Abstract
The essence of primary ovarian insufficiency (POI) is the premature exhaustion of primordial follicles in the follicle pool, which is caused by the excessive premature activation of primordial follicles after birth. Bisphenol A (BPA) exposure promotes the transition of primordial follicles to primary follicles, thus the number of primordial follicles in the primordial follicle pool decreases significantly. However, the molecular mechanisms underlying abnormal follicle activation are poorly understood. Phosphatase and tensin homologue (PTEN) signal system is a negative regulator of follicle activation, which is called the brake of follicle activation. Besides, BPA induces Michigan Cancer Foundation-7 breast cancer cells proliferation by dysregulating PTEN/serine/threonine kinase/p53 axis. Whether BPA initiates the excessive premature activation of primordial follicles in the mouse ovaries via PTEN signaling pathway is unclear. In this study, we treated 6-week-old female CD-1 mice with different concentrations of BPA to study the effect of BPA on follicular activation and development in vivo, as well as the role of PTEN signaling in this process. We observed that BPA in concentrations from 1 μg/kg to 10 mg/kg groups downregulated PTEN expression and initiated excessive premature activation of primordial follicles in the mouse ovaries, and this effect was partly reversible by PTEN overexpression. Our results improve the understanding of both the effect of BPA in occurrence of POI and molecular mechanisms underlying initiation of primordial follicle pool activation, thus providing insight for POI treatment and theoretical basis for reducing the risk of POI.
Introduction
Primary ovarian insufficiency (POI), referring to cessation of menses associated with secondary amenorrhea, sex steroid deficiency, and elevated serum levels of gonadotropins before the age of 40, 1 confers higher risks of osteoporosis, cardiovascular disease, neurocognitive disorders such as Parkinson disease, 2 endocrine and autoimmune disorders, as well as subfertility. 3 It is a heterogeneous disorder affecting approximately 1% of women aged <40 years. 4 The estimated incidence of POI is approximately 10% to 28% in patients with primary amenorrhea, and 4% to ∼18% in patients with secondary amenorrhea. 5 The causes of POI are not well understood but are thought to be complex, including genetic, environmental, and autoimmune factors. In particular, exposure to environmental factors, such as environmental endocrine disruptors through different ways such as dietary intake, respiration, and skin contact, may interfere with physiological effects of estrogen, androgen, thyroid hormone, and catecholamines. Environmental endocrine disruptors, especially BPA, are probably a cause of POI. 6 Such exposure has also been associated with the occurrence of many reproductive system disorders and even tumors. 7,8
BPA, 2,2-bis(4-hydroxyphenyl)propane or diphenol propane, is one of the most abundant chemical products in the world. 9 Widely used in mineral water bottles, food-can linings, and other daily plastic products, it is an important environmental endocrine disruptor. The chemical structure of BPA is similar to estrogen and has weak estrogenic activity, so BPA may interfere with normal biological endocrine function directly or through its derivatives. 10 –12 Accumulating evidence suggests that the effects of BPA on follicle development are related to stage of follicular development. 13 Previous animal studies have demonstrated that BPA could cause a significant decrease in the primordial follicle pool in newborn and immature rodents. 14 –17 However, the mechanisms underlying the effects of BPA on the follicles development in mature mice and human are not well understood.
The initiation of primordial follicle growth, during a gonadotropin-independent period, refers to the transformation of primordial follicles into primary follicles. Molecular regulatory mechanisms of primordial follicle development are complex and have not been well studied. Current studies suggest that most signal molecules involved in regulation of primordial follicle activation act through phosphatase and tensin homologue (PTEN)/PI3K signal pathway. 18,19 PI3K/AKT/mTORC1 pathway plays a very important role in overactivation of primordial follicles. 20 –22 PTEN, as a phosphatase to dephosphorylate phosphoinositide substrates, plays an important negative role in the dormancy and activation of primordial follicle pool which is called the brake of follicle activation. 23 –25 Previous study has shown that BPA may also promote proliferation of breast cancer through the PTEN-PI3K-AKT axis. 26 However, mechanisms of BPA-mediated primordial follicle activation remain unclear and few studies have been conducted.
Given roles for BPA and PTEN-PI3K-AKT in modulating the primordial follicle pool, we hypothesized that BPA might inhibit PTEN, to cause excessive premature activation of primordial follicles through PI3K/AKT signaling, which might be an important factor in the occurrence of POI. Therefore, we focused on the PTEN-PI3K-AKT signaling pathway in our examination of in vivo effect of BPA on follicular activation and development. In this study, 6-week-old female CD-1 mice were orally administered various concentrations of BPA for 14 to 42 days, and the effects of BPA on follicular activation and development as well as the depletion effect on primordial follicle pool were observed. We also assessed the modulation of the PTEN signaling pathway in the premature activation initiated by BPA.
Material and Methods
Ethics Statement
Female CD-1 mice were used in this study. All animal procedures were conducted under the protocol approved by the Ethics and Scientific Research Committee in Sichuan University. All efforts were made to minimize suffering.
Reagents
Bisphenol A (BPA, 99% purity; Sigma-Aldrich, St Louis, Missouri), corn oil (Sigma-Aldrich), dimethyl sulfoxide (DMSO; Sigma-Aldrich), human chorionic gonadotropin (hCG; Sigma-Aldrich), pregnant mare serum gonadotropin (PMSG; Sigma-Aldrich), rabbit anti-PTEN antibody (ab32199; Abcam, England), phospho-Akt (Thr308) antibody (9275 S; CST, USA), Phospho-Akt (Ser473) antibody (9271 S; CST), goat anti-rabbit IgG (H+L) highly cross-adsorbed secondary antibody (A11034; Alexa Fluor 488, Thermo Fisher, Waltham, USA), hyaluronidase (Sigma-Aldrich), M2 medium (Sigma-Aldrich), Ad-PTEN-eGFP (adenovirus expressing PTEN-enhanced green fluorescent protein; GeneChem, Shanghai, China), mouse progesterone (PROG) enzyme-linked immunosorbent assay (ELISA) kit (CSB-E05104m; CUSABIO, Wuhai, China), mouse estradiol (E)ELISA kit (CSB-E07280 m; CUSABIO), testosterone ELISA(T) kit (ADI-900-065; ENZO, Swiss Confederation, CH), TRIzol reagent (Ambion, California, CA), RevertAid First Strand cDNA Synthesis Kit (Thermo, Lithuania, EU), SYBR Green Master Mix (Applied Biosystems, Austin, TX, USA), RIPA buffer (Sigma Aldrich), protease inhibitors (Roche, Mannheim, Germany), BCA protein assay kit (Pierce, Rockford, Illinois), BSA (Amersco, Chengdu, China), chemiluminescence reagents (Millipore, Billerica, Massachusetts).
Preparation of Adenovirus Expressing PTEN
To construct an adenoviral vector, mouse PTEN cDNA was amplified using the following primer: 5′-AGGTCGACTCTAGAGGATCCCGCCACCATGACAGCCAT CATCAAAGAGATC-3′ and 5′-TCCTTGTAGTCCATACCGACTTTTGTAATTTGT GAATGCTG-3′ and was cloned into pGV314 vector. Ad-PTEN-eGFP, adenovirus with double expressing of PTEN and eGFP and Ad-eGFP, adenovirus expression GFP only, were prepared by GeneChem (Shanghai, China). A total of 2 × 109 plaque forming unit Ad-PTEN-eGFP or Ad-eGFP were injected into each mouse via tail vein.
