Abstract
The introduction of continuous glucose monitoring (CGM) has been considered a transformative monitoring tool in diabetes management. However, its adoption remains limited in the Gulf region, especially for patients with type 2 diabetes, due to cost, lack of reimbursement strategies, variability in healthcare infrastructure, and lack of trained health care providers (HCPs). The lack of regional guidelines tailored to the unique demographic, cultural, and health care needs of the Gulf population has resulted in low adoption and inconsistent use of CGM in clinical practice, leaving many patients without adequate advanced glucose monitoring options. This expert opinion statement evaluates the evidence for real-time CGM in the management of patients with type 2 diabetes and provides region-specific recommendations to guide HCPs in optimizing CGM use, improving patient outcomes, and addressing barriers to implementation in the Gulf region.
Keywords
Introduction
The burden of diabetes is increasing globally, with an estimated 6.7 million adults dying as a result of diabetes or its complications in 2021. 1 Notably, more than 90% of these cases are type 2 diabetes (T2D).1,2 The Gulf region—comprising Saudi Arabia, Kuwait, Bahrain, Oman, Qatar, and the United Arab Emirates (UAE)—has one of the highest prevalence rates of T2D, reaching up to 22% among adults. 3 This epidemic is influenced by a combination of genetic predisposition and lifestyle factors, such as physical inactivity, dietary patterns, and a high prevalence of obesity in this population.2,3 Effective glycemic control is critical to reducing the risk of long-term complications, such as cardiovascular disease, nephropathy, retinopathy, and neuropathy, particularly in patients on intensive insulin therapy (IIT).4-6
Monitoring glucose levels is a cornerstone of diabetes management. Previously, traditional capillary blood glucose (CBG) monitoring was commonly used. However, its limitations, such as pain, inconvenience, poor adherence, and incomplete glucose profiles, can hinder optimal decision-making. 7 In addition, laboratory-measured glycated hemoglobin (HbA1c), which is considered the gold standard for assessing glycemic control and risk of long-term complications in people with diabetes, has limitations of its own. HbA1c can only be reviewed infrequently and can mask the risk of hypoglycemia or extreme glucose fluctuations.8,9 The introduction of continuous glucose monitoring (CGM) has been considered a transformative monitoring tool, offering detailed insights into glucose variability and trends, allowing for more accurate assessment of mean glycaemia, hypoglycemia and glycemic variability, and informed adjustments to therapy.7 Real-time continuous glucose monitoring (rtCGM) has the ability to provide continuous glucose readings and real-time alerts for hyperglycemia and hypoglycemia, allowing timely intervention by patients and health care providers (HCPs). 10
Research has shown that CGM use improves glycemic control in individuals with type 1 diabetes (T1D) and T2D.10-17 As a result, several guidelines now recommend the early adoption of CGM for diabetes management, as early as at the time of diagnosis. 18
In the Gulf, Middle East, and North Africa (MENA) regions, the adoption of CGM technologies has been hindered by several barriers, including cost, lack of reimbursement strategies, variability in health care infrastructure, and limited number of trained HCPs. 19 Furthermore, there is a lack of regional guidelines tailored to the unique demographic, cultural, and health care needs of the Gulf population. This has resulted in low adoption and inconsistent use of CGM in clinical practice, leaving many patients without adequate advanced glucose monitoring options.19,20
This expert opinion article aims to evaluate the evidence for rtCGM in the management of patients with T2D and provide region-specific recommendations to guide HCPs in optimizing CGM use, improving patient outcomes, and addressing barriers to implementation in the Gulf region.
Methods
Expert Panel Selection
An expert panel of diabetes consultants and endocrinologists from the Gulf Cooperation Council (GCC) was convened. Experts were selected based on their clinical expertise in managing T2D, their experience with CGM technologies, and the need for regional representation to ensure relevance to the Gulf population.
