Abstract
Vulvar and vaginal atrophy (VVA) is a chronic, progressive medical condition prevalent among postmenopausal women, which produces symptoms such as dyspareunia, vaginal dryness, and vaginal irritation. Currently, the only prescription options are systemic and vaginal estrogen therapies that may be limited by concerns about long-term safety and breast cancer risk. Ospemifene is a tissue-selective estrogen agonist/antagonist (a selective estrogen receptor modulator) recently approved by the US Food and Drug Administration for treatment of dyspareunia, a symptom of VVA, due to menopause. Ospemifene, the first nonestrogen oral treatment for this indication, may provide an alternative to treatment with estrogen. Animal models with ospemifene suggest an inhibitory effect on growth of malignant breast tissue, but animal data cannot necessarily be extrapolated to humans. Clinical trials, including 3 long-term studies assessing the overall safety of ospemifene, support that ospemifene is generally well tolerated, with beneficial effects on the vagina, neutral effects on the breast, and minimal effects on the endometrium.
Introduction
Estrogen agonists/antagonists, also known as selective estrogen receptor modulators (SERMs), have been developed for a range of established clinical applications, including the treatment of breast cancer, osteoporosis, and infertility. 1 –3 Recently, SERMs have undergone clinical development for the treatment of vulvar and vaginal atrophy (VVA), a chronic medical condition that affects approximately 50% of postmenopausal women. VVA often produces symptoms such as dyspareunia, vaginal dryness, and vaginal irritation, any of which may negatively impact sexual and emotional health. 4 –7 Ospemifene is a new tissue-selective estrogen agonist/antagonist recently approved by the US Food and Drug Administration (FDA) for the treatment of moderate to severe dyspareunia, a common symptom of VVA in postmenopausal women. 8 –15 It is the first oral non-estrogen prescription alternative to estrogen therapies for the treatment of VVA.
Prior to the approval of ospemifene, the only prescription therapies available to women with VVA were systemically and vaginally administered estrogen treatments. 4,16 However, specific findings of the Women’s Health Initiative study raised concern about the safety of systemic estrogen therapy. 1,4,5,17 These concerns may have limited patient and physician acceptance of estrogen. Even with vaginal estrogen, safety concerns such as breast cancer risk can be a barrier to treatment. 18 Unless additional menopausal symptoms are present (beyond those associated with VVA), local vaginal estrogen treatment options, if effective, may be preferred and recommended over systemic estrogen. 19,20
Ideally, a treatment for VVA would have beneficial effects on the vagina as well as positive effects on bone and brain tissue, with neutral or antagonistic effects on the breast and the endometrium. As a therapeutic class, SERMs are characterized by their differential effects (estrogen agonistic, estrogen antagonistic, or neutral) in various tissues, and the molecular and clinical profile of each SERM is distinct.
The breast is an estrogen-responsive tissue in which the estrogen receptors (ERs; ERα and ERβ) are expressed in a distinct pattern. Both the receptors have a ligand-binding domain that can bind estrogen or an SERM. 21 –24 After binding, the receptors dimerize and recruit other factors involved in controlling the expression of estrogen-regulated genes. 21 –24 A critical factor controlling the agonistic or antagonistic tissue effects of ERs is the availability of coinducers and corepressors in each tissue type as well as their binding to the ERs. 22,23,25 Each SERM induces unique conformational changes in ERs, which can affect the relative binding of coactivators and corepressors. The tissue-specific activity of an individual SERM will depend on the structure of that SERM, the availability of cofactors in each cell type, and the effects of the SERM on the binding of cofactors to the ERs (Figure 1). The complexity of this regulatory system is just beginning to be fully appreciated, with more than 285 nuclear receptor coregulators having been identified thus far. 23 Although the specific coactivators and coinducers in various tissues have not been identified for ospemifene, some information is available regarding its effects on certain tissues (Table 1).