Animals and Experimental Design
CD-1 mice were obtained from Chengdu Dashuo Experimental Animal Co. Ltd (2015-030). All mice were housed in temperature-controlled (20-21°C) and light-controlled (12-hour light:12-hour dark cycle) conditions. Food (Formulab Diet for rodents; Dashuo, Chengdu, China) and high purity water were provided ad libitum.
Two-hundred and forty-six 6-week-old female CD-1 mice were divided into 3 parts to conduct 3 experiments (Supplementary Data 1).
Experiment 1: Evaluation of Follicles and the Activity of PTEN Signaling Pathway After BPA Treatment
Seventy-eight mice were divided into 6 groups and orally administered 0.5 mL of corn oil containing BPA-dissolved 0.1% DMSO at doses of 0, 1 μg, 10 μg, 100 μg, 1 mg, and 10 mg per/kg of body weight every day, respectively, for 28 days; mice were killed in the estrous cycle determined by vaginal lavage and ovaries were collected. The route of administration and doses of BPA was chosen based on prior studies utilizing this method of exposure, which could represent the actual conditions via oral administration. 9,27 Ovaries from one side were used for serial section and hematoxylin and eosin (H&E) staining to evaluate the follicles, while ovaries from the other side were used for fluorescence immunocytochemistry, reverse transcription-polymerase chain reaction (RT-PCR), and Western blotting to observe the expression of PTEN.
Experiment 2: PTEN Overexpression by Adenovirus Transfection
To validate the BPA effect on the PTEN axis, 78 mice were divided into 6 groups, orally administered BPA at doses of 0, 1 mg, and 10 mg per/kg body weight every day in 0.1% DMSO or DMSO alone as a vehicle control for 28 days. Mice were transfected with Ad-PTEN-eGFP or Ad-eGFP at the beginning and the 14th day, respectively. They were killed during the estrous cycle determined by vaginal lavage. Ovaries were collected and examined as previously described.
Experiment 3: Superovulation Test
To assess the depletion effect of BPA on primordial follicles, 90 mice were divided into 3 groups and orally administered BPA at doses of 0, 100 μg, and 10 mg per/kg body weight every day. In each group, mice were divided into 3 groups again and were induced superovulation on the 14th, 28th, and 42th days, respectively. After superovulation, mice were killed for evaluation of the structure of oocytes.
Ovarian Collection
All ovarian samples were free of oviduct, fat, and bursa. Ovaries from one side were immediately frozen in liquid nitrogen and subsequently stored at −80°C until RNA extraction and protein quantification. Ovaries from the other side were fixed in Dietrich’s solution for serial section and H&E staining.
H&E Staining and Follicles Counting
Ovaries (1 per mouse) were fixed in Dietrich’s solution for at least 72 hours before being processed for histology using standard protocols. Briefly, the ovaries were for routine paraffin embedding and serially sectioned (6-μm thickness) throughout the entire ovary. Sections were adhered and stained on slides in H&E using standard procedures. Follicle populations were counted at every third section of the entire ovary and were scored as primordial, primary, secondary, antral, or Graafian follicles, based on their morphological appearance, as detailed in the literature. 28
RNA Extraction and Quantitative RT-PCR Analysis
Tissues were homogenized in TRIzol reagent, and total RNA was extracted according to manufacturer’s instructions. Reverse transcription of 2 μg total RNA was performed with the RevertAid First Strand cDNA synthesis kit. Real-time PCR was performed with SYBR Green Master Mix in a 20-µL PCR reaction system, mixture consisting of 10 µL of SYBR Mix, 6 µL of cDNA, 1 µL of 10 µM forward/reverse primer, and 2 µL of RNase-free water. The quantitative PCR (qPCR) was performed on an Applied Biosystems 7500 Real-Time PCR System as follows: 50°C for 2 minutes, 95°C for 2 minutes, and 40 cycles of 95°C for 15 seconds, and 60°C for 60 seconds. The qPCR results were automatically analyzed using the Applied Biosystems 7500 system. The 2−ΔΔct method was used to calculate the relative gene expression. Sequences of reverse transcription primers are shown in Table 1.
Primers for RT-qPCR.
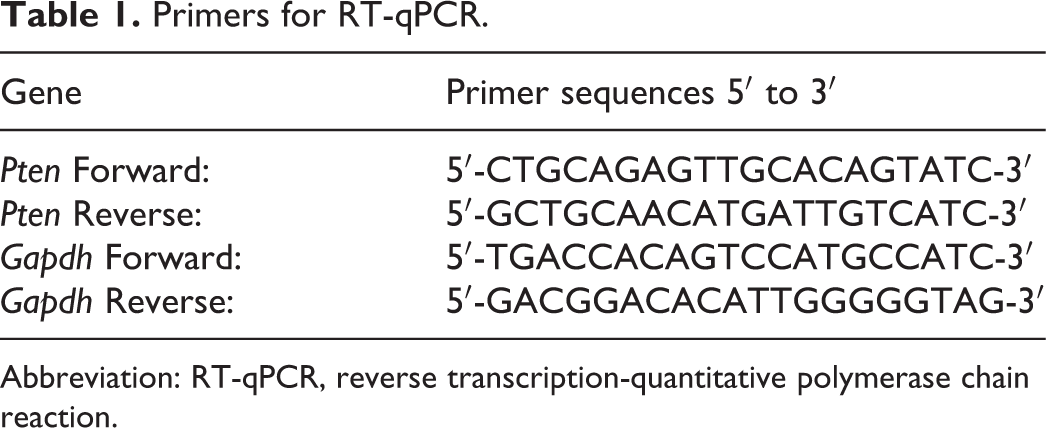
Abbreviation: RT-qPCR, reverse transcription-quantitative polymerase chain reaction.
Protein Quantification by Western Blotting
Lysates of total ovarian proteins were prepared in RIPA buffer containing protease inhibitors, and protein concentrations were determined using the BCA protein assay kit. Equal amounts of protein were loaded on 10% Sodium dodecylsulphate polyacrylamidegel electrophoresis (SDS-PAGE). After electrophoresis, proteins were transferred to polyvinylidene fluoride membranes. Blots were blocked with 5% BSA at room temperature for 1 hour and incubated with the indicated primary antibodies overnight at 4°C. Then the blots were washed 3 times with tris-buffered saline containing 0.1% Tween 20 (TBST) and incubated with Horseradish Peroxidase (HRP)-conjugated secondary antibodies for 1 hour at room temperature. After washing with TBST again 3 times for 15 minutes, blots were incubated with enhanced chemiluminescence reagents and then exposed to film for detection. The blots were quantified by densitometric analysis using the software Quantity One.
Fluorescence Immunocytochemistry
After deparaffinization, ovarian sections were blocked with normal goat serum for 30 minutes at room temperature. Rabbit anti-PTEN antibody, phospho-AKT (Thr308) antibody, and phospho-AKT (Ser473) antibody were applied as the primary antibodies. Sections were incubated in a sealed, humidified chamber for 2 hours at 37°C and then washed with phosphate-buffered saline (PBS). Specimens were then incubated with goat anti-rabbit IgG (H+L) highly cross-adsorbed secondary antibody for 1 hour at room temperature. After washed with PBS, sections were evaluated under a Leica DM4000 biological microscope equipped with epifluorescent illumination. Raw images were analyzed using Leica LAS Image Analysis by quantifying the average intensities. 29
Superovulation
To induce superovulation, animals were injected intraperitoneally (IP) with PMSG 6 IU and given hCG 6 IU IP 48 hours later. Animals were killed by an IP overdose of equithesin (4 mL/kg, 1% sodium pentobarbital), 13–15 hours after hCG administration. The mice were fasted overnight before being killed. Afterward, their oviducts were collected and then placed in the M2 medium, covered by mineral oil. Cumulus cell–oocyte complexes were removed from their oviductal ampullae, transferred to drops of M2 medium containing hyaluronidase solution (300 mL/mL) and manually denuded with a glass pipette to allow precise assessment of oocyte morphology. Maturation stage and oocyte structure were evaluated, as previously detailed in the literature. 30
Hormone Assays
Serum samples were obtained and stored at −20°C until use for hormone assays. Serum levels of estradiol (E2), progesterone (P), and testosterone (T) were tested by mouse estradiol (E2), progesterone, and testosterone (T) ELISA kit. Assay sensitivities were 25 pg/mL, 0.2 ng/mL, and 5.67 pg/mL for E2, P, and T, respectively.