Evidence Review and Data Synthesis
A literature review was conducted to evaluate the clinical effectiveness and safety of rtCGM, as well as their impact on glycemic control, hypoglycemia prevention, and quality of life. The review also considered regional challenges and factors influencing CGM adoption based on the available local evidence, clinical practice, expert insights, and real-world experiences from the Gulf region.
The literature search was performed using PubMed, with additional sources from regional health care reports and relevant international and regional guidelines. The search was conducted using a combination of Medical Subject Headings (MeSH) terms and free-text keywords related to CGM and T2D. The following key search terms were used, either alone or in combination:
“continuous glucose monitoring” OR “CGM” OR “real-time continuous glucose monitoring” OR “rtCGM”
“type 2 diabetes” OR “T2D”
“intensive insulin therapy” OR “multiple daily injections” OR “MDI”
“Gulf region” OR “Middle East” OR “MENA” OR specific country names (eg, Saudi Arabia, UAE, and Qatar)
The search was restricted to studies published in English and included randomized controlled trials (RCTs), observational studies, systematic reviews, meta-analyses, expert consensus reports, and the most recent international and regional guidelines that specifically addressed the use of CGM in T2D.
Consensus Development
The expert panel discussed and analyzed the evidence and clinical experiences. Key discussion points included the following:
The role of rtCGM in the management of T2D.
Patient selection criteria and indications for rtCGM.
Regional barriers and facilitators to CGM implementation.
Development of Recommendations
Recommendations were formulated based on the following:
Clinical evidence and published literature.
Expert insights and real-world experiences from the Gulf region.
Consideration of health care system capabilities, patient preferences, and cost-effectiveness in the Gulf.
Review and Approval
A draft of the consensus statement was reviewed and refined by all experts. Suggestions were incorporated as deemed appropriate by the authors. Final recommendations were agreed upon and approved unanimously.
Current T2D Landscape in the Gulf Region
The Gulf region is facing an increasing burden of T2D, marked by alarmingly high prevalence rates, poor glycemic control, and significant gaps in care delivery. 3 Projections indicate that the number of people with diabetes in the region could double by 2045, highlighting the urgent need for effective preventive and management strategies.1,3
Despite advancements in diabetes care and investments made in the GCC to control the disease, achieving optimal glycemic control remains a persistent challenge in the region, especially given common risk factors such as old age and obesity.3,21-23
A survey-based study aimed to describe the current status of diabetes control and diabetes management in Saudi Arabia, Kuwait, and the UAE. 24 In this study, the mean HbA1c level was 8.3%, which is considerably higher than the glycemic targets recommended by the American Diabetes Association (ADA) and International Diabetes Federation (IDF) [HbA1c <7.0% (53 mmol/mol)], indicating poor glycemic control in the Gulf region.24-26 This poor control was also shown in the high proportion of patients with micro and macrovascular complications.
In addition, the Gulf cohort of the observational A1chieve study demonstrated a mean baseline HbA1C of 9.4%. 27 In Saudi Arabia, less than 25% of patients achieve adequate glycemic control, reflecting significant gaps in diabetes care. 28 Another study revealed that the pooled prevalence of uncontrolled T2D in Saudi Arabia was 77.7%. 29 Similar results were reported in the national RIMODIS study, with more than two thirds of patients failing to achieve their glycemic target levels. 22
Despite strong evidence supporting the use of CGM for improving glycemic control, its uptake remains low due to cost, limited awareness, and accessibility challenges.19,30
A survey-based study conducted in Saudi Arabia among patients with diabetes demonstrated that only 11.3% of the surveyed population use CGM. 30 Many patients continue to rely on traditional CBG monitoring, which provides limited data and does not capture glycemic variability effectively.