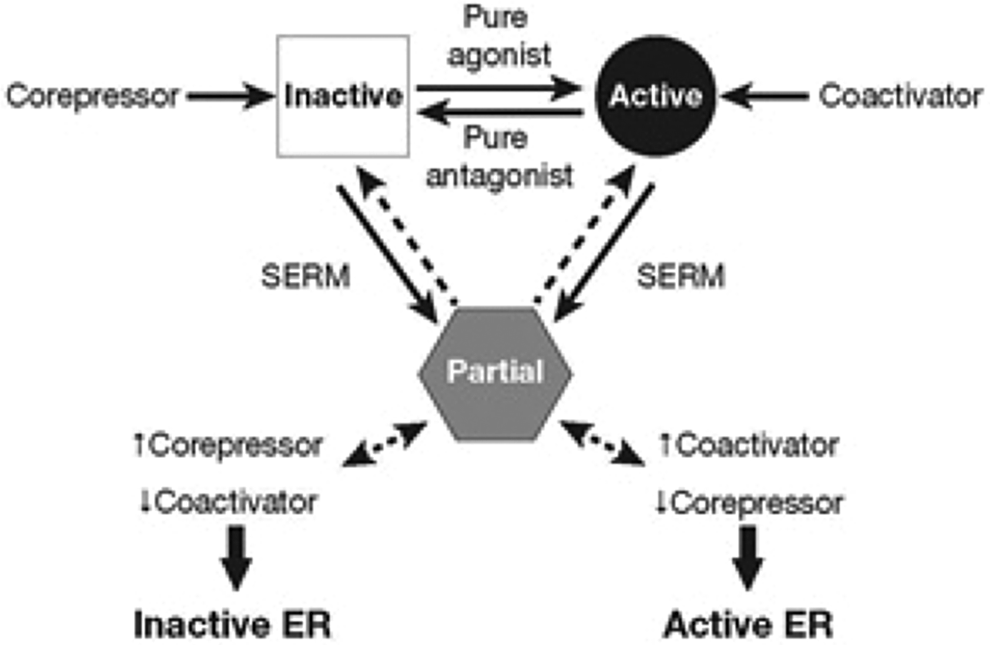
Tissue-specific agonist/antagonist effects of selective estrogen receptor modulators (SERMs) are determined by coactivators and corepressors. Adapted with permission from Jordan et al. 23 ER, estrogen receptor.
Ospemifene Activity Profile.a

Abbreviations: +, agonist effect; −, antagonist effect; 0, no or minimal effect.
a Refers to clinical data, unless otherwise indicated.
b Refers to preclinical data.
c Cardiovascular outcomes were not assessed, but effects on cardiometabolic parameters suggest a neutral overall impact.
In addition, recent research suggests that the ER can affect the activity of other proteins involved in gene regulation through mechanisms that do not require the ER to bind DNA (nongenomic). Some of these activities may control certain cell–cell interactions through the activation of signal transduction pathways. The significance of these nongenomic activities of ERs is just beginning to be investigated. Likewise, it is unclear how SERMs affect nongenomic ER activities. 24,26
The ERβ is the predominantly expressed ER in the adult mammary gland and may provide a protective mechanism in the breast by suppressing proliferation induced by ERα in epithelial cells of the breast. 27 The ERα is expressed only in epithelial cells that line the ducts and lobules, whereas ERβ is expressed in these cells as well as myoepithelial cells and some periductal stromal fibroblast cells. The exact mechanisms by which the ER subtypes control cell transformation and proliferation in the breast have not been established, but it has been speculated that ERβ expression in the myoepithelium may provide a barrier between proliferating ERα epithelial cells and the basement membrane. Additional data suggest that overexpression of ERβ in MCF-7 human breast cancer cells can suppress the proliferation of these cells—and in another cell line, T47D, expression of ERβ suppressed 1000 of 1400 genes induced by ERα. 28 Given the complexity of the interactions of SERMs in each target tissue type, predicting clinical implications may be difficult; however, it is essential to try to understand the effects of each SERM on tissues of relevance such as the breast. 26,29
In three phase 3 clinical trials, ospemifene has been demonstrated to be effective and generally well tolerated in the treatment of dyspareunia, a symptom of moderate to severe VVA in postmenopausal women. 13,30 –32 In addition, many preclinical evaluations of ospemifene have been conducted to determine its activity in breast cancer tissue (Table 2). 8 –10,14,33 Although breast cancer models differ from healthy breast tissue, the preclinical studies are a starting point for further research. In clinical studies evaluating long-term safety of ospemifene, mammograms (eg, at week 52 of treatment) were included in the overall safety assessments. 15,32
Overview of Preclinical Data for Ospemifene in the Breast.
