min−1. Peaks were monitored at 210 nm wavelength. Injection volume was 20 μL, and the temperature was maintained at 25°C. Analysis and determination of each TAs were carried out as described previously. 46 Standards of BA, OA, and UA were purchased from Sigma (Sigma-Aldrich Corporation, MO, USA). HPLC-grade methanol and phosphoric acid of analytical grade were obtained from Merck (Darmstadt, Germany). HPLC-grade water was used throughout the analysis.
Morphological Analysis
Phenotypic analyses were performed on fully flowered fresh plants and dried herbarium specimens. Nine morphological characteristics, that is, plant height (cm), plant width (cm), leaf length (cm), leaf width (cm), internode length (cm), node number (no.), inflorescence length (cm), fresh weight (g), and dry weight (g) were determined from fresh and herbarium samples.
The investigated morphological traits were related to the aerial parts, especially the important attributes influencing the amount of TAs. Some of the morphological characters were measured at the field or natural habitat and then the samples were transported to the laboratory for further examinations. Fresh weight and dry weight were measured using an electronic balance with 0.01 g precision.
Cytotoxicity Assay
Three cell lines including human breast adenocarcinoma (MCF-7), human neuroblastoma (SH-SY5Y), and human promyelocytic leukemia HL-60 cell lines were used. The MCF-7, SH-SY5Y, and HL-60 cells were grown in culture flasks using Dulbecco’s modified Eagle’s medium, supplemented with 10% heat-inactivated horse serum, 5% heat-inactivated fetal bovine serum, 100 U/mL of penicillin, and 100 µg/mL of streptomycin, in a humidified atmosphere at 37°C with 5% CO2. The growth medium was changed 3 times a week. Cell viability was measured by MTT assay. Cell lines received different treatments, as a control, and different concentrations of plant extract. Briefly, the cells were seeded into 96-well plates (3.0 × 103/well) and cultured for 24 hours. MTT solution (5 mg/mL; Sigma-Aldrich, St Louis, MO, USA) was added to each well and incubated for 4 hours. Next, 150 µL of dimethyl sulfoxide (Sigma-Aldrich) was added to dissolve the formazan precipitate. Absorbance was then measured at 570 nm using a BioTek microplate reader. The experiment was repeated at least 3 times. Cisplatin purchased from Cipla (India) was used as the positive control. Cell viability is expressed as a percentage relative to the untreated control.
Data Analysis
ANOVA was performed for all morphological traits as well as TA contents by SAS software (SAS Inst., 1990). Coefficients of variation (CV%) were determined as indicators of variability. All the experiments were carried out in triplicates. Mean values ± SD (standard deviations) were compared at the 5% (P ≤ 0.05) level of significance using Duncan’s multiple range tests.
Footnotes
Acknowledgment
We wish to thank Mrs. Fatemeh Goudarzi for her kind help in HPLC analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Shahid Beheshti University Research Council for financial support of this project.
