Abstract
Objective:
Continuous glucose monitoring (CGM) is increasingly applied in populations without diabetes, yet existing reference ranges are largely derived from middle-aged or older adults. This study characterized CGM metrics in young adults without diabetes and examined variation by sex, age, body mass index (BMI), and physical activity (PA).
Method:
Participants wore an unmasked Dexcom G7 CGM for up to 10 days under free-living conditions. Glycemic metrics were derived using the iglu R package and summarized as median [IQR]. Associations with sex, age, BMI, and PA were evaluated using Wilcoxon tests, Spearman correlations, and quantile regressions.
Results:
A total of 105 participants (age = 21 years [range: 18-26], BMI 23 kg/m2 [21-25]; 72% female; 72% non-White) provided ≥48hr of CGM data. Compared with females, males had higher mean sensor glucose (110 [103-119] vs 104 [99-108] mg/dL; P < .01), eA1c (5.4[5.2-5.8] vs 5.2[5.1-5.4]; P < .01), area under the curve (110[102-119] vs 103[99-108]; P = .01), and daily episodes >140 mg/dL (1.6[1.0-2.6] vs 1.3[0.7-2.0]; P = .03). Age correlated with CV (r = .20, P = .04). BMI was inversely correlated with CV (r = −.35), MAGE (r = −.35), and MODD (r = −.27), all P < .001. Physical activity was modestly associated with reduced glycemic burden.
Conclusion:
CGM revealed sex differences in young adults–males exhibited higher mean glucose and excursions–while both sexes maintained normoglycemic patterns. Age, BMI, and PA were linked to variability indices. Findings provide CGM reference data for young adults and highlight the importance of biological and behavioral factors in glycemic regulation.
Keywords
Introduction
Diabetes affects more than 38 million people in the United States, and another 97 million adults have prediabetes, placing them at elevated risk for type 2 diabetes and related complications. 1 Because diabetes prevalence rises sharply from early to mid-adulthood—about 3.6% in ages 20 to 39 compared with 17.7% in ages 40 to 59—characterizing glycemic patterns in young adults may provide an early window for prevention. 2 Continuous glucose monitoring (CGM), a cornerstone of diabetes care, is increasingly being used in populations without diabetes to establish reference ranges and to study behavioral influences on glucose regulation.3-7
Descriptive studies to date suggest that healthy individuals maintain stable glucose patterns, with early reports showing that adults spend ~91% of the day between 71 and 120 mg/dL and <1% above 140 mg/dL.3-7 Shah et al 7 reported mean glucose of 98 to 99 mg/dL across most age groups and ~96% of values between 70 and 140 mg/dL. More recent large-scale studies confirmed the reliability of CGM benchmarks in mid-to-older adulthood: Keshet et al 3 analyzed >7000 adults aged 40 to 70 years, and Spartano et al 4 reported normative ranges in >1000 community-dwelling adults around age 60.
However, young adults remain underrepresented in prior CGM research, comprising roughly 22% and 19% of participants in the two studies that included this age group.5,7 Furthermore, existing studies often pool across wide age ranges, obscuring developmental differences, and sex-specific benchmarks are rarely reported. For example, Shah et al 7 included a small subgroup of young adults aged 18 to 25 years (n = 29) but did not examine sex differences, while studies such as Keshet et al 3 and Spartano et al 4 did not include young adults in their samples. At the same time, CGM has demonstrated sensitivity to lifestyle- and behavior-related variation in glucose regulation—including effects of physical activity, diet, and stress8,9—factors that are particularly variable in young adulthood.
To aid interpretation, we created a summary table (Table 1) that reports age ranges, sample sizes, study duration, CGM device, and the primary outcomes (TIR, time in range; TBR, time below range; TAR, time above range; CV, coefficient of variation) from existing CGM studies in nondiabetic populations, including those overlapping with the young adult age range comparable with the present cohort. This study addresses these gaps by focusing on young adults aged 18 to 26 years. We aimed to characterize CGM-derived metrics in this population and to examine associations with sex, age, body mass index (BMI), and physical activity. By generating sex-stratified reference values, this work contributes to refining population-level benchmarks and advancing understanding of how biological and behavioral factors shape glycemic profiles during early adulthood.
Summary of Studies Reporting CGM Metrics in Sample Inclusive of Adults Ages 18 to 26 Without Diabetes.
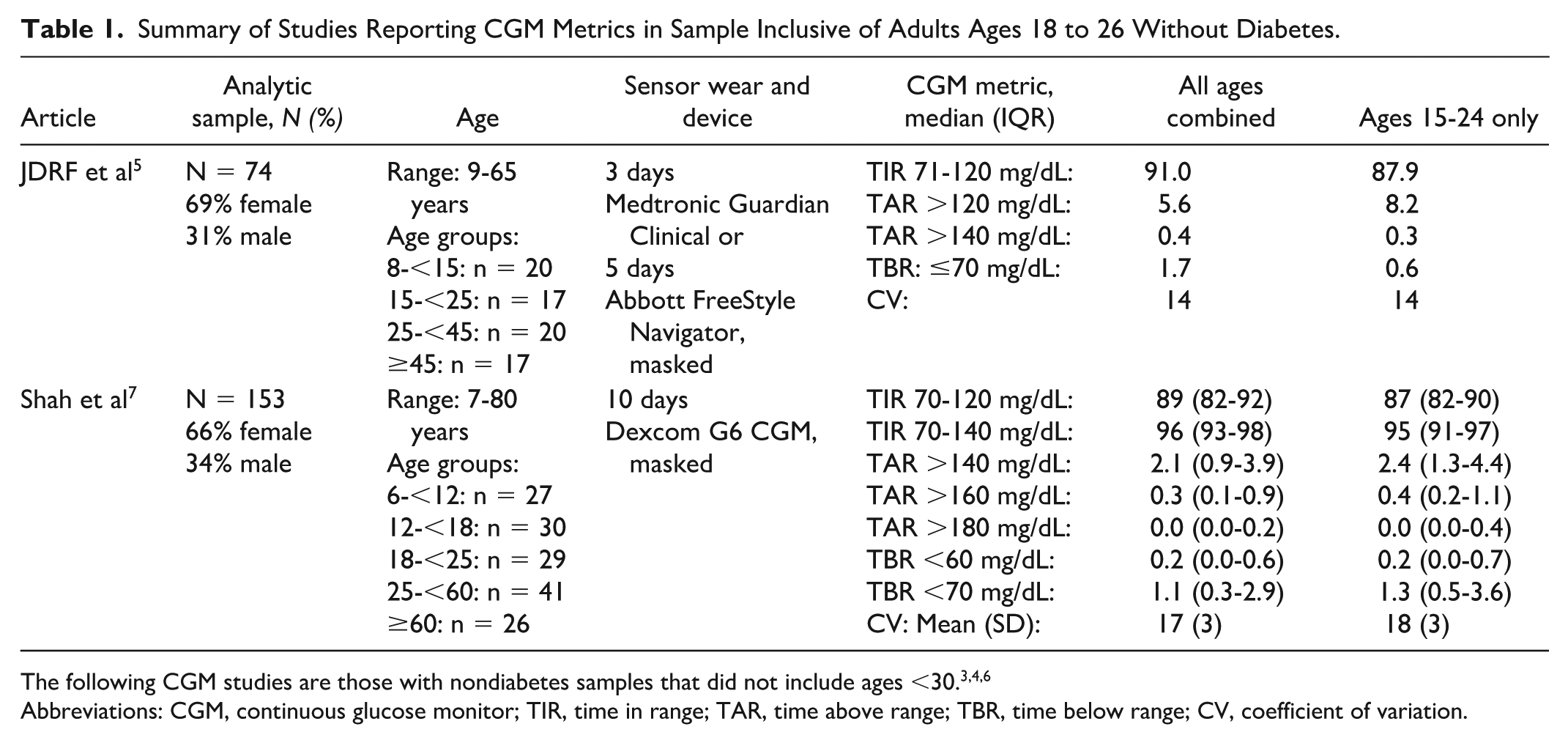
Abbreviations: CGM, continuous glucose monitor; TIR, time in range; TAR, time above range; TBR, time below range; CV, coefficient of variation.