Statistics
Analysis of variance was performed to evaluate significant differences among groups in the vivo assays. All statistical tests were 2-sided, and P values less than .05 were considered to be statistically significant. SPSS software (version 22.0) was used for all statistical analyses.
Results
BPA Exposure Promotes the Premature Activation of Primordial Follicles In Vivo
Six-week-old female CD-1 mice were orally given BPA (at doses of 1 μg/kg, 10 μg/kg, 100 μg/kg, 1 mg/kg, and 10 mg/kg per day) for 28 consecutive days. The mice were weekly weighed, and the data were graphically represented. Weekly weight gain was noticed in all mice; however, there were no significant difference in body weights in each treatment group and control group at each time point (Figure 1E). Mouse ovarian serial sections with H&E staining were evaluated by counting the different stages of follicles, defined as primordial, primary, secondary, antral, or Graafian follicles (Figure 1A), and calculating the proportions of primordial follicles. The ratios of primordial follicles to primary follicles and ratios of primordial follicles to total follicles in animals administered various concentrations of BPA (10 mg/kg, 1 mg/kg, 100 μg/kg, and 10μg/kg) were significantly lower than that in the control group, with a significant dose–response relationship (Figure 1B,C). These data suggested BPA exposure promoted the premature activation of primordial follicles. Compared with other groups, the group administered 10 mg/kg BPA exhibited significantly higher T levels in hormone assays, and there was a trend that BPA-treated groups exhibited higher E2 levels, however, with no statistical significance, whereas values of P were similar among groups (Figure 1D).
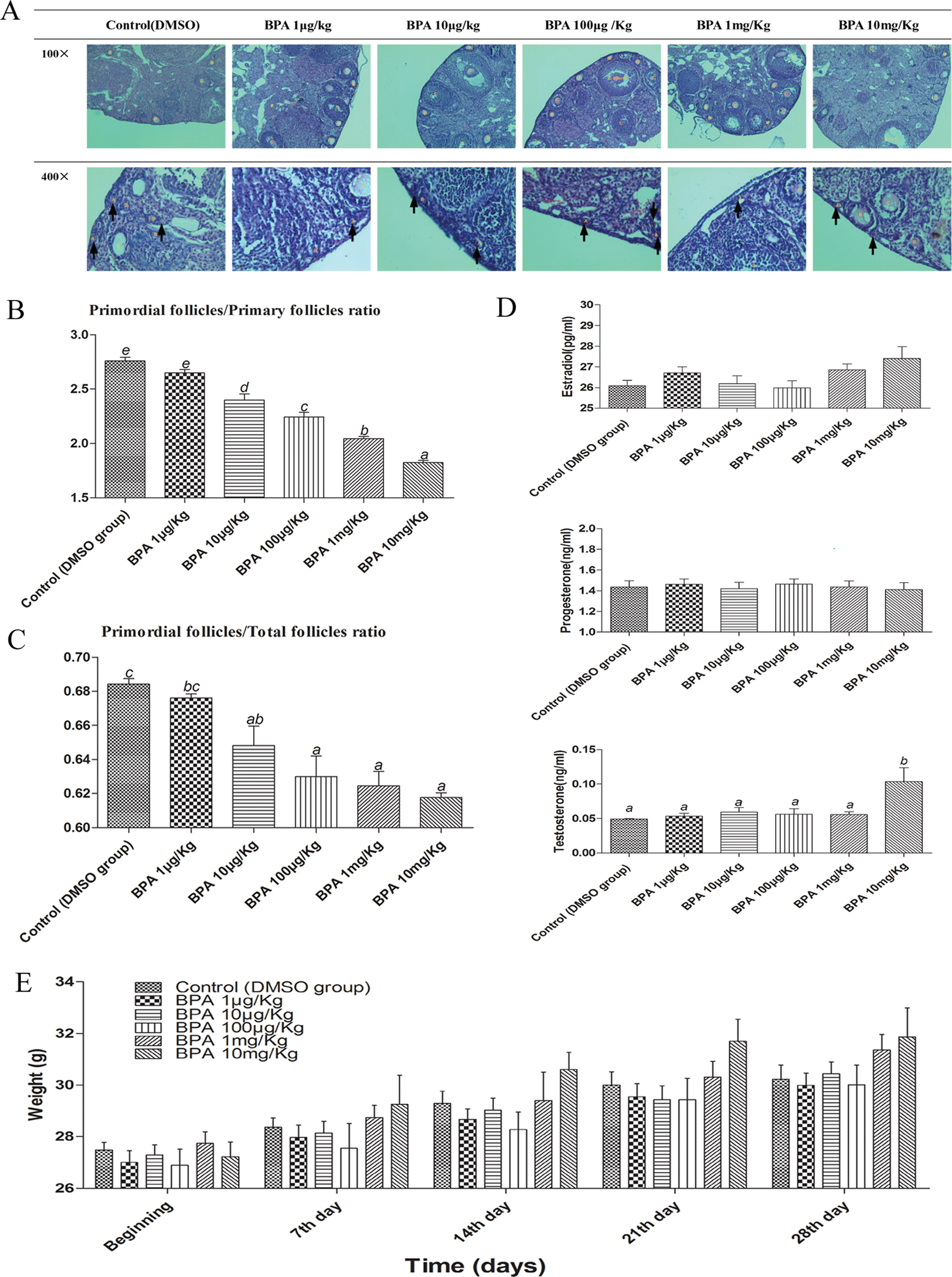
BPA exposure promotes the premature activation of primordial follicles in vivo. A, H&E staining of different stages of follicles in different concentrations of BPA (A-primordial follicles, B-primary follicles, C-secondary follicles, D-antral follicles, E-atretic follicles, and G-Graafian follicles, and arrows pointed to A-primordial follicles). B, Primordial follicles to primary follicles ratio in ovarian analysis in vivo, n = 8. C, Primordial follicles to total follicles ratio in ovarian analysis in vivo, the total follicles means the total number of primordial, primary, secondary, antral, atretic, and Graafian follicles, n = 8. D, Hormones (estradiol, progesterone, and testosterone) in different concentrations of BPA, n = 10. E, Mouse weights in different concentrations of BPA, n = 10. The results are expressed as mean ± SEM; different lower-case letters above the columns, such as a, b, c, d, and e, indicate P < .05, and if 2 columns have the same lower case letter, it indicates no statistical significance. BPA indicates bisphenol A; SEM, standard error of the mean.