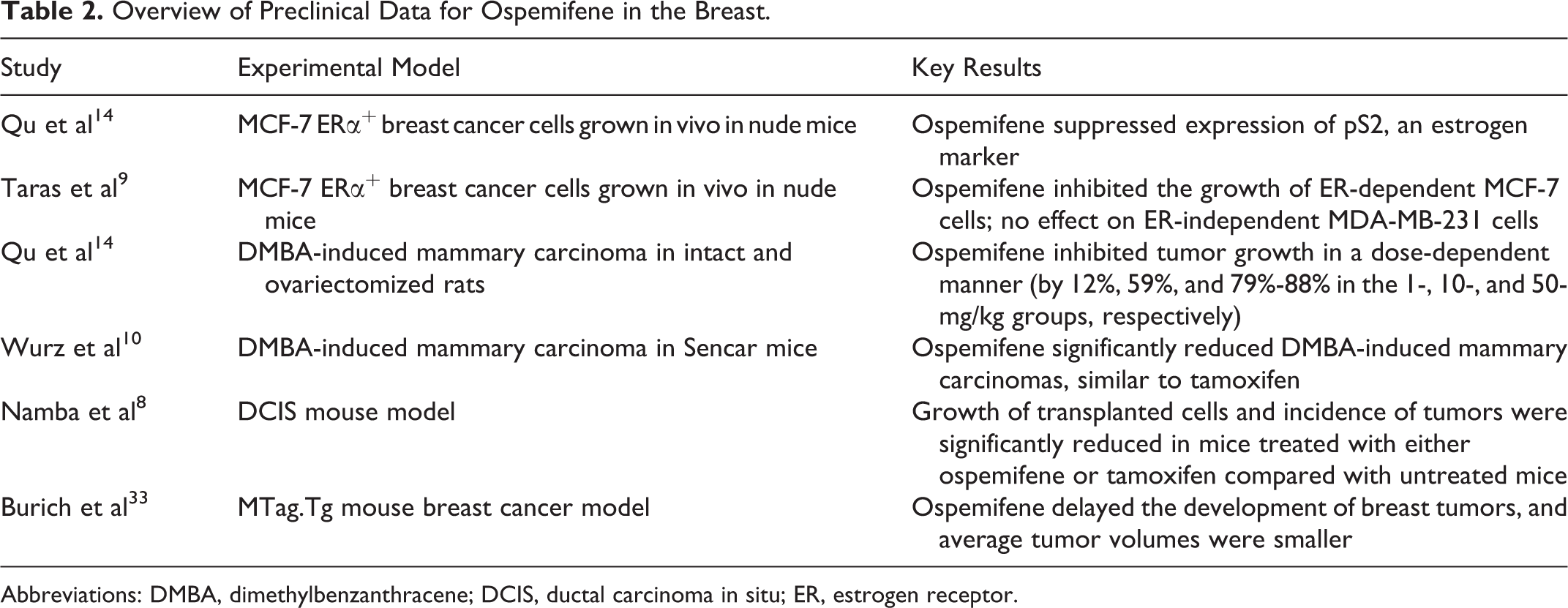
Abbreviations: DMBA, dimethylbenzanthracene; DCIS, ductal carcinoma in situ; ER, estrogen receptor.