Methods
Study Design
We conducted an observational, continuous-measurement study to characterize CGM-derived glycemic metrics among young adults under free-living conditions. Each participant wore an unmasked sensor for up to 10 consecutive days. No intervention, randomization, or comparator was used. The objective was to generate descriptive data in young adults without a history of diabetes diagnosis.
Participants and Procedures
A total of 105 young adults from a private university were enrolled between October 2023 and April 2025. Recruitment occurred via campus flyers and brief in-class presentations. At baseline, eligibility was confirmed, and written informed consent was obtained. Participants completed demographic and health surveys in REDCap,10,11 received standardized CGM safety/application training, and then applied an unblinded Dexcom G7 sensor with an overpatch under staff supervision. The Dexcom app was installed and paired; data were shared via Dexcom Clarity to enable real-time capture.
Participants were instructed to wear the CGM continuously for up to 10 days during normal activities. Sensors were removed at day 10 or earlier if dislodged. Follow-up occurred within 1 week, with post-monitoring surveys completed in REDCap. Eligibility criteria included age 18 to 26 and current university enrollment. Exclusion criteria were self-reported diabetes diagnosis, glucose-lowering or steroid medications, or chronic conditions likely to affect glycemia. The protocol was approved by the University IRB (UP-23-00751) and adhered to the Declaration of Helsinki.
Measures
Continuous glucose monitoring
Interstitial glucose was measured using the Dexcom G7 system. 12 Participants self-applied the sensor to their upper posterior arm, following the manufacturer’s guidance. An adhesive overlay patch secured the sensor to the skin. Sensors paired with the G7 smartphone app via Bluetooth recorded glucose every five minutes. Participants shared data with the study team through Dexcom Clarity. 13 CGM metrics followed international consensus recommendations 14 and recent large-scale studies. 3 Metrics included mean glucose (mg/dL), estimated A1c (%), glucose management indicator (GMI), area under the curve (AUC; mg/dL), percent time above range (%TOR; >140 mg/dL and >180 mg/dL), percent time in range (%TIR; 63-140 mg/dL), average daily excursions >140 mg/dL, high blood glucose index (HBGI), low blood glucose index (LBGI), coefficient of variation (CV), J-index, mean amplitude of glycemic excursions (MAGE; mg/dL), and mean of daily differences (MODD; mg/dL). GMI was derived from mean glucose using the equation proposed by Bergenstal et al. 15 As recommended, GMI was used here as a CGM-derived summary metric rather than a diagnostic substitute for measured HbA1c. 15 MAGE was computed as the mean absolute difference between consecutive peaks and nadirs exceeding 1 SD of mean glucose. 16 MODD was the mean absolute difference between paired values at the same clock time on consecutive days. 8
Survey assessments
Sociodemographic, biologic, anthropometric, and behavioral data were collected via self-report at baseline. Age (years) and biological sex at birth (male/female) were self-reported. Height (inches) and weight (pounds) were reported and used to calculate body mass index (BMI) with the CDC formula: BMI = (weight [lbs]/height [in2]) × 703. 17 Race (White, African American, American Indian/Alaska Native, Asian, Native Hawaiian/Pacific Islander, Other), ethnicity (Hispanic/Latino/a or not Hispanic/Latino/a), and academic year (freshman, sophomore, junior, senior, graduate) were collected as descriptive variables. Physical activity was measured at baseline and follow-up using the PROMIS eight-item pediatric short form. 18 Because PROMIS pediatric T-scores are not age-normed for adults, scores were applied within-sample to rank relative scores. Diabetes status was captured by self-reported history of diagnosis from a physician. Participants were asked whether they had ever been diagnosed with high blood pressure, Type 1 diabetes, Type 2 diabetes, or abnormal cholesterol, with the option to select “none of the above” or “other” (Table 2).
Sample Characteristics Overall and by Sex, N (%) or Median (Q1, Q3).
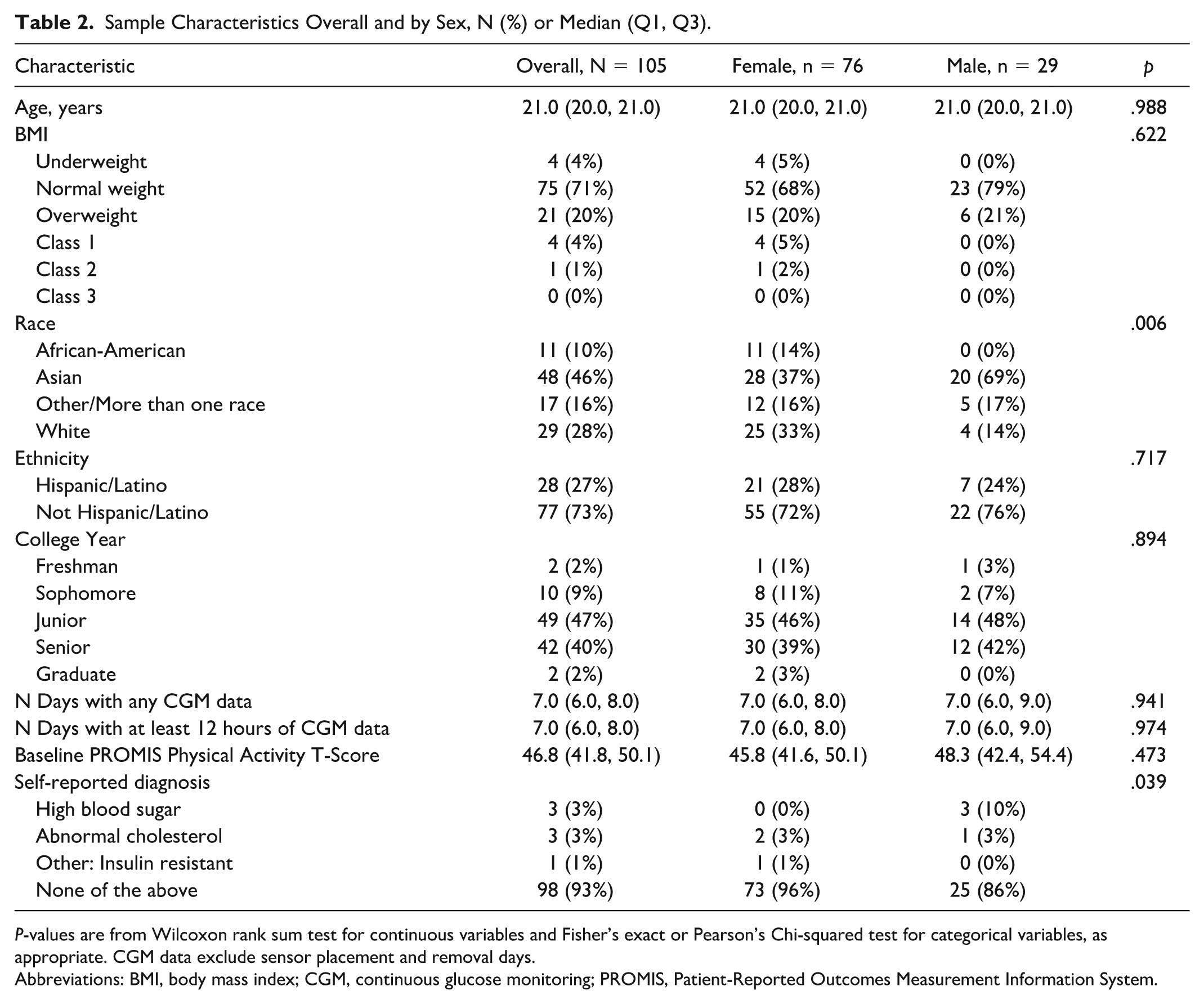
P-values are from Wilcoxon rank sum test for continuous variables and Fisher’s exact or Pearson’s Chi-squared test for categorical variables, as appropriate. CGM data exclude sensor placement and removal days.