BPA Activates PTEN/PI3K/AKT Signaling in Mouse Ovaries
In all BPA and control groups, phosphorylated AKT (p-AKT) at Thr308 and Ser473 were all expressed in mouse ovaries, primordial and primary follicles which we especially observed (Table 2). Compared with the control group, in BPA-treated groups, PTEN expression was suppressed significantly, whereas the expression of p-AKT(Thr308) and p-AKT(Ser473) was increased by fluorescence immunocytochemistry and Western blotting, especially at comparatively high concentrations. These data suggested a role for the PTEN/PI3K/AKT axis in modulating the premature follicular activation effects of BPA (Figure 2).
Summary of Fluorescence Immunocytochemistry for Localization of PTEN, p-AKT(Thr308), and p-AKT(Ser473) in BPA and Control Groups.
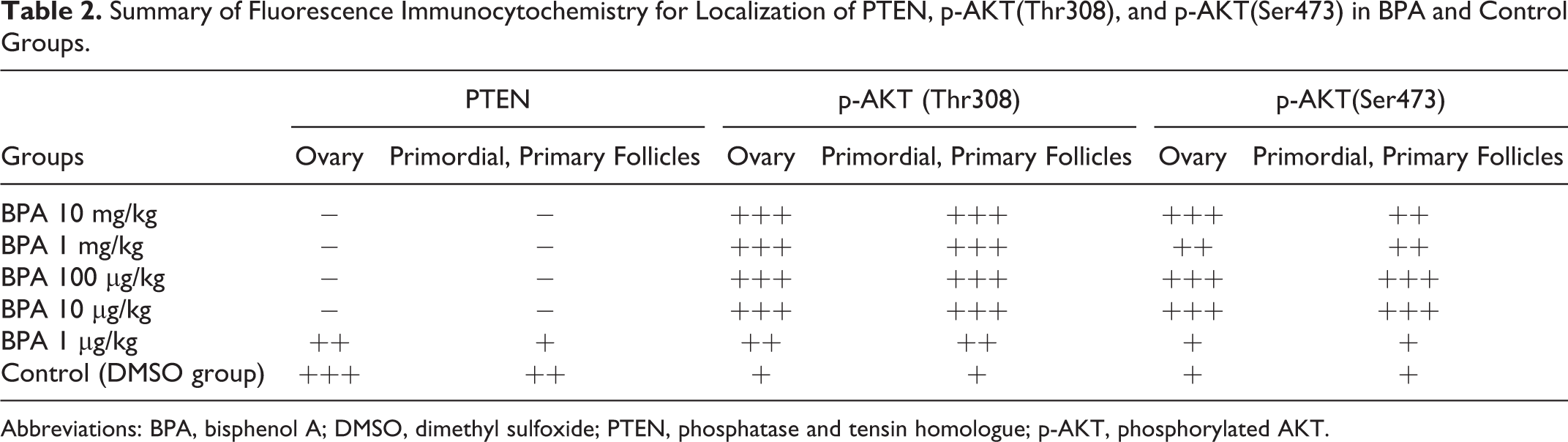
Abbreviations: BPA, bisphenol A; DMSO, dimethyl sulfoxide; PTEN, phosphatase and tensin homologue; p-AKT, phosphorylated AKT.
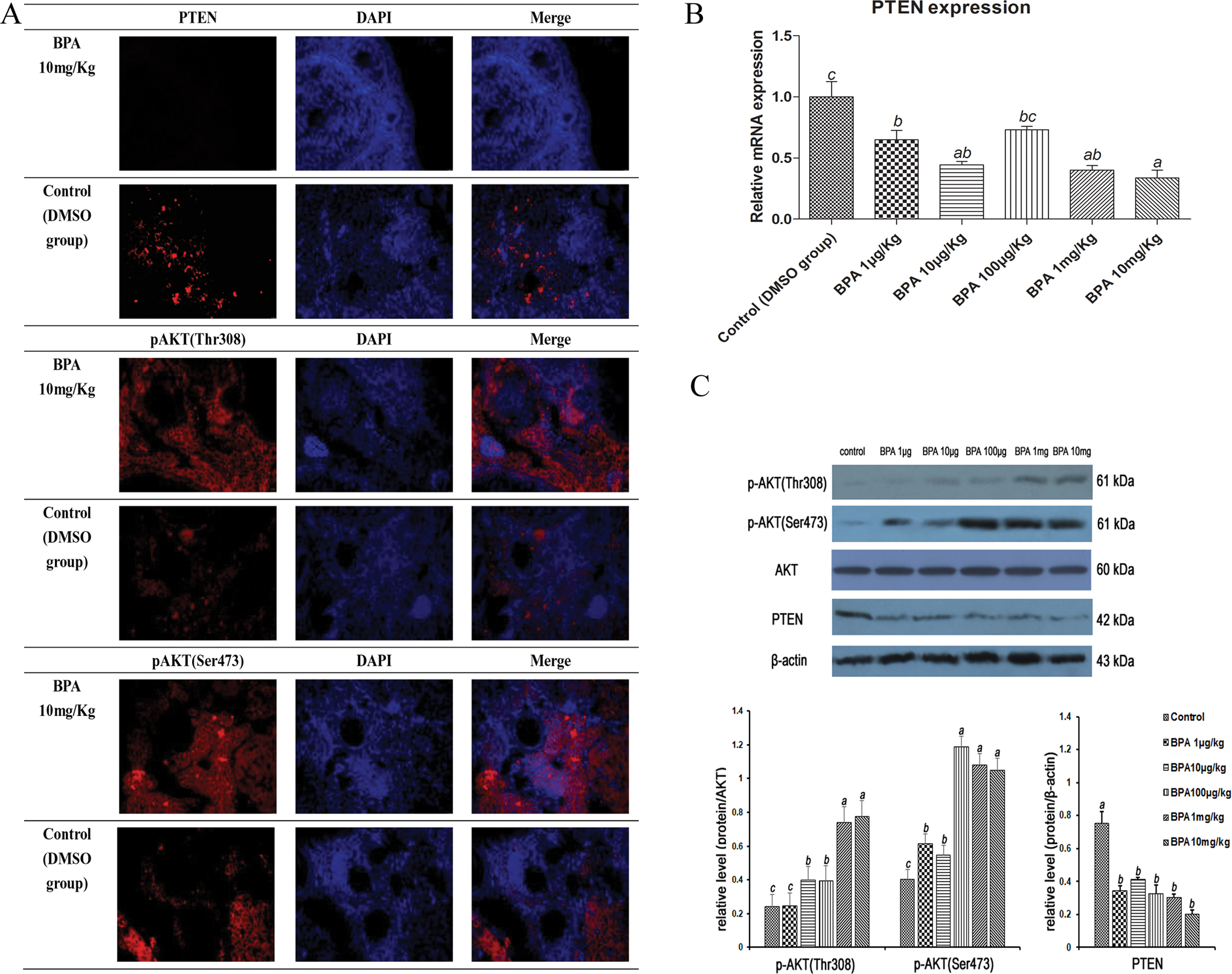
BPA activates PTEN/PI3K/AKT signaling in mouse ovaries. A, Fluorescence immunocytochemistry for localization of PTEN, p-AKT(Thr308), and p-AKT(Ser473). B, PTEN expression by RT-PCR, n = 5. C, Expression of PTEN, p-AKT(Thr308), and p-AKT(Ser473) by Western blotting. The results are expressed as mean ± SEM; different lower case letters above the columns, such as a, b, c, d, and e, indicate P < .05; and if 2 columns have the same lower case letter, it indicates no statistical significance. BPA indicates bisphenol A; PTEN, phosphatase and tensin homologue; RT-PCR, reverse transcription-polymerase chain reaction; p-AKT, phosphorylated AKT; SEM, standard error of the mean.