The objectives of the present review are to summarize the published breast-related preclinical data (breast cancer models) for SERMs and describe the available clinical safety data on the effects of ospemifene in the breast, based on palpation and mammography. Articles included in this review were initially identified through the PubMed database (search terms “ospemifene” OR “FC1271a”) and then screened for relevance (criteria for inclusion were English-language articles on preclinical breast data and/or human clinical safety data). The search was last updated in January 2013. A supplemental EMBASE search identified clinical abstract data that had not yet been published.
Ospemifene Pharmacology
Like tamoxifen, ospemifene is a triphenylethylene (Figure 2) and thus has the estrogen agonist/antagonist tissue-selective effects of other SERMs. 1 Ospemifene has a half-life of 26 hours and undergoes metabolism by cytochrome P450 enzymes to 4-hydroxyospemifene (major metabolite). 11,12,34
Chemical structures of ospemifene, tamoxifen, and estradiol.
Summary of Ospemifene Preclinical Breast Data: In Vitro and In Vivo Studies
Ospemifene binding to ERs
In competition studies with 17β-[3H]estradiol, the reported IC50 values of ospemifene for ERα and ERβ were found to be 827 nm and 1633 nm, respectively. Ospemifene binds to ERα slightly more than to ERβ, with relative binding affinities of 0.8% and 0.6%, respectively, compared to estradiol. This is similar to the binding affinities of toremifene and tamoxifen to the ERs. 14
In vitro research on MCF-7 cells
The MCF-7 ERα-positive and MDA-MB-231 ER-negative breast cancer cells were utilized to show that the effects of ospemifene on the breast are ER dependent. 9 In this in vitro study, ospemifene inhibited the growth of MCF-7 cells and the expression of a gene that is known to be induced by estrogen called pS2. Ospemifene had no effect on the MDA-MB-231 cells that lack the ERs. Although the inhibitory effect of ospemifene on the growth of MCF-7 cells in vitro was not as great as that of toremifene and raloxifene, the effect of ospemifene was shown to be dose dependent. At ospemifene concentrations of 0.1 , 1.0 , 5.0 , and 10.0 μmol/L, the cells grew at rates of 95.4% ± 7.7%, 79.6% ± 9.8%, 66.2% ± 12.5%, and 56.4% ± 11.1%, respectively, of the nontreated cells. 9
In vivo research in rats and mice
The MCF-7 cells have provided a model system for studying in vivo tumor growth in mice and rats. 35 When the MCF-7 breast cancer cells were studied as xenografts in athymic, ovariectomized mice, ospemifene inhibited the growth of ER-dependent MCF-7 cells in vivo but had no effect on the growth of ER-independent MDA-MB-231 cells. 9
In a study of nude mice by Qu et al, 14 ospemifene successfully suppressed the expression of the pS2 estrogen marker in human MCF-7 ER-positive cells despite the presence of estrogen. However, in the study by Taras et al, 9 pS2 was suppressed in vitro but not in vivo in athymic, ovariectomized mice.
Another model system used to investigate breast cancer in animals is the chemical induction of mammary tumors with dimethylbenzanthracene (DMBA). In rats, the inhibition of DMBA-induced mammary tumors by ospemifene was shown to be dose dependent. The 1-mg/kg dose used in these animal studies approximates the physiologic human dose of 60 mg (a 60-mg dose in a 70-kg person is equivalent to 0.86 mg/kg), whereas the 10- and 50-mg/kg doses would represent supraphysiologic human doses. 19 Based on 100% growth of tumors in the untreated rats, there was 88%, 41%, and 21% tumor growth in the groups treated with the 1-, 10-, and 50-mg/kg doses of ospemifene, respectively. In fact, at the highest dose of ospemifene (50 mg/kg), 4% of the tumors disappeared. 14 When the number of tumors was measured 6 weeks after treatment with ospemifene had been stopped, the rats treated with 50 mg/kg ospemifene had only 35% of the tumor burden of the untreated group.