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; PROMIS, Patient-Reported Outcomes Measurement Information System.
Statistical Analysis
Baseline characteristics were summarized overall and by sex as median (IQR, interquartile range) for continuous variables and n (%) for categorical variables. Between-sex differences used Wilcoxon rank-sum (continuous) and Fisher’s exact or Chi-squared tests (categorical), as appropriate. To ensure CGM data quality, day 1 of any new sensor wear and the removal day were excluded due to calibration/transition artifacts observed in early wear; analyses were restricted to participants with ≥48 cumulative hours of valid CGM data.
Standardized glycemic variability (GV) metrics were computed in R using iglu. 19 Distributions were inspected via histograms and Shapiro-Wilk tests; because all GV metrics were nonnormal (Figure 1), between-sex comparisons are reported as median (IQR) and tested with Wilcoxon. Associations of age, BMI, and physical activity T-scores with GV metrics were assessed using Spearman correlations. To model associations while accounting for nonnormality, quantile regression (quantreg) was applied for each GV metric with sex, age, BMI, and physical activity (categorical split) as predictors. 20 A two-sided alpha of .05 defined statistical significance.
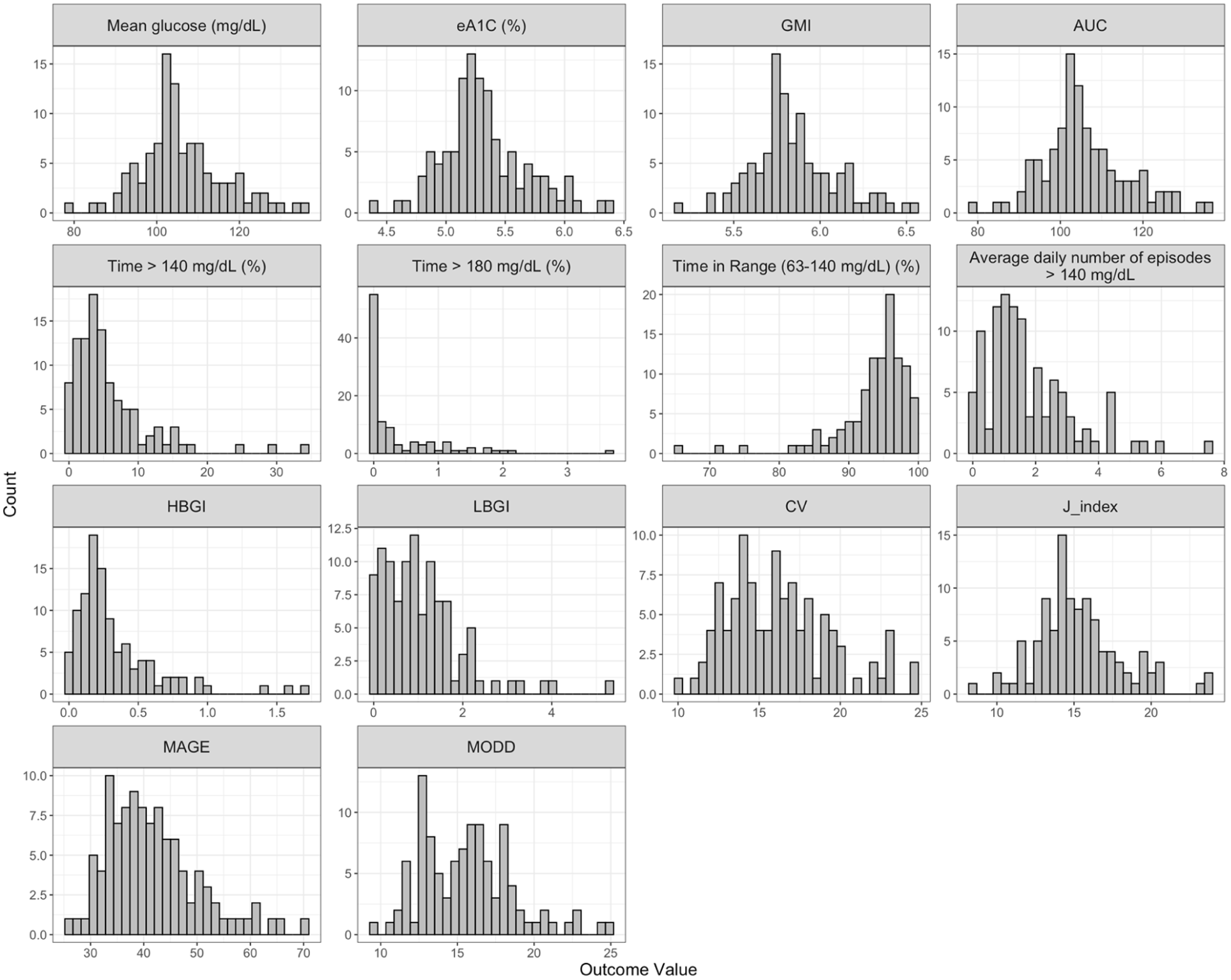
Distribution of CGM metrics. Histograms of CGM metrics in the study cohort; each panel displays the frequency of participants with values in the given range.
Aggregate daily overlay plots were generated by rounding each timepoint to the nearest 10-minute mark and applying LOESS smoothing (Figure 2). Hourly %TOR >140 mg/dL and >180 mg/dL was calculated across the wear period and visualized as heatmaps (Figure 3). Cumulative %TOR >140 mg/dL was computed as the running sum divided by total %TOR across wear time and plotted by day; the denominator was the total %TOR >140 mg/dL for the entire wear time. The cumulative percentage of %TOR >140 mg/dL was calculated by dividing the numerator by the denominator for each day, starting at day 1. The cumulative percentage of %TOR >140 mg/dL was plotted on the y-axis, with the corresponding day number on the x-axis. The simple slope of cumulative percentage of %TOR >140 mg/dL over time was calculated as the median change in cumulative percentage of %TOR >140 mg/dL for every increase in wear day for the full cohort and for each sex group via a linear quantile mixed model (lqmm) to account for the within-subject correlation (Figure 4). 21 Analyses were conducted in R version 4.5.0.
