Abstract
Background:
Technosphere insulin (TI) is an ultra-rapid-acting inhaled insulin approved for glucose management in adults with diabetes mellitus. Using a higher modified initial conversion dose than in the current United States Prescribing Information, this study assessed supplementing or replacing automated insulin delivery (AID) systems with TI.
Methods:
Adult participants with type 1 diabetes (glycated hemoglobin [HbA1c], 7%-11%) using an AID system were randomized into TI + AID (TI for meals and AID for basal and corrections), TI + insulin degludec (TI for meals and corrections and insulin degludec for basal), or control group (remaining on AID) and treated for 90 days. HbA1c, forced expiratory volume in 1 second (FEV1), hypoglycemic events, and adverse events (AEs) were assessed.
Results:
Of 33 enrolled participants, 24 completed the study. All groups demonstrated comparable declines in HbA1c from baseline to end of treatment (statistically significant decline for control group). No within- or between-group statistical differences were observed in FEV1. Incidence and event rate of hypoglycemia <70 mg/dL and <54 mg/dL were similar between groups, and no severe hypoglycemic events were reported. No treatment-related serious AEs were reported, and 2 participants experienced AEs of special interest related to TI (clinically relevant decline in pulmonary function and wheezing).
Conclusions:
This proof-of-concept study demonstrated the safety and efficacy of TI, at a higher modified dose conversion, when added for mealtime control to an AID system or was used for glycemic control with basal insulin.
Introduction
Subcutaneous (SC) insulin is a commonly used diabetes treatment delivered via syringes, pens, and/or pumps, including automated insulin delivery (AID) systems. 1 SC administration is associated with substantial injection burden and adherence concerns.2,3 Meal bolus timing with SC rapid-acting insulin analogs (RAAs) fails to meet physiologic needs owing to delayed capillary absorption of injectable formulations. 4 This delayed onset of action and elimination requires SC insulin to be injected approximately 5 to 30 minutes before meals to minimize early postprandial hyperglycemia and late postprandial overcorrection, insulin stacking, 4 and consequent hypoglycemia. 5 Premeal administration is complicated by the need to estimate doses and account for variations in meal size and composition, which may leave residual insulin between meals, raising the risk for hypoglycemia. These complexities and associated consequences persist despite advances in glucose monitoring and AID systems, and only a minority of people with type 1 diabetes (T1D) meet the American Diabetes Association glycated hemoglobin (HbA1c) goals. 6
Inhaled insulin’s dry powder formulation maintains insulin in a monomeric state, enabling ultra-rapid absorption through the lungs and into the bloodstream.7-10 As a result, inhaled insulin achieves a faster onset of action and elimination, allowing dosing at the start of a meal or exercise and enabling faster corrections compared with SC RAA insulins. 11 Technosphere insulin (TI; MannKind Corporation), an inhaled recombinant monomeric human insulin with a first measurable effect observed within 12 minutes, is US Food and Drug Administration (FDA) approved for glucose management in adults with type 1 or 2 diabetes.10,12 Common adverse events (AEs; ≥2%) for TI include hypoglycemia, cough, and throat pain or irritation. 12 The cough is generally mild, intermittent, and limited to a single event, with its incidence declining by 4 to 8 weeks of continued TI use. 13 Based on pharmacokinetic and bioavailability differences between SC RAA and TI, the average conversion ratio of SC insulin to inhaled insulin (1:≈1.3) recommended in TI’s current United States Prescribing Information (USPI) 12 may be inadequate. Prior research has demonstrated that some participants required additional TI doses to achieve target glycemic outcomes. 14 Glucose clamp studies and clinical experience with TI suggest that a higher dosing conversion ratio of 1.5 to 2.8 may be needed to optimize mealtime glucose control.4,15 However, limited data exist for patients switching from an AID system to inhaled administration or supplementing AID with TI at mealtimes. A previous proof-of-concept study showed significant improvements in mean postprandial glucose excursions with a higher modified TI dose conversion compared with the lower USPI dose conversion with no safety concerns. However, the study lacked additional comparator arms. 16
This study aimed to evaluate the efficacy and safety of a 90-day treatment with TI plus AID or plus insulin degludec in participants with T1D currently using an AID system. The primary outcome was change in HbA1c, and safety outcomes included change in forced expiratory volume in 1 second (FEV1) and incidence of hypoglycemia and AEs over 90 days of treatment.