Chemically induced breast cancer in mice was a model used by Wurz et al 10 to study the effects of ospemifene on breast cells. Mammary carcinomas were DMBA induced in female Sencar mice, with concomitant treatment with 50 mg/kg of ospemifene, tamoxifen, or raloxifene for 37 weeks each. Ospemifene significantly reduced DMBA-induced mammary carcinomas in these mice compared with the untreated DMBA-induced control mice (P = .003). Tumor reduction over 37 weeks was similar for ospemifene and tamoxifen treatment in these mice, but raloxifene had no significant suppressive effect on tumor reduction compared with the controls. Results were similar when these experiments were repeated with ospemifene and tamoxifen over 52 weeks. Both ospemifene and tamoxifen reduced the number of DMBA-induced carcinomas in these mice compared with untreated mice (P = .01 and P = .004, respectively; Figure 3).
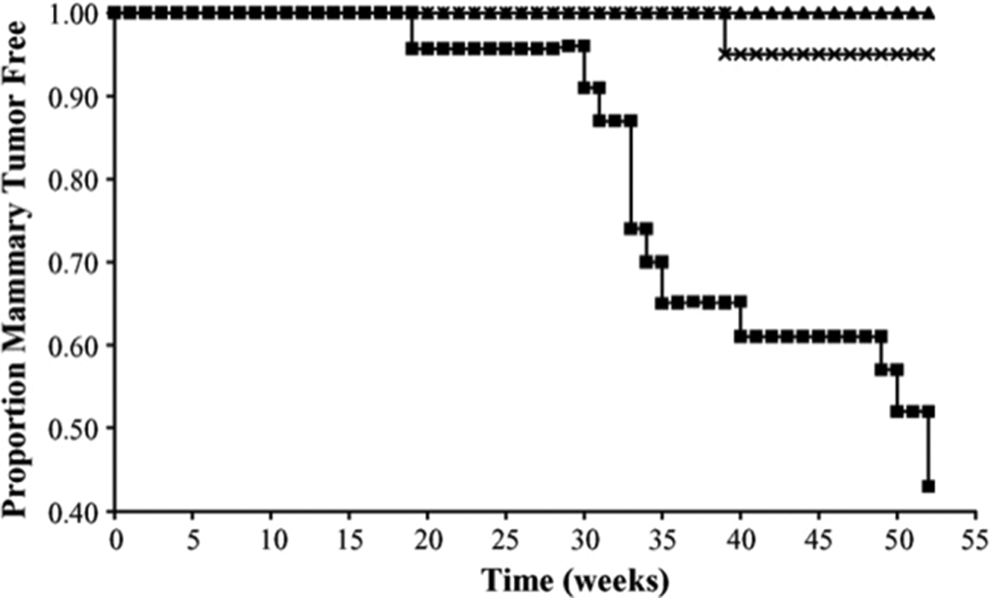
Suppression of dimethylbenzanthracene (DMBA)-induced tumors in mice. Reprinted with permission from Wurz et al. 10
The ductal carcinoma in situ (DCIS) mouse model uses stably transformed mouse cells to induce intraepithelial neoplasia outgrowths in mice. 8 These outgrowths share similarities with human DCIS, including the ability to develop into invasive tumors, thereby providing an excellent model for studying DCIS. In this model, the local mammary lesions are taken from transgenic mice that carry the mouse mammary tumor virus polyomavirus middle T (PyV-mT) gene (MTag.Tg). The transformed cells from these transgenic mice are transplanted into the mammary fat pads of a wild-type mouse of the same genetic background as the transgenic mice.