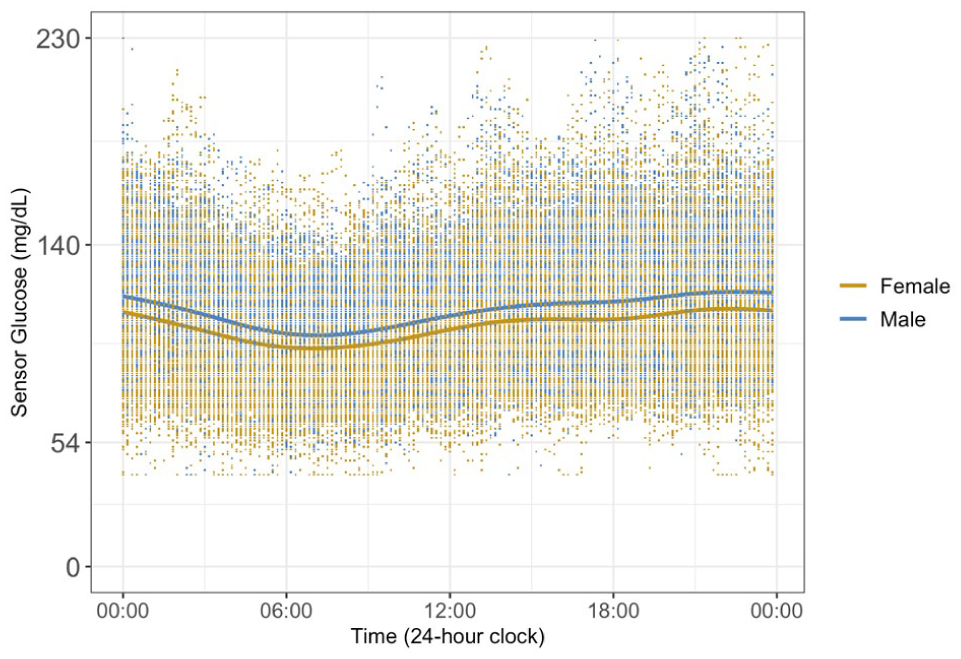
Daily glucose patterns by sex. Average daily glucose profiles with LOESS smoothing aggregated by hour of the day, stratified by sex.
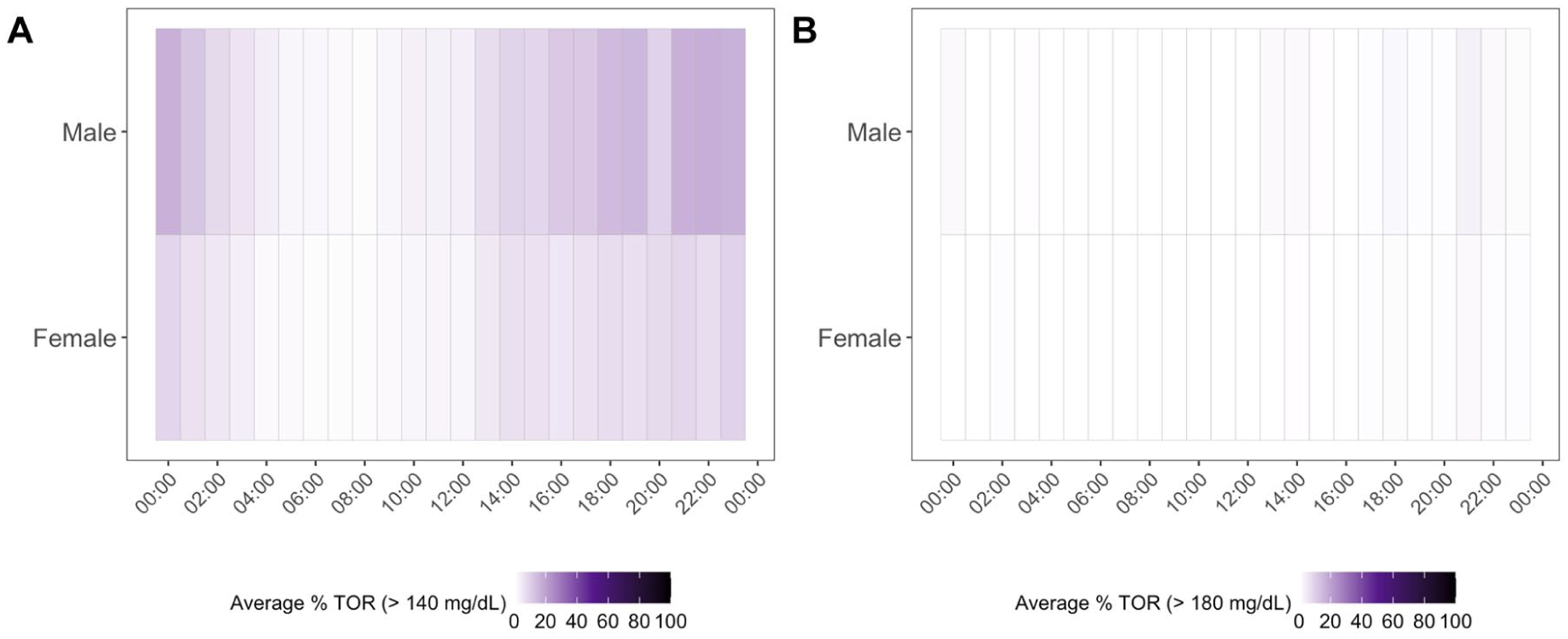
Heatmaps of percent time out of range by sex. Each heatmap cell displays percentages on a scale from 0% to 100%, with higher percentages represented as darker colors. Heatmaps showing the percent of time out of range (%TOR) above (a) 140 mg/dL and (b) 180 mg/dL across each hour of the day, stratified by sex. Darker shading indicates higher %TOR.
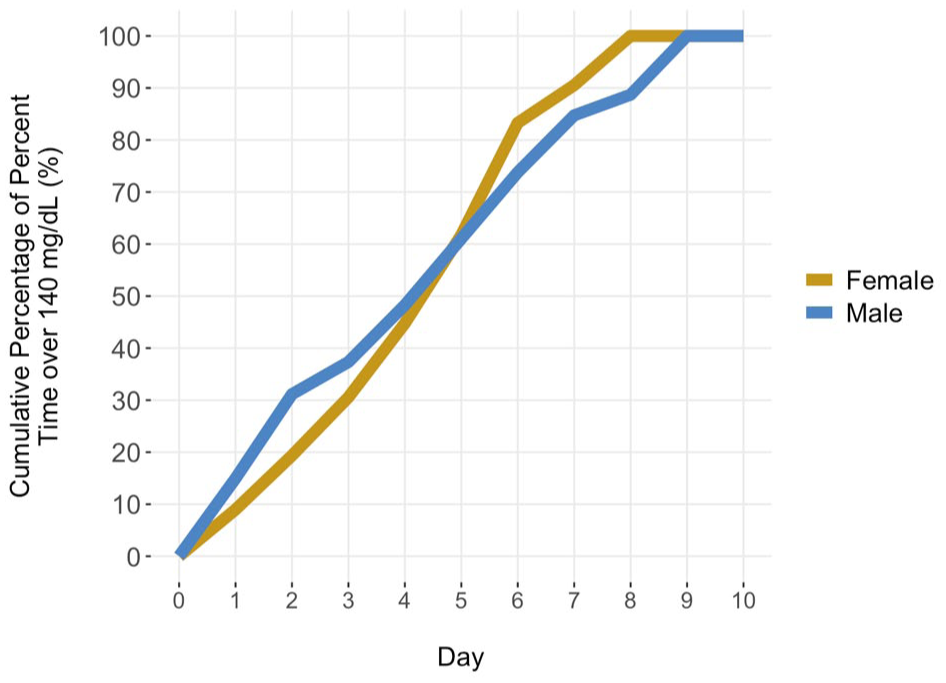
Cumulative percentage of %TOR > 140 mg/dL by sex. Median trajectories are shown as solid lines. The median cumulative exposure increased by 11.1% per day overall (95% CI: 10.1%, 12.0%), with similar rates for females (11.5%, 95% CI: 10.4%, 12.6%) and males (10.6%, 95% CI: 9.2%, 12.0%). Day 0 and the final day were excluded to avoid fixed endpoints (0 and 100%) that bias slope estimation and inflate fit statistics.