Increasing adoption of CGM in clinical practice has the potential to improve diabetes care in the region, enabling better glucose management, reduced hypoglycemia, and improved patient satisfaction. 19 Furthermore, it provides the potential to transform the current standard model of care, through telemedicine and virtual diabetes clinic, and to address the increasing cost of diabetes care, given the strong evidence for cost-effectiveness in selected cohorts.31,32
Overview of CGM
Adequate diabetes management requires close monitoring of glucose levels. 33 Over the past 100 years, multiple innovations have improved glucose monitoring methods to address this need. The introduction of CGM technologies has been the highlight of these advancements, with numerous studies supporting the glycemic benefits and value associated with its use for individuals with T2D.
Real time CGM systems, measure glucose values and automatically display a recent reading while offering alerts and alarms for current or expected glycemic events, including hyperglycemia and hypoglycemia. 34 These predictive alerts provide additional safety advantages, allowing patients to avoid hypoglycemic events as well as eliminate their fear of hypoglycemia.34-36
The ambulatory glucose profile (AGP) is a report summarizing CGM data over a period of 5 days to 3 months. 37 It has been recommended as a standardized tool that serves as a fast and easy-to-assess entry point for physicians and caregivers to analyze CGM data.34,38 The current international consensus report on the use of CGM systems indicates that to effectively manage diabetes therapy, time in range (TIR) should be considered alongside HbA1c decision-making (Figure 1).34,39
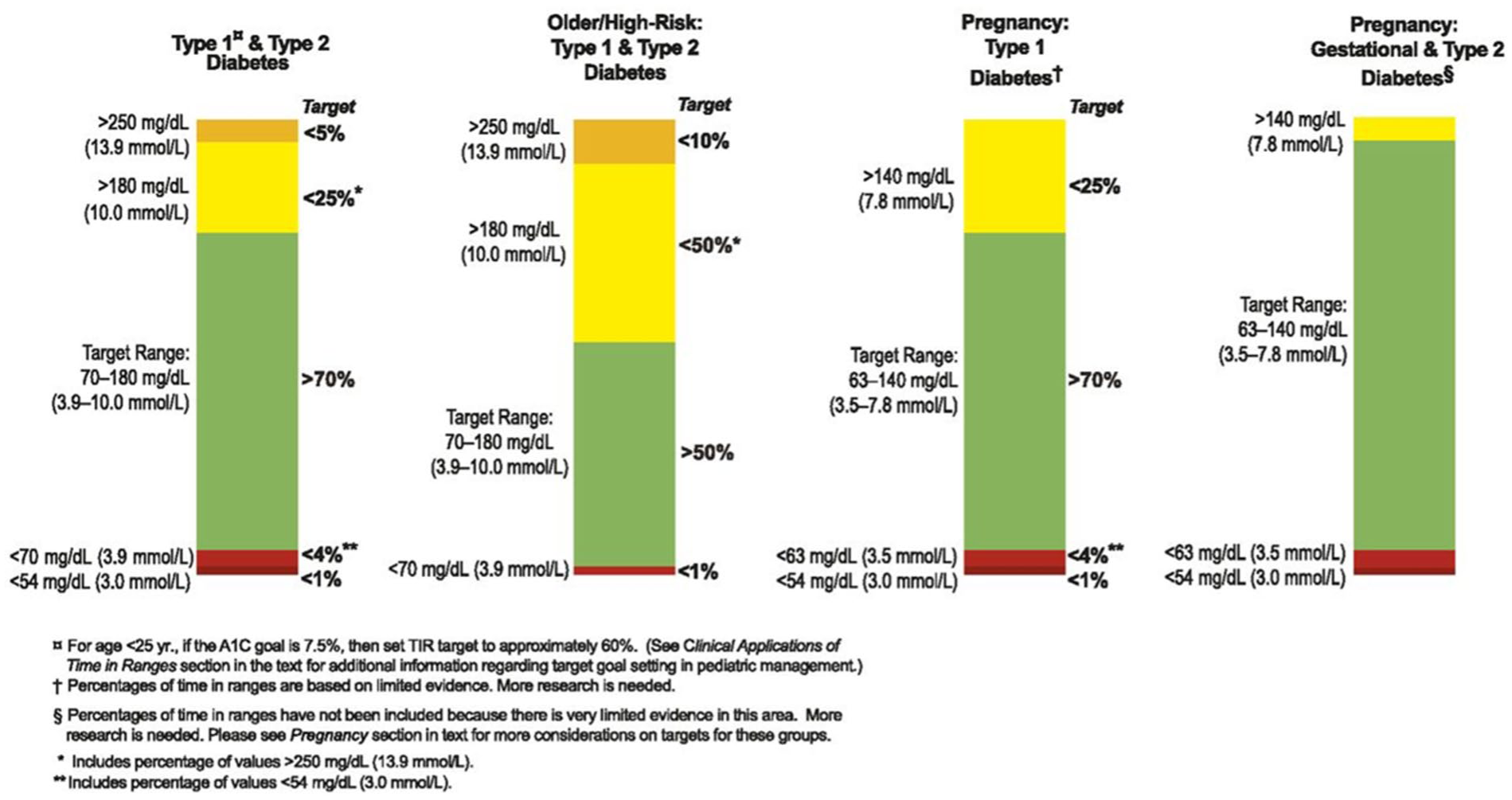
GM-based targets for different diabetes populations. 39
Having standardized metrics for evaluating CGM data has significantly enhanced and streamlined the analysis and interpretation, allowing clinicians and patients to make better-informed therapy modifications.
However, there continues to be a lack of adoption of this technology for T2D worldwide and in the Gulf region. The under-adoption of CGM for T2D could be driven by a lack of regional data, guidelines, and awareness among many clinicians, who may be unfamiliar with CGM and how the use of these devices may help individuals with T2D achieve their glycemic targets.19,33 Other limitations could be the cost of these devices compared to traditional CBG and the lack of reimbursement.
Evidence for rtCGM in T2D