Methods
Study Design and Population
MKC-TI-192 (NCT05243628) was an open-label, multicenter, proof-of-concept study evaluating the efficacy of TI in controlling postprandial glucose in adults with T1D using an AID system. Participants were randomized into 3 treatment groups: TI + insulin degludec, TI + AID, or AID control (Table 1). All participants were provided with a Dexcom G6 Professional (G6 Pro) for collection of blinded continuous glucose monitor (CGM) data during the screening period and the last 10 days of the study, while continuing to use their own CGM devices for real-time treatment decisions. CGM analysis used blinded data (Supplementary Material). The study was conducted at 2 centers (Loma Linda, CA, and Austin, TX) between March 31, 2022, and October 17, 2022. It consisted of 5 clinic visits (screening, baseline, 2 follow-ups, and an end-of-treatment visit) and 9 telephone visits over 90 treatment days. The screening visit included baseline HbA1c measurements. The baseline visit included an FEV1 test, randomization, a meal challenge to establish a starting dose of prandial insulin (Supplementary Material), and initial administration of mealtime insulin. The final study visit (≥90 days after, unless participant discontinued prematurely) included HbA1c and FEV1 measurements (Figure 1). Eligibility criteria are listed in Table 2.
Treatment Groups.
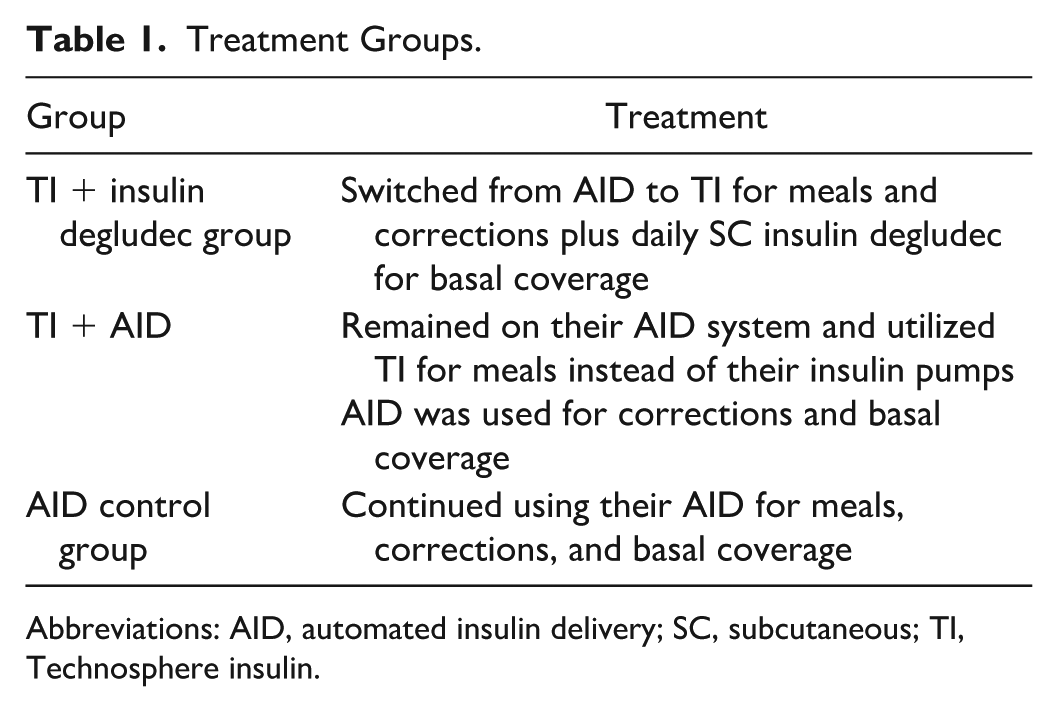
Abbreviations: AID, automated insulin delivery; SC, subcutaneous; TI, Technosphere insulin.