Results
Participant Characteristics
A total of 105 participants made up the analytic sample (72% female, n = 76, and 28% male, n = 29) (Table 2). The median age was 21 years (IQR: 20, 21). Most were of normal weight (71%), with 4% classified underweight and 25% as overweight or obese. The median number of days with any CGM data was 7.0 (IQR: 6.0, 8.0); results were similar when restricted to days with ≥12 hours of wear. Baseline physical activity T-score median was 46.8 (IQR: 41.8, 50.1). No statistically significant sex differences were observed for age, BMI category, physical activity T-score, or number of days with any or at least 12 hours of CGM data. Self-reported physician diagnoses included high blood sugar (10% male, n = 3; and 0% female), abnormal cholesterol (3% male, n = 1; 3% female, n = 2), and insulin resistant (1% female, n = 1; 0% male). The majority of participants (93%) reported no diagnosis (96% female, n = 73; 86% male, n = 25).
CGM-Derived Metrics in Young Adults
The overall median for mean glucose was 104.6 mg/dL (IQR: 100.4, 110.9). Males had significantly higher mean glucose than females (109.6 vs 103.6 mg/dL; P = .009) (Table 3, Figure 1). Similar sex differences were observed for estimated A1c (5.4% vs 5.2%; P = .009), GMI (5.9 vs 5.8; P = .009), and AUC (109.5 vs 103.4; P = .010). %TOR>140 mg/dL and 180 mg/dL were low overall (median 4.2% and 0.1%, respectively) and did not differ by sex (P = .065 and P = .076). %TIR of 63 to 140 mg/dL was high (median 95.1%) with no sex group difference (P = .13). Males had a higher median daily number of excursions >140 mg/dL (1.6 vs 1.3; P = .027) and higher HBGI scores (0.3 vs 0.2; P = .041), whereas females had higher LBGI (1.0 vs 0.4; P = .004). GV metrics (CV, MAGE, MODD) did not differ by sex (all Ps > .05).
CGM Metric Values Overall and by Sex, Median (Q1, Q3).
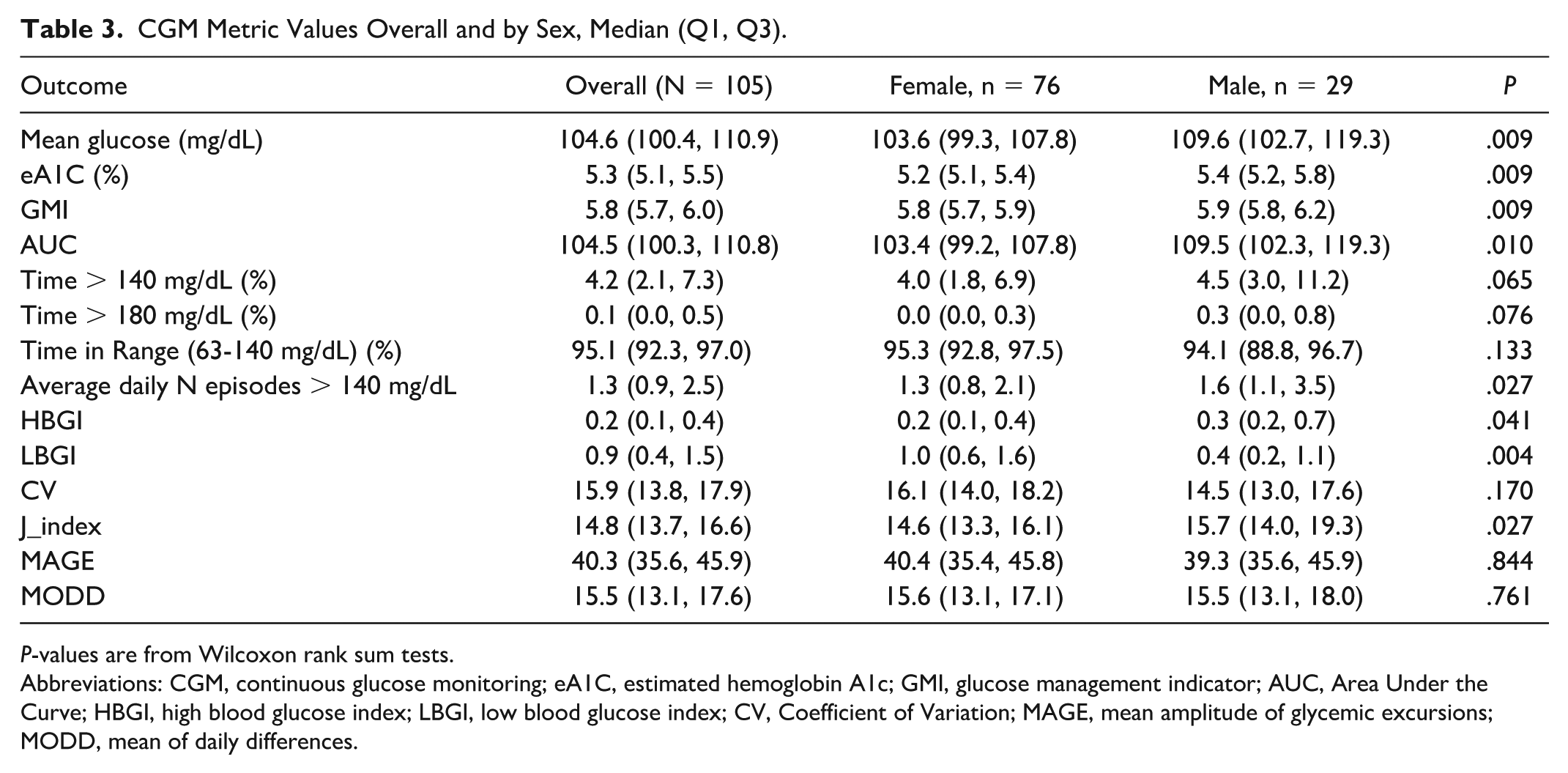
P-values are from Wilcoxon rank sum tests.
Abbreviations: CGM, continuous glucose monitoring; eA1C, estimated hemoglobin A1c; GMI, glucose management indicator; AUC, Area Under the Curve; HBGI, high blood glucose index; LBGI, low blood glucose index; CV, Coefficient of Variation; MAGE, mean amplitude of glycemic excursions; MODD, mean of daily differences.
Aggregated Glucose Trends and Peaks
Aggregated hourly curves (Figure 2) and %TOR heatmaps (Figure 3) showed that males maintained higher glucose levels throughout the day and exhibited more pronounced peaks, particularly after noon and 6 pm. Cumulative %TOR >140 mg/dL increased by 11.1% per day overall (95% CI: 10.1%, 12.0%), with similar slopes for females (11.5%) and males (10.6%) (Figure 4).
Associations of Age, BMI, and Physical Activity With CGM Metrics
Figures 5 to 7 display correlations of age, BMI, and physical activity T-scores with CGM metrics. Higher BMI correlated with lower glycemic variability: CV (r = −.35, P < .001), MAGE (r = −.35, P < .001), and MODD (r = −.27, P < .001). BMI showed small, nonsignificant positive correlations with mean glucose, eA1c, GMI, and AUC (all r ≈ 0.14; P ≈ .15-.16). Age was minimally associated with mean glucose indices but positively associated with variability measures (CV r = .20, P = .043; MAGE r = .14, P = .15; MODD r = .18, P = .063). Higher physical activity showed modest, nonsignificant associations with lower exposure metrics (e.g., mean glucose r = −0.11, P = .28; %TAR >140 r = −0.19, P = .051).