PTEN Overexpression Can Reverse the Excessive Activation of Primordial Follicles
To investigate whether the suppression of PTEN was initiated by BPA, we carried out a PTEN overexpression experiment. We transfected the mice by injection of adenovirus vector expressing PTEN and/or eGFP via tail vein, 2 weeks after the second injection, eGFP signals appeared in the ovaries in both Ad-PTEN-eGFP and Ad-eGFP groups (Supplementary Data 2), while the overexpression of PTEN was achieved in Ad-PTEN-eGFP group by fluorescence immunocytochemistry, qPCR, and Western blot.
In Ad-eGFP groups, the ratios of primordial follicles to primary follicles and ratios of primordial follicles to total follicles in BPA 1 mg/kg and 10 mg/kg group were significantly lower than DMSO control, and a dose–response relationship which was similar to the above results was observed. However, in Ad-PTEN-eGFP groups, these ratios in BPA-treated groups were only slightly lower than the control group and had no statistical significance (Figure 3B,C). These results show that PTEN overexpression reversed the excessive activation of primordial follicles caused by BPA.
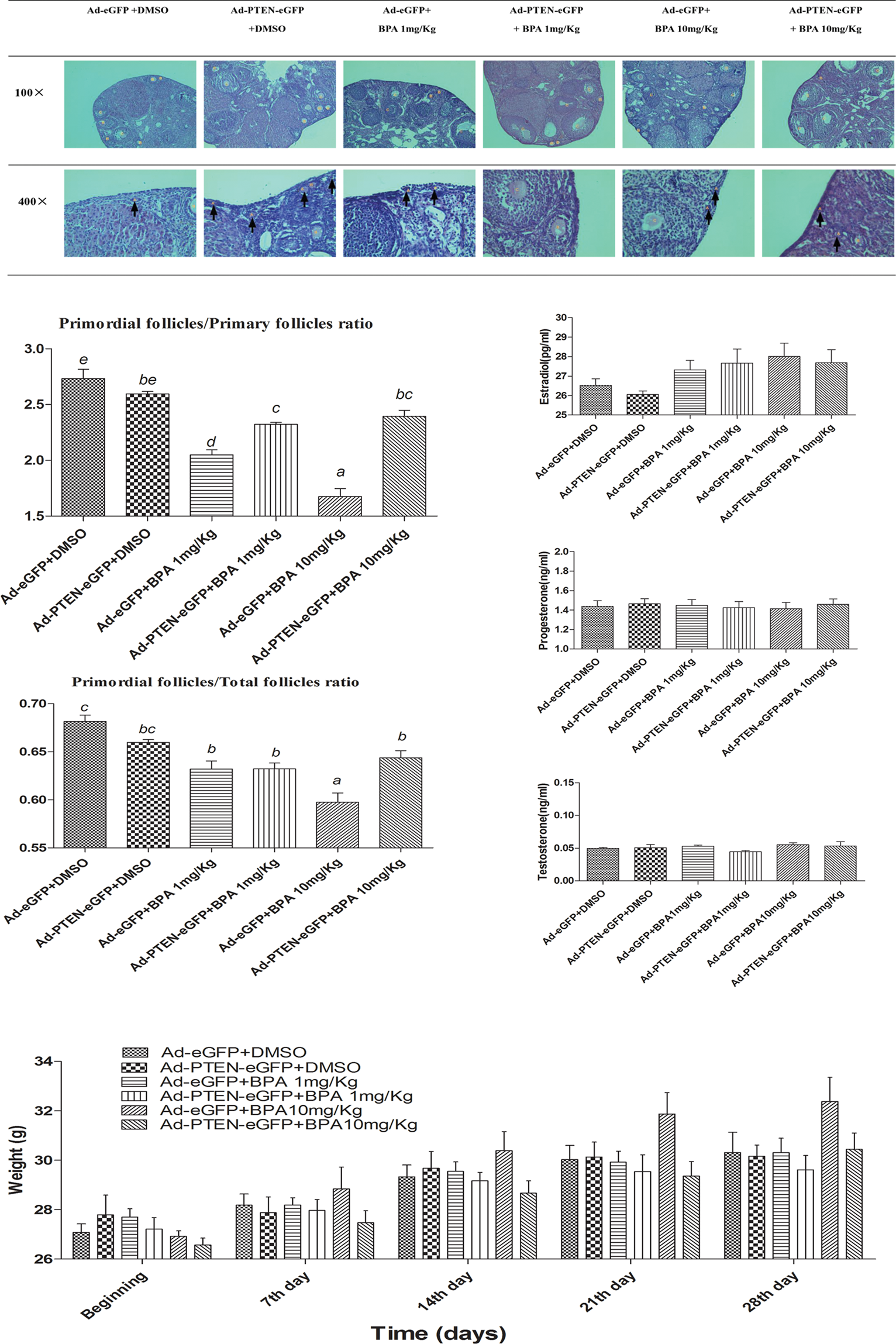
PTEN overexpression can reverse the excessive activation of primordial follicles. A, H&E staining of different stages of follicles in Ad-PTEN-eGFP groups and Ad-eGFP groups (A-primordial follicles, B-primary follicles, C-secondary follicles, D-antral follicles, E-atretic follicles, and G-Graafian follicles, and arrows pointed to A-primordial follicles). B, Primordial follicles to primary follicles ratio in ovarian analysis in vivo, n = 8. C, Primordial follicles to total follicles ratio in ovarian analysis in vivo, the total follicles means the total number of primordial, primary, secondary, antral, atretic, and Graafian follicles, n = 8. D, Hormones (estradiol, progesterone, and testosterone) in Ad-PTEN-eGFP groups and Ad-eGFP groups, n =10. E, Mouse weights in Ad-PTEN-eGFP groups and Ad-eGFP groups, n = 10. The results are expressed as mean ± SEM, different lower case letters above the columns, such as a, b, c, d, and e, indicate P < .05; and if 2 columns have the same lower case letter, it indicates no statistical significance. PTEN indicates phosphatase and tensin homologue; Ad, adenovirus; SEM, standard error of the mean; eGFP, enhanced green fluorescent protein; RT-PCR, reverse transcription-polymerase chain reaction.
We also noticed the mice body weights weekly after virus injection and found the change in body weights was same to the above experiment, and there was no significant difference among each group (Figure 3E). In addition, the values of E2, P, and T were also similar by hormone assay (Figure 3D).
PTEN Overexpression Can Reverse Excessive PI3 K/AKT Signaling Induced by BPA
In Ad-eGFP groups, the dose–response suppressed expression of PTEN was detected after BPA treatment; in the meantime, the increased expressions of p-AKT(Thr308) and p-AKT (Ser473) in BPA-treated groups were demonstrated by Western blot and fluorescence immunocytochemistry (Table 3). In PTEN overexpression groups, transfected with Ad-PTEN-eGFP, the expression level of PTEN was higher than that in Ad-eGFP group; even though the dose–response was suppressed after BPA treatment, it was still higher than that in Ad-eGFP groups. Compared with Ad-eGFP groups, the expressions of p-AKT(Thr308) and p-AKT(Ser473) in Ad-PTEN-eGFP groups were reduced remarkably. After BPA treatment, p-AKT(Thr308) and p-AKT(Ser473) expressions were slightly increased, but they were still remarkably less than that in Ad-eGFP groups, respectively, which was consistent with the change in PTEN expression. These data suggested that PTEN overexpression can partly reverse BPA-mediated excessive activation of the PI3K/AKT signaling pathway (Figure 4).