Either ospemifene or tamoxifen was administered to mice before transplantation of the transformed cells in order to study the ability of these treatments to prevent tumor growth and outgrowth in this model system. 8 Compared with control mice, those treated with ospemifene or tamoxifen had a reduction in the growth of transplanted cells and a lower incidence of tumors (P < .01) over 3 or 10 weeks of posttransplantation. Cell proliferation was reduced significantly by ospemifene (P < .05) and tamoxifen (P < .01) as measured by the proliferation marker Ki-67. 8
In a recent study, Burich et al 33 assessed the impact of ospemifene on the development of mammary tumors directly in immunologically intact mice carrying the PyV-mT antigen fusion gene. Female C57BL/6 transgenic mice that were confirmed to carry the PyV-mT antigen were administered ospemifene and 4-hydroxyospemifene (5, 25, or 50 mg/kg) starting at 4 weeks of age. At the time point when the tumors were first measurable, the average tumor volumes were significantly larger in control mice than in the mice that received the ospemifene 50-mg/kg dose (P < .05; Figure 4). This group of ospemifene-treated mice also had a significantly higher survival rate over 150 days (P =.0085 vs controls). In another set of experiments, MTag.Tg mice were not treated with ospemifene until they were 16 weeks old, which allowed more time for the development of tumors. Survival over 125 days was significantly longer for mice that received the 50-mg/kg dose of ospemifene (P = .0143 vs controls).
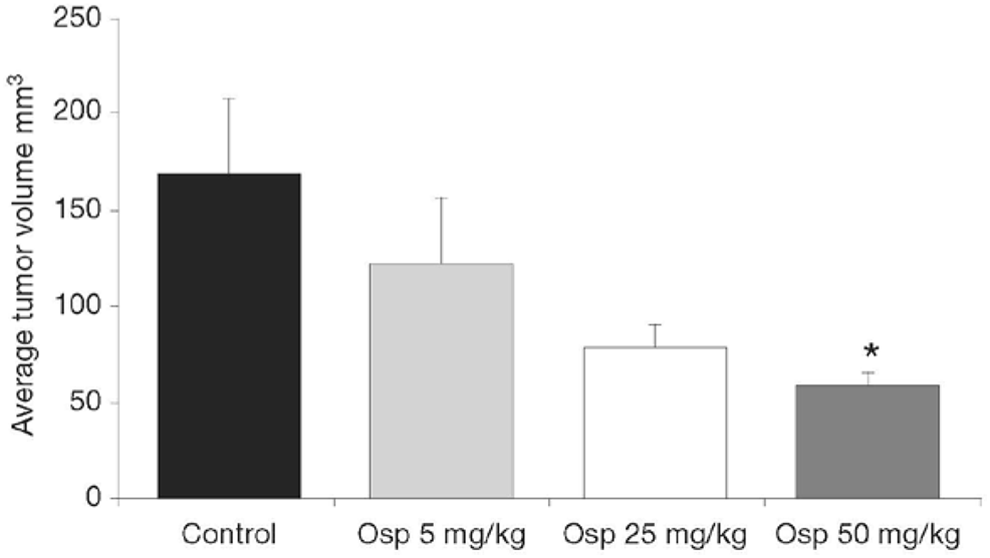
First measurable tumor volumes in MTag.Tg mice. *P <.05 compared with control. Reprinted with permission from Burich et al. 33 Osp, ospemifene.
Clinical Studies of Ospemifene
Given the promising preclinical data in vaginal epithelial tissues, as well as favorable preclinical safety data (including the breast data summarized above), it was determined that ospemifene studies in humans were warranted. 11,12,34 A brief overview of safety and tolerability data from ospemifene clinical studies, particularly breast and endometrial safety, is provided below.