Several studies have investigated the effectiveness of rtCGM in patients with T2D. The findings of the Steno2tech Study support the use of CGM in individuals with T2D treated with insulin. Over a 12-month period, CGM was superior to CBG monitoring in improving glucose control and patient-reported outcomes, including satisfaction, well-being, and health behavior. 40 Similarly, in the DIaMonD (T2D) study, a high percentage of adults who received multiple daily insulin injections for T2D used CGM on a daily or near-daily basis for 24 weeks and had improved glycemic control. 17
In the real-world Landmark study, rtCGM use was correlated with improved HbA1c levels for individuals with T1D or T2D who use IIT. 41 Patients reported a significant reduction in diabetes stress and hypoglycemia concerns, and the majority were satisfied or very satisfied with the use of rtCGM. 41
The benefits of rtCGM use have also been reported in individuals with T2D who are managed with or without intensive insulin treatment.17,42,43 The MOBILE study showed that CGM use for patients on basal insulin was associated with significantly lower HbA1c and improved glycemic control at 8 months.10 These outcomes were almost completely lost upon the discontinuation of CGM in the MOBILE Follow up study, indicating that the long-term use of a rtCGM is necessary to maintain the glycemic benefits and contribute to risk reduction for long-term complications. 44
Similarly, results from the GRACE study revealed that rtCGM use was associated with significant glycemic improvements in T2D patients treated with basal insulin only or noninsulin therapy. 45
Recent studies are exploring the effectiveness of rtCGM in patients with T2D on noninsulin therapies.46,47 The use of CGM in this patient population was shown to reduce the glycemic variability and increase TIR.
Several studies have reported that the use of rtCGM was associated with a reduced risk of diabetes-related complications and that its use was considered cost-effective against CBG in individuals with T2D, including those on non-intensive therapies.48-51
A retrospective analysis of one of the largest administrative claims research databases in the United States revealed that the use of rtCGM in patients with T2D for more than 6 months was associated with decreased diabetes-related medical costs, inpatient hospital admissions, and total hospital admissions. 51 The study also highlighted that increased access to rtCGM may help reduce the diabetes-related cost of care.