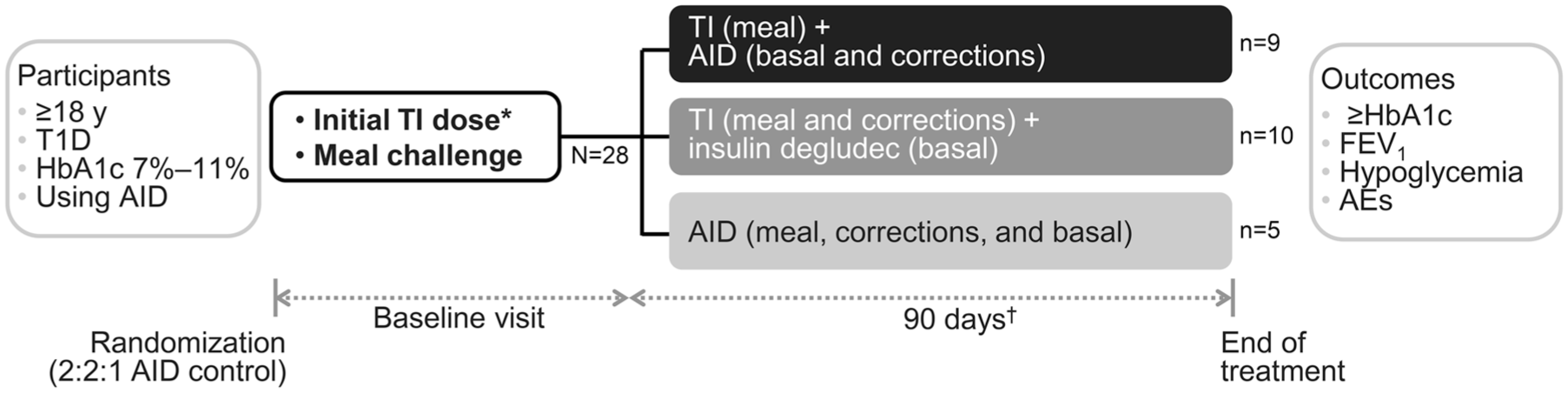
Study design.
Eligibility Criteria.
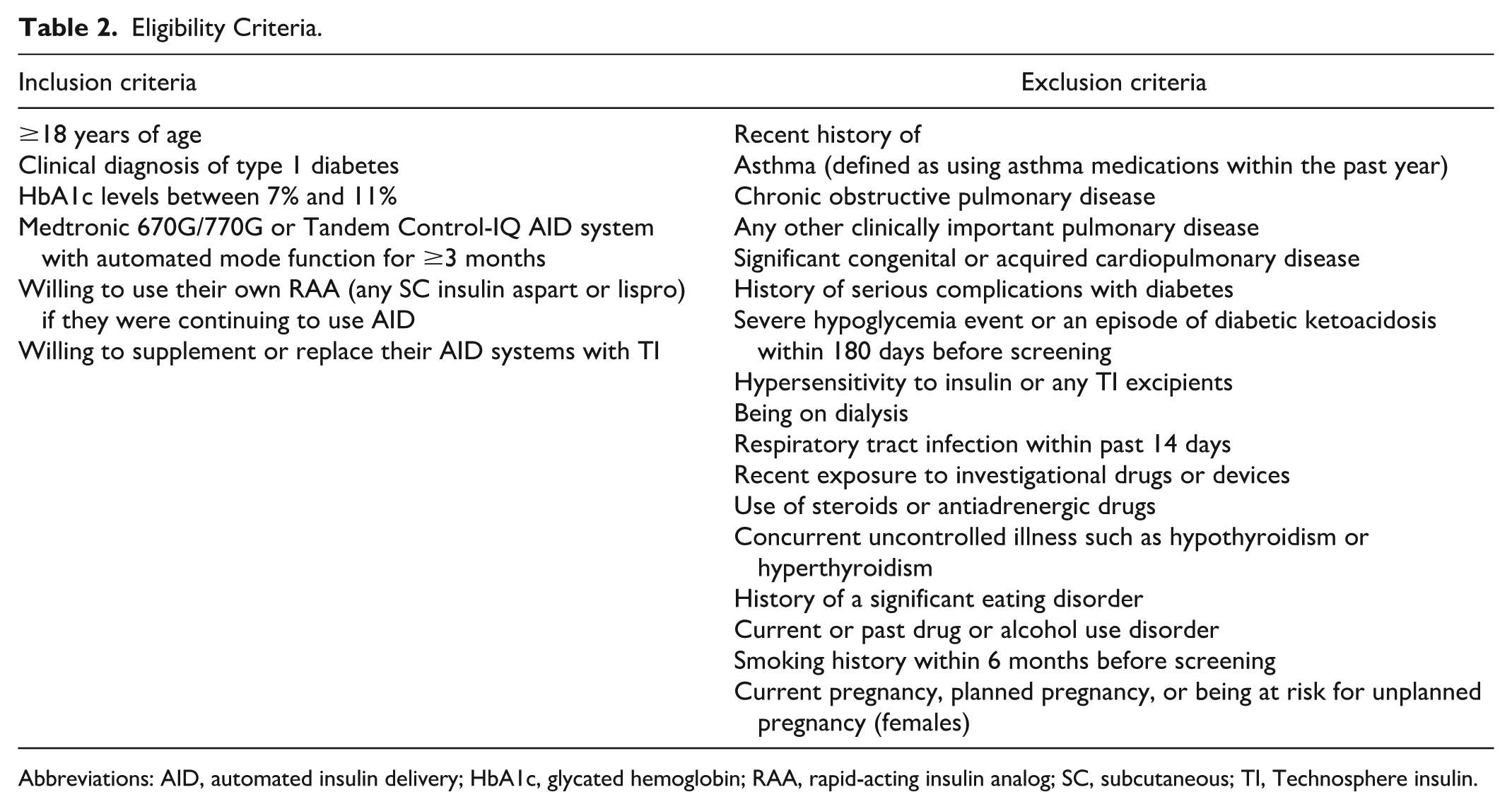
Abbreviations: AID, automated insulin delivery; HbA1c, glycated hemoglobin; RAA, rapid-acting insulin analog; SC, subcutaneous; TI, Technosphere insulin.
The protocol, informed consent form, advertising materials, and amendments associated with this study were reviewed and approved by the Advarra Institutional Review Board. The study was conducted in compliance with FDA guidelines, local regulations where applicable, International Council for Harmonization Good Clinical Practice guidelines, and the Declaration of Helsinki. All participants provided written informed consent prior to any study-specific procedures or assessments.
Study Treatment and Insulin Dosing
Based on glycemic responses to the meal challenge, participants in both TI groups were provided a TI dose to be administered at the start of each meal (breakfast, lunch, and dinner). TI doses were calculated using the higher modified dose conversion shown in Table 3 (compared with 1:≈1.3, the average conversion ratio of SC insulin to inhaled insulin across the 1-24 units RAA range). Participants in the TI + insulin degludec group were provided a dose of TI for corrections and instructed to discontinue their AID system and start once-daily SC insulin degludec administration. Participants in the TI + AID group continued to use their personal AID system with an RAA (either fast-acting insulin aspart or lispro, based on their preference) for corrections and basal coverage. Postmeal insulin corrections with TI in the TI + insulin degludec group were permitted no sooner than 60 to 90 minutes after the mealtime dose. The TI dose was titrated to glycemic effect throughout the study, per investigator discretion. Corrections could be administered during the meal challenge or throughout the study if the participant’s glucose value exceeded 160 mg/dL, or per investigator discretion. Participants in the control and AID + TI groups adjusted active insulin time if using the Medtronic 670G/770G or the basal correction dose if using the Tandem Control-IQ.
Higher Modified Dose Conversion Used in This Study vs Current Insulin Dose Conversion in the USPI.