Summary of Fluorescence Immunocytochemistry for Localization of PTEN, p-AKT(Thr308), and p-AKT(Ser473) in Ad-PTEN-eGFP Groups and Ad-eGFP Groups.
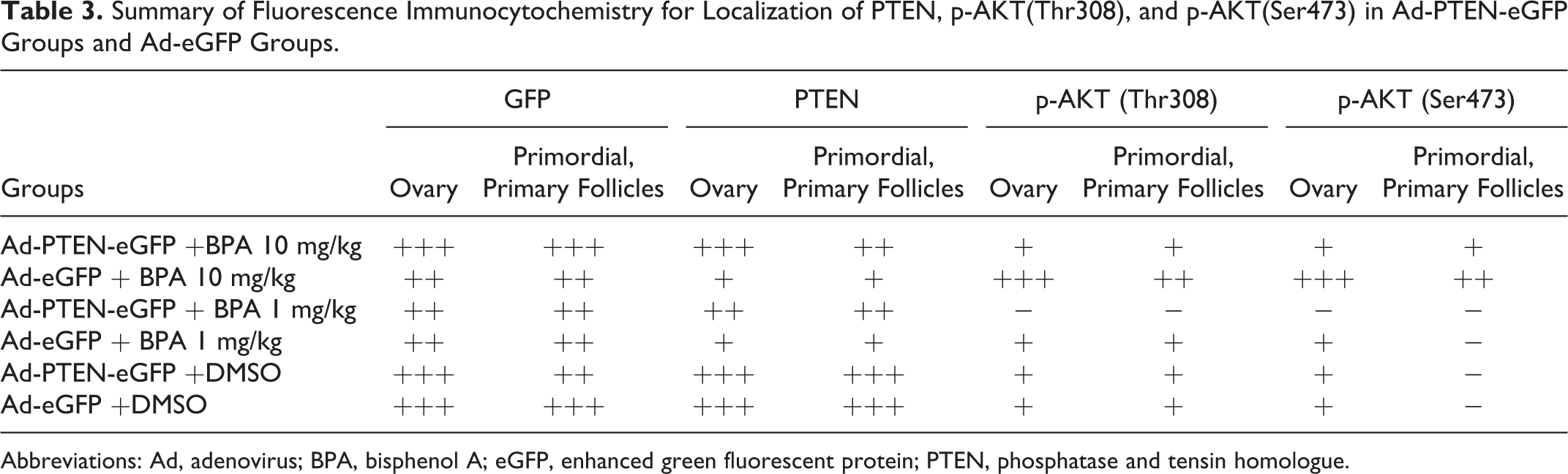
Abbreviations: Ad, adenovirus; BPA, bisphenol A; eGFP, enhanced green fluorescent protein; PTEN, phosphatase and tensin homologue.
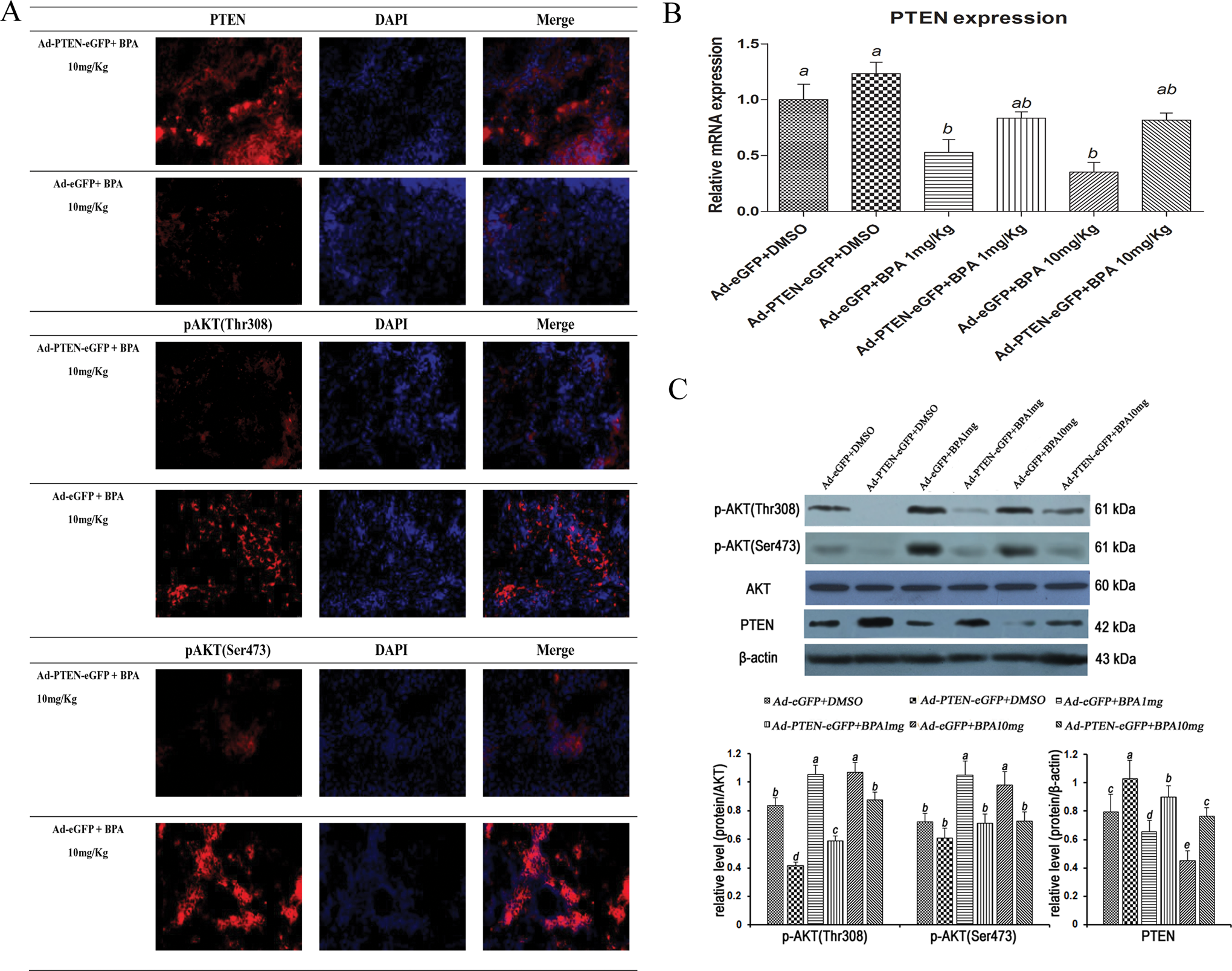
PTEN overexpression can reverse excessive PI3K/AKT signaling induced by BPA. A, Fluorescence immunocytochemistry for localization of PTEN, p-AKT(Thr308), p-AKT (Ser473) in Ad-PTEN-eGFP groups and Ad-eGFP groups. B, PTEN expression by RT-PCR, n = 5. C, Expression of PTEN, p-AKT(Thr308), and p-AKT (Ser473) by Western blotting. The results are expressed as mean ± SEM; different lower-case letters above the columns, such as a, b, c, d, and e, indicate P < .05; and if 2 columns have the same lower case letter, it indicates no statistical significance. PTEN indicates phosphatase and tensin homologue; BPA, bisphenol A; Ad, adenovirus; SEM, standard error of the mean; eGFP, enhanced green fluorescent protein; p-AKT, phosphorylated AKT; RT-PCR, reverse transcription-polymerase chain reaction.