Expert Recommendations for the Use of CGM in T2D
Several international and regional guidelines have emphasized the importance of CGM as a tool to optimize glycemic management in individuals with diabetes, including those with T2D.18,34,52-56
In alignment with these guidelines, the expert panel has put together region-specific recommendations to guide HCPs in the Gulf region in the effective implementation of CGM technologies, aiming to standardize care, improve patient outcomes, prevent complications, and enhance quality of life (Table 1).
Expert Recommendations for the Use of CGM in T2D.

Limitations and Gaps in Knowledge
Despite the growing global evidence supporting the use of rtCGM in T2D, significant gaps remain in the regional context, particularly in the Gulf region. One of the primary limitations is the lack of comprehensive regional data on the clinical effectiveness, safety, and long-term outcomes of rtCGM use in patients with T2D. Most of the available evidence is derived from studies conducted in Western populations, which may not fully account for the unique demographic, cultural, and health care characteristics of the Gulf region.
In addition, there is limited research addressing the economic feasibility of rtCGM implementation in the region, including cost-effectiveness analyses and the impact of reimbursement strategies on adoption rates. The variability in health care infrastructure across Gulf countries further complicates the generalizability of existing data, highlighting the need for country-specific evaluations.
Further research is required to explore the long-term impact of rtCGM on glycemic control, hypoglycemia prevention, and quality of life in Gulf populations, particularly in adults with T2D on IIT. These studies should be extended to patients receiving bolus insulin as well as noninsulin users since the benefit of rtCGM has been established in these populations as well. Studies assessing HCP attitudes, adoption rates, patient adherence, and barriers to CGM adoption are also critical to developing tailored strategies for widespread implementation. Future efforts should be directed toward conducting gulf-specific RCTs, cost-effectiveness studies, and real-world studies. Finally, collaborative efforts to generate robust, region-specific evidence will be essential for optimizing diabetes care and addressing the growing burden of T2D in the Gulf region.
Conclusion
In conclusion, rtCGM is a transformative tool for managing T2D. While these benefits apply to a wide range of patients, those on IIT would currently benefit the most. Other type 2 patients, like those on basal insulin, would also benefit from using the device. rtCGM is regarded for its accuracy, multiple insertion sites, customizable alerts, and ease of use. Integrating rtCGM into standard care can significantly improve clinical outcomes, supported by robust evidence, international guidelines, and expert consensus in the Gulf region.
Footnotes
Acknowledgements
The authors acknowledge the work of Huda Sobhy for the editorial and medical writing assistance.
Abbreviations
CGM, continuous glucose monitoring; HCPs, healthcare providers; T2D, type 2 diabetes; UAE, United Arab Emirates; IIT, intensive insulin therapy; CBG, capillary blood glucose; HbA1c, glycated hemoglobin; rtCGM, real-time continuous glucose monitoring; T1D, type 1 diabetes; MENA, Middle East and North Africa; GCC, Gulf Cooperation Council; MeSH, medical subject headings; RCTs, randomized controlled trials; ADA, American Diabetes Association; IDF, International Diabetes Federation; AGP, ambulatory glucose profile; TIR, time in range; TBR, time below range; TAR, time above range; GRI, glycemic risk index.
Authorship Confirmation/Contribution Statement
All authors contributed to the conceptualization, discussion, and development of the expert recommendations. All authors provided critical revisions, contributed to the final manuscript, and approved the submitted version.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MEA has received research fund from Medtronic and Sanofi; served on an advisory panel for Medtronic, Insulet, Abbott, VitalAire, Sanofi; and received honoraria for speaking from Abbott, Eli Lilly, Medtronic, Novo Nordisk, Sanofi, and VitalAire. FA has attended as a speaker for educational activities/ advisory board member sponsored by Novo Nordisk, Lilly, BI, Dexcom, Servier, Amgen, Abbott & Bayer. AB has received an honorarium from Dexcom for an educational activity. The rest of the authors declare no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dexcom has funded the medical writing and publication of the manuscript.