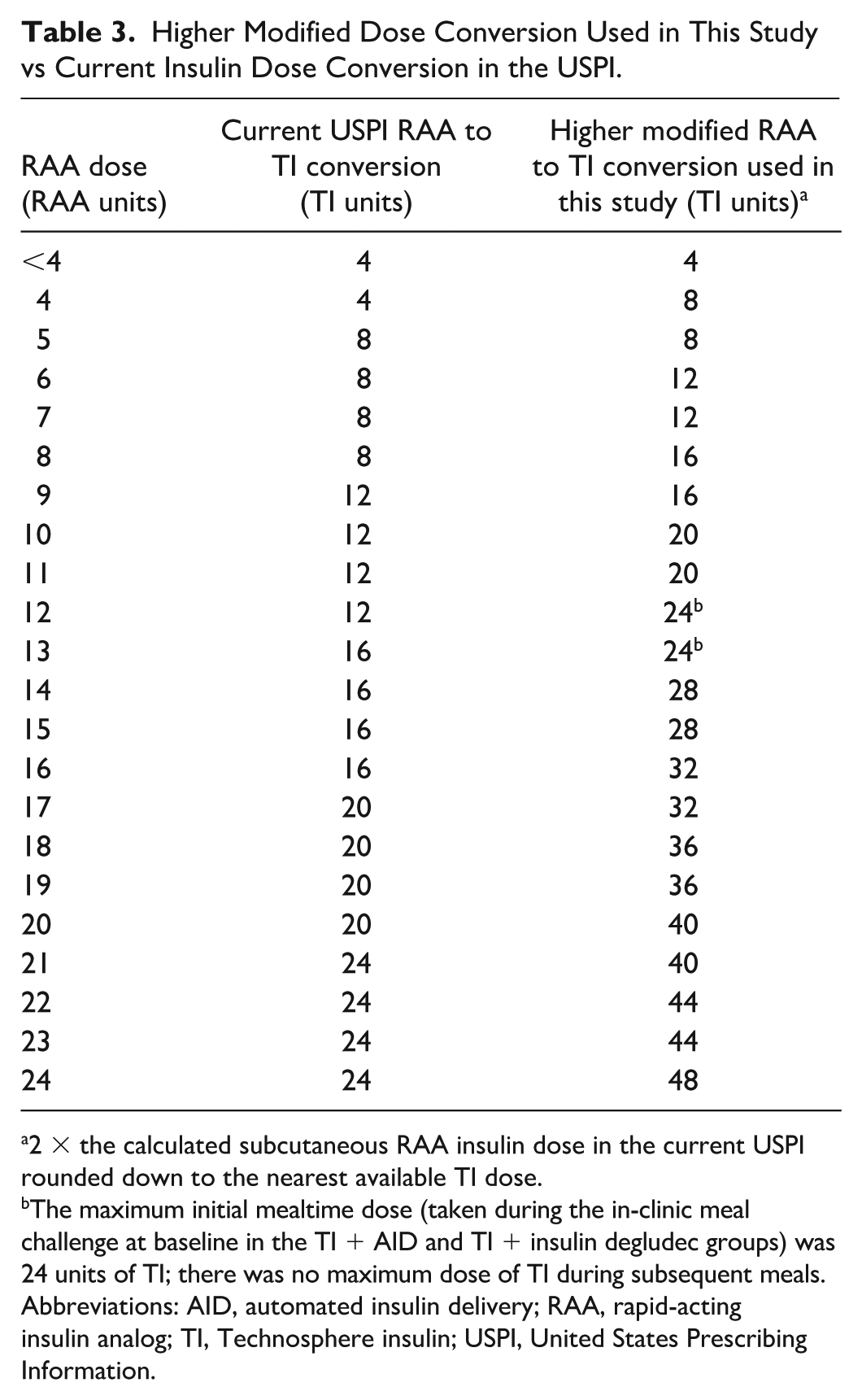
2 × the calculated subcutaneous RAA insulin dose in the current USPI rounded down to the nearest available TI dose.
The maximum initial mealtime dose (taken during the in-clinic meal challenge at baseline in the TI + AID and TI + insulin degludec groups) was 24 units of TI; there was no maximum dose of TI during subsequent meals.
Abbreviations: AID, automated insulin delivery; RAA, rapid-acting insulin analog; TI, Technosphere insulin; USPI, United States Prescribing Information.
Participants in the control group continued to manage their diabetes using their personal AID systems with their preferred RAA (either SC fast-acting insulin aspart or lispro) and were instructed to dose 5 to 15 minutes before meal start (following the RAA insulin label and investigator discretion). After the baseline visit, participants’ doses in all 3 groups were titrated through telehealth visits over the next 4 study weeks, adjusting mealtime and basal insulin based on glycemic response.