Breast safety
Three clinical studies to evaluate the overall long-term safety of ospemifene included mammograms to assess breast safety. A 6:1 randomized, double-blind, placebo-controlled study evaluated the safety and efficacy of ospemifene 60 mg over 52 weeks in 426 women with VVA and an intact uterus (ospemifene, n = 363; placebo, n = 63). 31,32,36 Breast safety was assessed by palpation at screening, then at weeks 12, 26, 52, and at the posttreatment follow-up visit (≥4 weeks after treatment completion). Mammography was conducted at screening and week 52 (or end of treatment). No clinically significant changes from baseline to week 52 were noted on breast examination or mammography for any study participant at any time point. No cases of breast cancer occurred during this study.
Two long-term extension studies of a 12-week, randomized, double-blind, placebo-controlled clinical study were performed to evaluate the safety of ospemifene once daily in postmenopausal women with moderate to severe symptoms of VVA. 15 The initial 12-week study evaluated the efficacy and tolerability of ospemifene 30 mg/d and 60 mg/d in 826 women (ospemifene 30 mg, n = 282; ospemifene 60 mg, n = 276; placebo, n = 268). 13 After completing the initial study, women with a uterus were eligible to continue blinded treatment for a total of 52 weeks (12 weeks in the initial study plus 40 weeks in the safety extension [N = 180]). Safety assessments of the breast included mammograms and palpation on physical examination performed at week 52 or study discontinuation. 15
The majority of breast palpations at week 52 had normal results, and the findings were similar for all the study groups (normal results: 100% for placebo, 100% for ospemifene 30 mg/d, and 98.3% for ospemifene 60 mg/d). 15 Furthermore, results of mammograms performed at week 52 were normal for all the subjects in all study groups, with the exception of 1 subject in the ospemifene 60-mg/d group who had an abnormal mammogram finding that subsequently resolved during follow-up after completion of the study. Only 2 subjects experienced a breast-related serious treatment-emergent adverse event (TEAE; 1 event in each): 1 (2%) subject in the placebo group had breast cancer in situ, and 1 (1.4%) subject in the ospemifene 60-mg group had breast prosthesis implantation surgery. Six subjects experienced a nonserious breast-related TEAE: 1 (2.0%) subject in the placebo group had a breast cyst; 2 (3.2%) subjects in the ospemifene 30-mg/d group had evidence of a breast mass; and 1 (4.3%) each in the ospemifene 60-mg/d group had a breast mass, breast microcalcification, or an abnormal mammogram (as noted previously). All 6 nonserious breast-related TEAEs were mild in severity.
Similar breast safety results were achieved in a second long-term, open-label safety extension of the same initial 12-week study. In this assessment of women without a uterus treated with ospemifene 60 mg/d for 52 weeks, no clinically significant changes in overall breast safety were observed at week 26 or week 52 (N = 301; data on file). All breast-related TEAEs were considered mild or moderate in severity. One subject in the ospemifene group had a report of a breast-related TEAE (breast mass), assessed as mild and unlikely related to the study drug, that was ongoing at the end of the study and at the 4-week follow-up visit. Subsequent mammograms during routine care visits after study completion were reported to be normal.