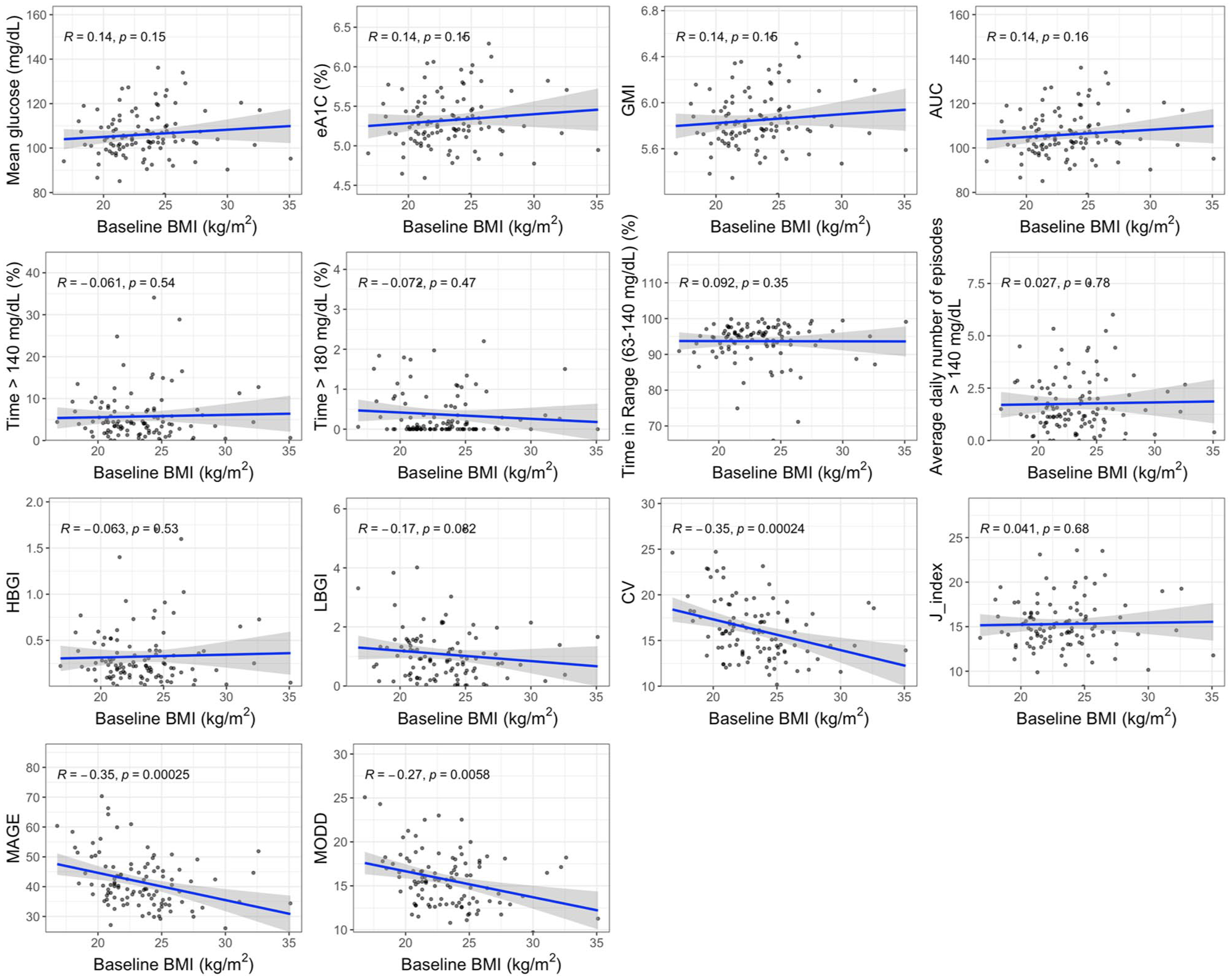
Associations between BMI and CGM metrics. Scatter plots with regression lines and Spearman’s correlations and P-values.
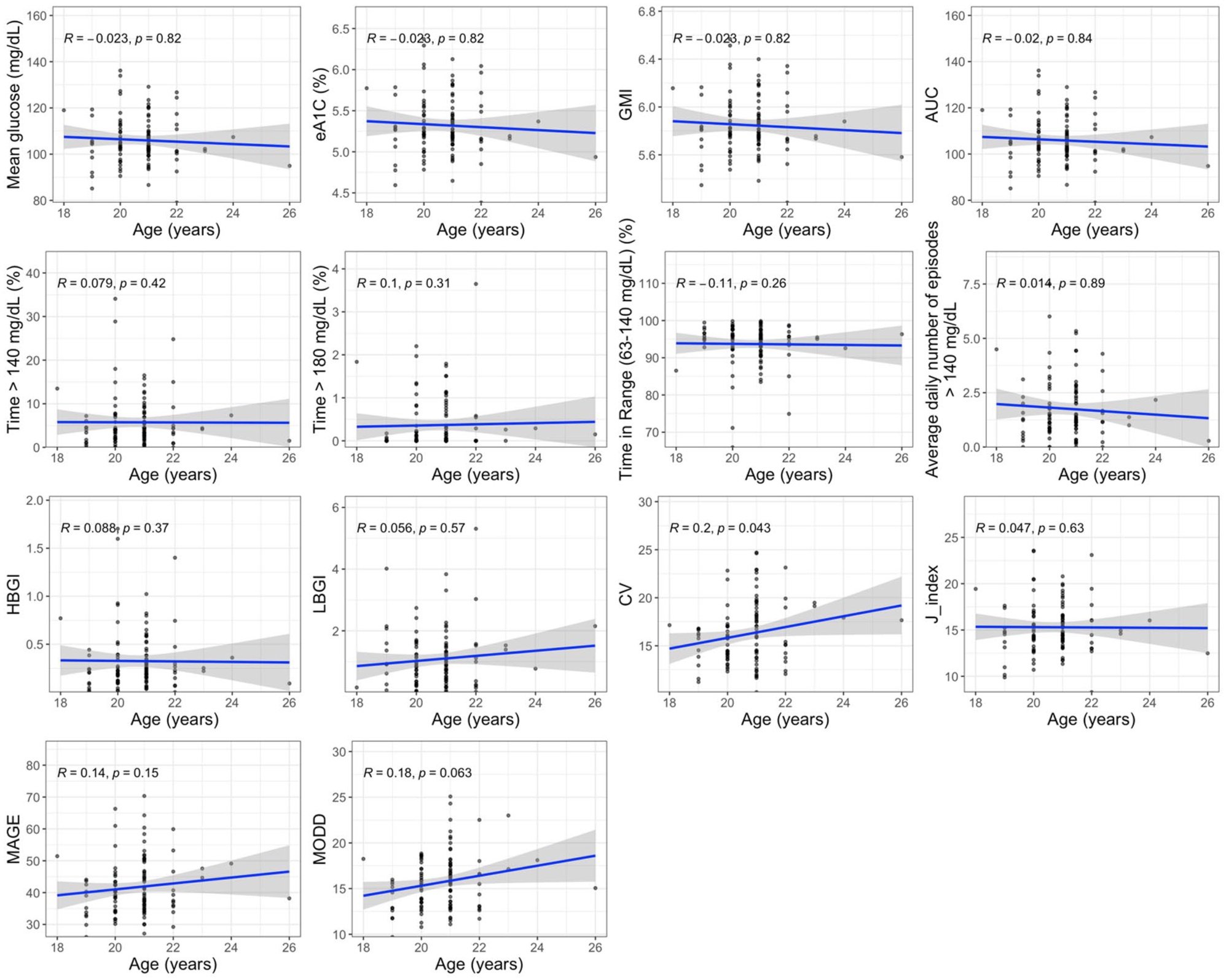
Associations between age and CGM metrics. Scatter plots with regression lines and Spearman’s correlations and P-values.