BPA Accelerates Primordial Follicle Pool Depletion
To assess the depletion effect of BPA on primordial follicles, superovulation was performed on the mice which differed from the dose and length of BPA. When the mice were treated for 14 days and 28 days, the percentages of healthy oocytes retrieved were similar in treatment groups and control group. However, when the mice were administered with BPA 10 mg/kg for 42 days, the percentages of healthy oocytes were significantly lower than that in 100 μg/kg BPA-treated group and control group with same time length (Figure 5). Collectively, these data indicate that longer time and higher dose exposure of BPA may accelerate depletion of the primordial follicle pool.
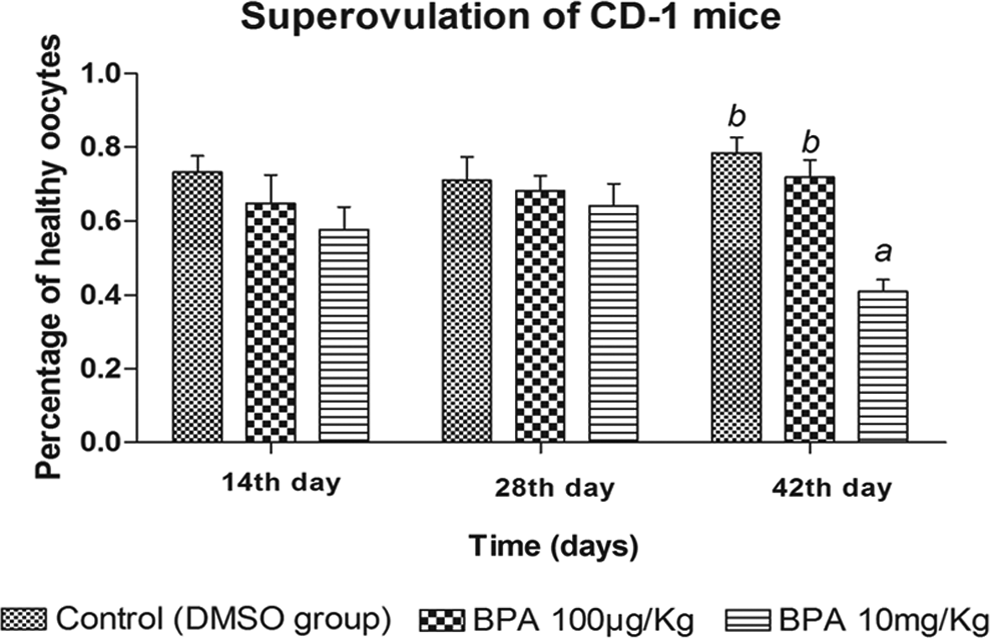
BPA accelerates primordial follicle pool depletion. Percentages of healthy oocytes retrieved after superovulation in BPA groups (10 mg/kg, 100 μg/kg) and control group, the percentages of healthy oocytes = healthy oocytes/(healthy oocyte s+ immature oocytes and/or presence of morphologic abnormalities), n = 10. The results are expressed as mean ± SEM; different lower case letters above the columns, such as a, b, c, d, and e, indicate P < .05; and if 2 columns have the same lower case letter, it indicates no statistical significance. BPA indicates bisphenol A; SEM, standard error of the mean.
Discussion
Folliculogenesis can be divided into 3 stages: follicle recruitment, selection, and ovulation, and has 2 phases: the initial gonadotropin-independent phase and the later gonadotropin-dependent phase. 31 The dynamic change of primordial follicle pool depends not only on the number of primordial follicles in the embryonic stage but also on the rate of follicular depletion after birth, which are thought to be 2 major mechanisms of POI. 32 Clinical epidemiological data have shown an increasing incidence of POI in recent decades. In addition to genetic factors, BPA, as the most widespread environmental endocrine disruptor, may account for the occurrence of POI partially. 6
Currently, the effect that BPA promotes the transition of primordial to primary follicles has been noticed. 13 Animal experiments have shown that exposure of BPA on pregnant mice reduced the number of primordial follicles on their off-spring. 16 Also the exposure of BPA on newborn mice or immature female rat significantly decreased the ratio of primordial/primary follicles in their ovaries. 15,17 Rivera et al. 14 proposed that BPA could stimulate both initial recruitment of the primordial follicle pool and subsequent follicular development, with accelerated folliculogenesis resulting in a significantly decreased number of primordial follicles. However, no existing studies till now reveal that whether BPA can affect the follicular development of the mature mice. In this study, we treated sexual mature CD-1 mice with 5 consecutive concentration gradients of BPA (1 μg/kg, 10 μg/kg, 100 μg/kg, 1 mg/kg, and 10 mg/kg) for 28 days and found that compared with the control group, the ratios of primordial follicles to primary follicles and ratios of primordial follicles to total follicles were significantly decreased in animals administered various concentrations of BPA, especially above 10 μg/kg, and a dose–response relationship was observed. Therefore, it is obvious that BPA exposure can promote the transition of primordial into primary follicles and leads to premature activation of primordial follicles not only in immature animals but also in mature animals. During the experiment, we observed that mice treated with BPA of different doses had irregular estrus cycles, with a prolonged proestrus and diestrus compared to controls, which was similar with previous studies. 33 –35 We also found that there was no statistical significance of CL in different groups. Consistently, Moore-Ambriz et al. reported that young adult female C57BL/6 J mice (39 days of age) orally administered 50 μg/kg BPA daily for a period encompassing the first 3 reproductive cycles (12–15 days) did not exhibit altered parameters related to ovulation after BPA exposure, 36 and same findings of CL were also reported in other studies. 33,34 Until recently, evidence showed that BPA exposure of <50 mg/kg/day did not affect much on periodic recruitment or the ovulation in animals, but more and more studies and our experiment confirmed the role of BPA on the dynamics of primordial follicle pool and the excessive premature activation of primordial follicles.
It has been suggested that BPA can interfere with endocrine function of hypothalamic–pituitary axis 37 and impairs ovulation. Fernández et al. 33 found that irreversible alteration in hypothalamic–pituitary–gonadal axis caused by exposure to 500 μg/kg/day BPA for 10 days in rats could lead to anovulation and infertility. Moore-Ambriz et al. 36 reported that young adult female C57BL/6 J mice (39 days of age) orally administered 50 μg/kg BPA daily for a period encompassing the first 3 reproductive cycles (12–15 days) did not exhibit altered parameters related to ovulation after BPA exposure, but instead influenced oocyte quality in terms of its fertilization ability. In our superovulation test, we treated the 6-week female CD-1 mice with 3 different concentrations (high-dose BPA 10 mg/kg, low-dose BPA 100 μg/kg, and blank control group) for 14 days, 28 days, and 42 days, respectively, to examine long-term effects of continuous BPA on ovulation. The results showed that the percentage of healthy oocytes was significantly decreased only in high-dose and longer time group (BPA 10 mg/kg, for 42 days). Similar results were found in in vitro studies in both animal and human. Nakano et al. reported that oocytes exposed to high doses of BPA (50 μg/mL and 100 μg/mL) experienced a cell cycle delay, but managed to progress to MII when the culture period was prolonged, which inhibited oocyte maturation. 38,39 These data indicate that continuous long-term exposure to BPA, especially high concentrations, may accelerate depletion of primordial follicle pool. It is hard to define a harmful dose range of BPA on ovulation since the methodological limitations preclude the comparison of these study results.