Endometrial safety
In addition to the breast safety documented in these clinical trials, ospemifene was generally well tolerated, with no clinically significant endometrial effects (based on transvaginal ultrasonography and biopsy). 15,32,36 In the 52-week safety and efficacy study, endometrial thickness increased by 0.68 mm from baseline to week 52. On biopsy, there were 3 cases of active proliferation, 1 of which was diagnosed as simple hyperplasia without atypia. 36 In the 40-week safety extension study of women with an intact uterus, ospemifene 30 mg/d and 60 mg/d resulted in an increase of 0.68 mm and 1.14 mm, respectively, in mean endometrial thickness by week 52. The majority of endometrial biopsy samples were atrophic or inactive. Weakly proliferative findings were noted both at baseline and at week 52 in 1 subject who received ospemifene 30 mg and in 1 who received ospemifene 60 mg; there were no cases of endometrial hyperplasia or carcinoma. Overall, the incidence (per thousand women) of endometrial effects with ospemifene was 60.1 (thickening of ≥5 mm), 86.1 (any type of proliferative endometrium), and 5.9 (uterine polyps) versus 21.2, 13.3, and 1.8 for placebo, respectively; given that ospemifene has estrogen agonistic effects on the endometrium, a black box warning regarding the potential for endometrial stimulation is included in the labeling. 37
Additional safety/tolerability data
As expected with an SERM, hot flushes were the most commonly reported adverse effects; however, the rate of discontinuation due to hot flushes was low (≤1%) in all the studies. Overall, the frequency of hot flushes attributed to ospemifene 60 mg in randomized clinical trials is 7.5%. 37 Only 1 case of venous thromboembolism was noted in long-term safety studies. 15,32,36 However, overall, the incidence rates per thousand women of thromboembolic events with ospemifene 60 mg in clinical trials (duration of treatment of up to 15 months) were 0.72 (thromboembolic stroke; 1 case), 1.45 (hemorrhagic stroke; 2 cases), and 1.45 (deep vein thrombosis [DVT]; 2 cases), whereas for placebo these rates were 1.04 (1 case), 0 (no cases), and 1.04 (1 case), respectively, and a black box warning on DVT and stroke is included in the labeling. 37
Summary
Ospemifene has been recently approved by the FDA for the treatment of postmenopausal women with moderate to severe dyspareunia, a prevalent symptom of VVA. A chronic and progressive condition, VVA also elicits other distressing symptoms, such as vaginal dryness and irritation. Phase 3 trials have established that non-estrogen ospemifene addresses the underlying physiological changes that occur in VVA by increasing the proportion of superficial cells in the vaginal epithelium, reducing the number of parabasal cells, and decreasing vaginal pH, which leads to improvement in VVA symptoms, including dyspareunia and vaginal dryness. 13,30 The results of long-term safety extension studies also support that oral ospemifene is well tolerated, with beneficial estrogen agonist effects in the vagina and neutral to minimal effects in the breast and endometrium. 13,15,30 –32
Concerns about long-term safety are potential barriers to estrogen-based therapies for VVA. Thus, a nonestrogen VVA treatment alternative would be a welcome option for women who cannot or will not use estrogen therapy. 38
As detailed in this review, investigations in animal models suggest that ospemifene may have an inhibitory effect on the growth of malignant breast tissue. 8 –10,14,33 However, any extrapolation of preclinical animal models to humans must be done with caution. As a class, SERMs appear to have antiestrogenic or neutral effects on the breast. The antiestrogenic effects of tamoxifen and raloxifene are well established in clinical studies, and these agents are indicated for the treatment (tamoxifen only) or prevention (tamoxifen and raloxifene) of certain breast cancers. 39 –42 Among investigational SERMs, antiestrogenic effects have been reported for lasofoxifene (clinical) and bazedoxifene (preclinical). 43,44
The available clinical data for ospemifene, including long-term (52-week) safety evaluations in postmenopausal women with VVA, support a neutral effect in the breast. It is important to note that human data on the breast effects of ospemifene are limited, and therefore a definitive statement on the long-term effects in human breast tissue cannot be made at this time. Future areas of investigation for ER agonist/antagonist compounds in the breast include effects on proliferation markers such as Ki-67 in women and gene expression in human breast tissue.
Footnotes
Acknowledgments
The author acknowledges Rosemary Reinke, PhD, Kulvinder K. Singh, PharmD, and The Medicine Group for editorial assistance in the development of this manuscript.
Author’s Note
The author had full control over content, material, writing, and editing, and takes full responsibility.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Sarah L. Berga has consulted for Shionogi Inc., Pfizer, Teva, Watson, Noven, AHC Media, LLC, and Agile Therapeutics. She serves on the editorial board of the American Journal of Obstetrics and Gynecology, the Journal of Clinical Endocrinology and Infertility, and Menopause.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development of this manuscript was funded by Shionogi Inc. The manuscript discusses ospemifene, which is a product of Shionogi Inc.