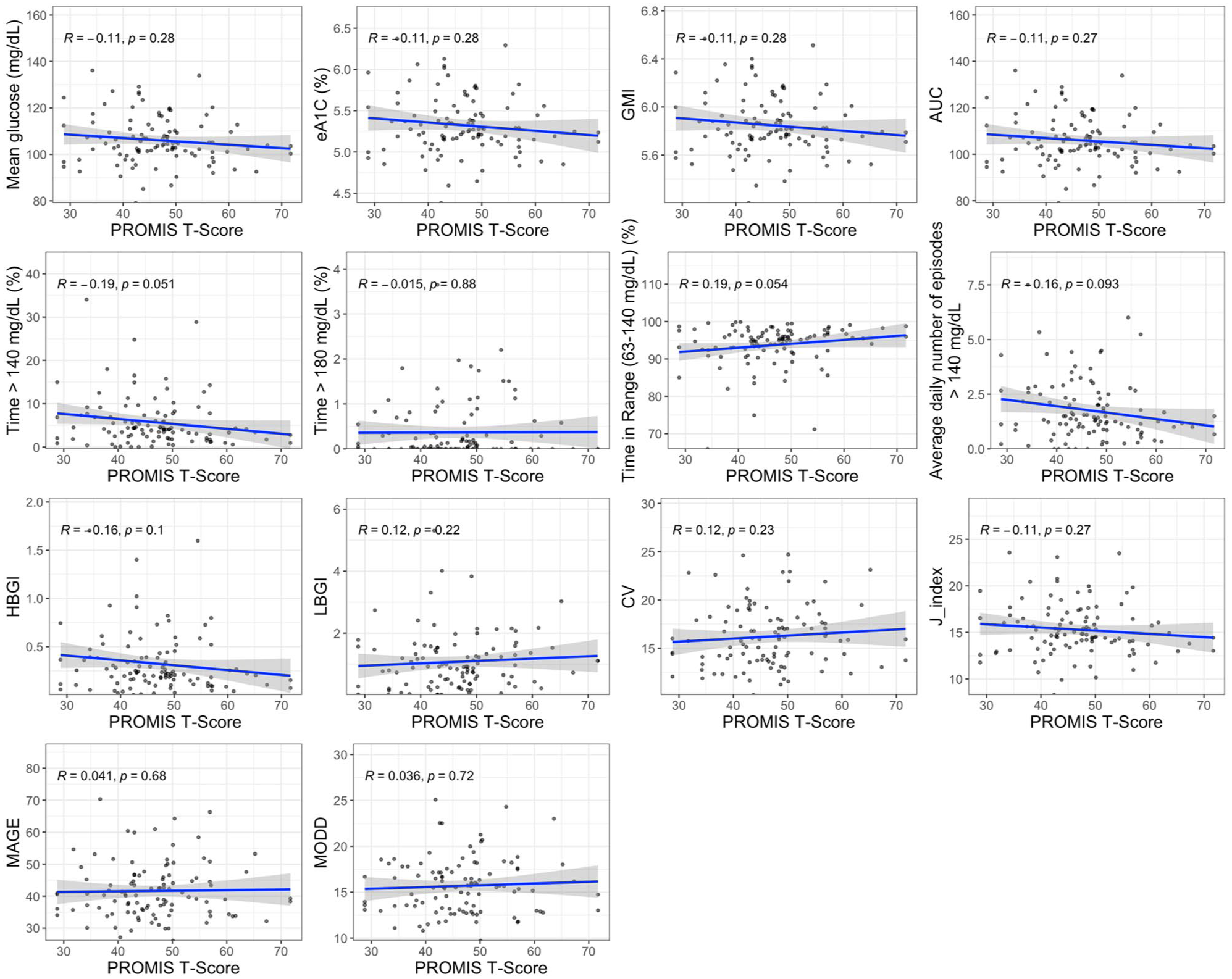
Associations between PROMIS physical activity measure T-score and CGM metrics. Scatter plots with regression lines and Spearman’s correlations and P-values.
In multivariable models (sex, BMI, age, physical activity categorized at the median) (Table 4), males had higher mean glucose (+6.4 mg/dL; 95% CI: 0.5, 12.4; P = .035), eA1c (+0.2%; 95% CI: 0.0, 0.4; P = .035), GMI (+0.2; 95% CI: 0.0, 0.3; P = .035), and AUC (+6.4; 95% CI: 0.4, 12.4; P = .036), but lower LBGI (−0.6; 95% CI: −1.0, −0.3; P = .001) than females. Higher BMI was associated with lower variability (CV −0.3; 95% CI: −0.5, −0.1; P = .004; MODD −0.3; 95% CI: −0.5, −0.1; P = .017). Each additional year of age predicted lower %TIR (−0.3 percentage points; 95% CI: −0.5, −0.1; P = .001) and higher LBGI (+0.2; 95% CI: 0.0, 0.3; P = .011) and CV (+0.7; 95% CI: 0.1, 1.3; P = .026). Participants at or above the median for physical activity T-score had slightly higher %TIR (+1.5 percentage points; 95% CI: −0.1, 3.1; P = .063), with no other significant differences.
Quantile Regression Estimates of CGM Metrics by Sex, BMI, Age, and Physical Activity.
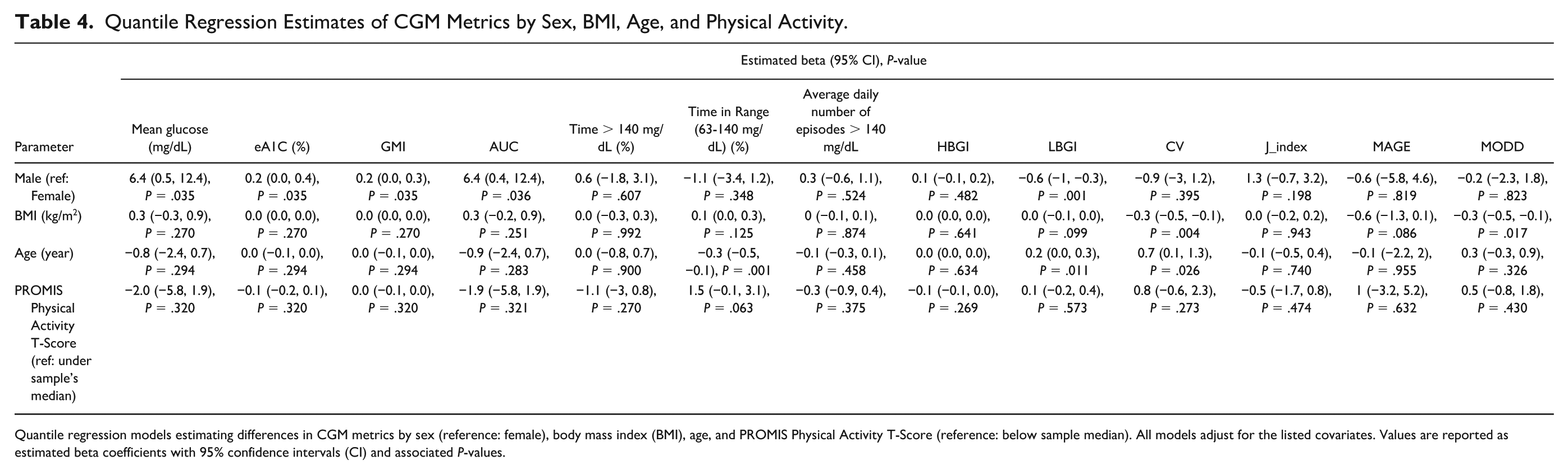
Quantile regression models estimating differences in CGM metrics by sex (reference: female), body mass index (BMI), age, and PROMIS Physical Activity T-Score (reference: below sample median). All models adjust for the listed covariates. Values are reported as estimated beta coefficients with 95% confidence intervals (CI) and associated P-values.