Most signaling molecules involved in the regulation of primordial follicle activation act through the PTEN/PI3 K signaling pathway. 18,19 Reddy et al. 23 first used transgenic technology to selectively knock out PTEN gene in mice oocytes and found that primordial follicles with Pten deletion had largely been activated before sexual maturity, leading to premature ovarian failure and infertility subsequent to exhaustion of primordial follicles in early adulthood. Zhao et al. 40 treated an in vitro neonatal ovarian culture system with BPA 0.1 μM, 1 μM, and 10 μM for 10 days and found that BPA induced primordial follicle activation through the PI3K/Akt pathway. In this study, by fluorescence immunocytochemistry, RT-qPCR, and Western blot, we observed that BPA downregulated the expression of PTEN, along with increased levels of p-AKT(Thr308) and p-AKT (Ser473) after 28-day treatment with BPA, which was partially reversed by overexpression of PTEN after transfecting with Ad-PTEN-eGFP. Our data support an important role of the PTEN/PI3K/AKT axis in modulating premature activation effects of BPA in vivo. The repression of PTEN induced by BPA was also revealed in other studies. 26 Xiaoting Li et al. found that BPA downregulated the expression of PTEN in MCF-7 cells, associated with expression of miR-19a and miR-19b. 26 MiR-19 may be involved in the mechanism of downregulation of PTEN by BPA; however, it is still unclear how BPA decreases the expression of PTEN in human and animal ovaries. This will be explored in future projects.
In our study, after transfection with adenovirus vector, the expression of PTEN-eGFP was detected in ovarian follicles such as primordial, primary follicles, and granulosa cells in mouse ovaries. The overexpression of PTEN inhibited premature activation of primordial follicles caused by BPA and partly reversed BPA-mediated excessive activation on the PI3K/AKT signaling pathway. We do not know whether this process had occurred in oocytes and granulose cells in a similar manner or not, and the specific effect of BPA on the above two cells, respectively. It is confirmed that granulosa cells played an important role in ovarian physiology by producing steroid hormones and other factors during the follicular development. 41 Besides that, ovarian steroidogenesis occurs according to the 2-cell/2-gonadotropin theory. 42 Accumulated studies demonstrate BPA may disrupt granulosa cells’ function and have a significant impact on folliculogenesis. Xu et al. 43 found that murine ovarian granulosa cells cultured with 100 fM to 100 µM BPA for 24 to 72 hours had decreased cell viability, increased cell apoptosis, and increased follicular atresia. It is also reported that higher BPA levels increased E2 synthesized in vivo 33 and affected folliculogenesis process, 44,45 as well as short exposure to supraphysiological concentrations of BPA on human membrane granulosa cells altered a series-associated gene expression. 46
Using selective PTEN deletion experiment, the role of PTEN on oocytes and granulosa cells has been investigated separately. When PTEN knock out mice oocytes, primordial follicles had been largely activated before sexual maturity in these mice. 23 When PTEN expression was selectively disrupted in granulosa cells, the PTEN mutant mice ovulated more oocytes and produced moderately more pups than the control mice. 47 These findings provide the novel evidence that PTEN plays very important regulation role in suppression the overovulation and depletion of primordial follicle pool by controlling the survival and activation of primordial follicles, as well as the proliferation and differentiation of granulosa cells through the PTEN-PI3K signaling networks. In our study, contrarily, we conducted the PTEN overexpression in mouse ovaries and found the overexpression of PTEN inhibited premature activation of primordial follicles induced by BPA. These results are consistent with the above, although we use the reverse method.
In hormone assays of this study, T levels of the group administered 10 mg/kg BPA were significantly higher than in other groups. Notably, a similar tendency was reported in rat ovarian theca-interstitial cells whereby BPA increased T synthesis at concentrations of 10−7 to 10−4 M after a 72-hour incubation period. 44 It can be explained that BPA alters androgen metabolism in the liver and, acting as a potent sex hormone–binding globulin binder, displaces androgens resulting in increased levels of serum free androgens. 48 However, BPA did not change the levels of E2 and P in this study; it might imply that during the transition of primordial to primary follicles, impact on the 2 hormones is very small.
In conclusion, this study demonstrates that BPA can initiate excessive premature activation of primordial follicles in the mature mouse ovaries via the PTEN/PI3K /AKT signaling pathway by downregulating PTEN expression in vivo. Our results improve the understanding of both the effect of BPA in occurrence of POI and molecular mechanisms underlying activation of primordial follicle pool, thus providing insight for POI treatment and theoretical basis for reducing the risk of POI.
Supplemental Material
Supplemental Material, Supplementary_data1_Flow_Chart - Bisphenol A Initiates Excessive Premature Activation of Primordial Follicles in Mouse Ovaries via the PTEN Signaling Pathway
Supplemental Material, Supplementary_data1_Flow_Chart for Bisphenol A Initiates Excessive Premature Activation of Primordial Follicles in Mouse Ovaries via the PTEN Signaling Pathway by Ying Hu, Dong-zhi Yuan, Yi Wu, Lin-lin Yu, Liang-zhi Xu, Li-min Yue, Lin Liu, Wen-ming Xu, Xiao-yong Qiao, Ru-jun Zeng, Zhi-lan Yang, Wei-yao Yin, Ya-xian Ma, and Ying Nie in Reproductive Sciences
Supplemental Material
Supplemental Material, Supplementary_data2 - Bisphenol A Initiates Excessive Premature Activation of Primordial Follicles in Mouse Ovaries via the PTEN Signaling Pathway
Supplemental Material, Supplementary_data2 for Bisphenol A Initiates Excessive Premature Activation of Primordial Follicles in Mouse Ovaries via the PTEN Signaling Pathway by Ying Hu, Dong-zhi Yuan, Yi Wu, Lin-lin Yu, Liang-zhi Xu, Li-min Yue, Lin Liu, Wen-ming Xu, Xiao-yong Qiao, Ru-jun Zeng, Zhi-lan Yang, Wei-yao Yin, Ya-xian Ma, and Ying Nie in Reproductive Sciences
Supplemental Material
Supplemental Material, Supplementary_data3 - Bisphenol A Initiates Excessive Premature Activation of Primordial Follicles in Mouse Ovaries via the PTEN Signaling Pathway
Supplemental Material, Supplementary_data3 for Bisphenol A Initiates Excessive Premature Activation of Primordial Follicles in Mouse Ovaries via the PTEN Signaling Pathway by Ying Hu, Dong-zhi Yuan, Yi Wu, Lin-lin Yu, Liang-zhi Xu, Li-min Yue, Lin Liu, Wen-ming Xu, Xiao-yong Qiao, Ru-jun Zeng, Zhi-lan Yang, Wei-yao Yin, Ya-xian Ma, and Ying Nie in Reproductive Sciences
Footnotes
Authors’ Note
Y.H., D.Z.Y., and Y.W. planned and performed the majority of the experiments and data analysis and wrote the first draft of the manuscript. L.L.Y. performed the immunofluorescence assays. L.Z.X. contributed to the experimental planning, to data analysis and discussion, and critically reviewed the manuscript. L.M.Y., L.L., and W.M.X. critically reviewed the manuscript. X.Y.Q. helped in the Western blotting assays. R.J.Z. and Z.L.Y. participated in the RT-PCR assays. W.Y.Y., Y.X.M., and Y.N. took care of the CD-1 mice, did the oral gavage job, and helped in ovary collection. Y.H. and D.Z.Y. contributed equally to this work.
Acknowledgment
The authors would like to thank the expert technical assistance of Mr Xiang Zheng and Mr Jinhu Zhang in West China School of Preclinical and Forensic Medicine, Sichuan University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation for Youth (China, NO. 21507094) and Clinical Research Fund of Sichuan University West China Second University Hospital in 2016 (NO.KL008).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