Discussion
In this CGM-based glucose profiling study of young adults aged 18 to 26 years, we report a full characterization of conventional metrics as well as observed statistically significant sex differences in CGM-derived exposure metrics. Males had higher mean glucose, GMI, estimated A1c, overall exposure (AUC), and more daily excursions >140 mg/dL than females, whereas both sexes maintained high time in range and minimal hyperglycemia. Indices of glycemic variability (CV, MAGE, MODD) were similar by sex. These patterns remained after adjustment for BMI, age, and physical activity, indicating that sex differences in early-adult glycemic exposure are detectable even among young adults displaying normoglycemic glucose patterns. Prior work15,22 has shown individual-level discrepancies between CGM-derived GMI and laboratory HbA1c; we include GMI in this study to align with standardized CGM reporting and facilitate comparison with prior literature, not to define glycemic status.
Our findings align with and extend prior CGM metrics reporting that has largely focused on middle-to-older adulthood or pooled wide age ranges.3-5,7 Consistent with those reports, young adults in the present study exhibited high glycemic stability, spending 95% of time in range (63-140 mg/dL), with time greater than 140 mg/dL at 4.2% and over 180 mg/dL at 0.1% over 180 mg/dL. These values are comparable with the ~96% TIR (70-140 mg/dL) reported by Shah et al. 7 While Keshet et al 3 and Spartano et al 4 reported similarly low TOR in middle-aged and older adults, our study extends the reliability of these profiles in young adults, a developmental phase not well represented in CGM research among people without diabetes. Notably, although Keshet et al 3 included sex-stratified analyses, their cohort did not encompass the 18- to 26-year age range examined in the current study. As summarized in Table 1, only two prior CGM studies in nondiabetic populations included participants with the young adult age range examined here.5,7 Together, these cohorts represented a total of 46 individuals aged 18 to 25, a subset of samples spanning childhood through adulthood. The present study builds on this work by focusing exclusively on young adults aged 18 to 26 years, providing a more targeted characterization of glycemic patterns and sex-based differences during early adulthood.
Differences in CGM system accuracy and study design should also be considered when comparing across normative cohorts. The current study used an unblinded Dexcom G7, which demonstrated high accuracy with a mean absolute relative difference (MARD) of approximately 12% when compared with venous reference measurements, comparable with other CGM systems. 23 Further, the use of CGM functions as a digital feedback tool, and thus glucose values could have been brought under greater control through contingency-shaped learning, which may differ from outcomes obtained with masked CGM and the data yielded in masked protocols. 24
The combination of higher average exposure in males with no between-sex differences in variability suggests a right-shifted distribution rather than greater instability. Although we did not measure potential mediators (e.g., body composition, insulin sensitivity, or menstrual phase), this interpretation is consistent with a general shift in central tendency. The higher LBGI among females, while still low in absolute terms, indicates relatively greater exposure to lower glucose values without evidence of clinically meaningful hypoglycemia in this cohort.
Body mass index showed inverse associations with variability metrics (CV, MAGE, MODD). While lower variability can be interpreted as favorable, these results warrant caution because they occurred alongside small, nonsignificant positive correlations with exposure metrics (e.g., mean glucose, AUC). Variability should therefore be interpreted in the context of overall exposure rather than in isolation. Age exhibited small, directionally consistent associations—higher variability and lower TIR with increasing age—within this restricted 18- to 26-year range. Physical activity showed modest, nonsignificant trends toward lower exposure, which may reflect limitations of self-report and the available sample size for these analyses.
Conclusions
Strengths of this research include a focus on an underrepresented age group, unmasked CGM use under free-living conditions, consensus-guided metrics, and multivariable modeling of key behavioral and biological covariates. Our sample size for the given young adult age range serves as a strength when compared with prior literature. Limitations include a single-university sample with a female-skewed distribution, self-reported anthropometrics, use of a pediatric-form physical activity instrument not normed for adults, and lack of synchronized data on diet, sleep, or body composition. Pregnancy status was not assessed, which may have introduced minor heterogeneity in glucose variability among female participants. However, the prevalence of pregnancy among college-aged women is low—estimated at roughly 3.5% annually—suggesting that the absence of this measure is unlikely to have materially affected our findings. 25 Although no participants self-reported a diabetes diagnosis (Table 2), diabetes status was not objectively confirmed using laboratory measures such as HbA1c, fasting plasma glucose, or oral glucose tolerance testing at baseline. As a result, it is possible that some individuals with undiagnosed prediabetes were included, and the cohort may therefore not represent a fully normative glycemic population. In addition, BMI-based weight classifications may underestimate adiposity risk among Asian participants, who comprised 46% of our sample. Lower BMI thresholds (≥23 kg/m2 for overweight, ≥27.5 kg/m2 for obesity) have been recommended for Asian populations, 26 suggesting that some participants categorized as “normal weight” may fall within ranges for central adiposity or metabolic dysregulation. However, all inferential analyses (e.g., correlations and regression models) utilized BMI as a continuous variable. This analytic approach reduced potential bias related to differences in BMI cutoffs across racial/ethnic groups and preserved information on the full range of BMI values.
The observational design precludes causal inference. Future research with larger and more diverse young adult cohorts can incorporate direct assessments of body composition and physical fitness levels, reported menstrual phase, HbA1c and fasting plasma glucose testing, and additional behavioral assessments such as daily diet and sleep.
Footnotes
Acknowledgements
None.
Abbreviations
AUC, Area Under the Curve; BMI, Body Mass Index; CGM, Continuous Glucose Monitoring; eA1C, Estimated A1c; GMI, Glucose Management Indicator; GV, Glycemic Variability; HBGI, High Blood Glucose Indicator; IQR, Interquartile Range; LBGI, Low Blood Glucose Indicator; MAGE, Mean Amplitude of Glycemic Excursions; MODD, Mean of Daily Differences; PROMIS, Patient-Reported Outcomes Measurements Information System; REDCap, Research Electronic Data Capture; TIR, Time In Range; TOR, Time Out of Range.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The Authors declare that there is no conflict of interest that would affect the data analysis or interpretation. DSB maintains an active research agreement with Dexcom Inc. for the procurement of sensors and devices intended exclusively for research use.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project received partial support during a pilot phase focused on protocol development and data validation as part of a larger project funded by the National Institutes of Health (NIH), National Institute on Minority Health and Health Disparities (NIMHD), under award number 3P50MD017344-03S3 to author D.B. The funder had no role in the study design, data collection, data analysis, interpretation of data, manuscript preparation, or the decision to submit the manuscript for publication.