In the TI + insulin degludec group, end-of-study TI dose (relative to basal dose) was compared with the starting prescribed dose. Prescribed doses for TI bolus insulin (at breakfast, lunch, and dinner) and basal insulin were recorded at the start and end of study. Average percentage of bolus insulin to total insulin (basal + bolus) was estimated, with total bolus insulin calculated as the sum of prescribed breakfast, lunch, and dinner doses.
HbA1c
Blood samples for baseline and end-of-study HbA1c assessments were obtained at the study site’s clinic using standardized methods and equipment. All samples were analyzed by the same central laboratory (ACM Global) for consistency.
Lung Function
Pulmonary function testing was conducted using the Vitalograph asma-1 spirometer (Vitalograph Inc.) following American Thoracic Society and European Respiratory Society guidelines. 17 Multiple FEV1 tests were performed with a spirometer until 3 successful measurements were obtained, and the highest FEV1 value was used for analysis. Percent predicted FEV1 was calculated with the Global Lung Function Initiative calculator using participant age, height, and race. 18 To minimize potential measurement bias, participants with upper respiratory tract infections within 14 days before screening were asked to return 14 days after symptom resolution for rescreening to determine readiness for the baseline visit.
Hypoglycemic Events
Hypoglycemia was defined using 2 thresholds: any self-monitored blood glucose (SMBG) measurement of <70 mg/dL (level 1/mild) or <54 mg/dL (level 2/moderate). 19 Severe hypoglycemic events were defined as those requiring assistance from another person to actively administer carbohydrate, glucagon, or other resuscitative actions and were documented as serious AEs (SAEs). Nonsevere hypoglycemic events were not documented as AEs.
Incidence of hypoglycemia events was the number of SMBG measurements >30 minutes apart. Event rate was the number of SMBG measurements >30 minutes apart across length of exposures, corresponding to incidence divided by number of days of exposure (eg, 9 SMBG measurements >30 minutes apart over 90 days exposure is an event rate of 10%).
Participants were instructed to take fingerstick SMBG measurements using study-provided glucose meters if they experienced symptoms of hypoglycemia or CGM readings <70 mg/dL. To inform treatment decisions, participants were instructed to obtain an SMBG value if CGM readings did not match their symptoms or expectations (as required by CGM per label) 20 or they suspected diabetic ketoacidosis (DKA) accompanied by a CGM value >250 mg/dL.
AEs
Adverse events were recorded throughout the study and defined as any untoward medical occurrence or clinical investigation (not necessarily causally related to study treatment) occurring after enrollment and during the 90-day study. For each AE, a description of the event, start and end date, seriousness, severity (grading scale available in Supplementary Material), action taken, and relationship to study drug were recorded. AEs were followed until resolved or stable, and outcomes were documented. AEs of special interest (AESIs) were clinically relevant decline in pulmonary function (>20% decline from baseline in percent predicted FEV1 or respiratory symptoms not associated with upper respiratory infection), hypersensitivity reactions (including anaphylaxis), and DKA.
Statistical Analysis
Descriptive statistics (n, mean, SEM or SD, range) were calculated for baseline and end-of-study variables assessing efficacy and safety. Frequency counts and percentages were generated for categorical data. No imputation method was applied for missing postbaseline HbA1c or FEV1 measurements, and participants who prematurely discontinued the study were excluded from analyses. Box plots of FEV1 data were used to visually assess variations in measurements across participants. A paired
Results
Participants
A total of 33 participants consented to participate; however, 4 did not meet inclusion criteria, and 1 withdrew from the study prior to randomization (Supplemental Figure 1). Of 28 randomized participants, 26 performed the in-clinic standardized meal challenge, while 2 were excluded owing to blood glucose levels >300 mg/dL. A total of 24 participants completed the final study visit ≥90 days after baseline. Population includes the 24 participants that completed 90 days of treatment. Participants were predominantly white and female, with a mean age of 45 years and mean AID use time of 3 years (Table 4).
Baseline Characteristics.
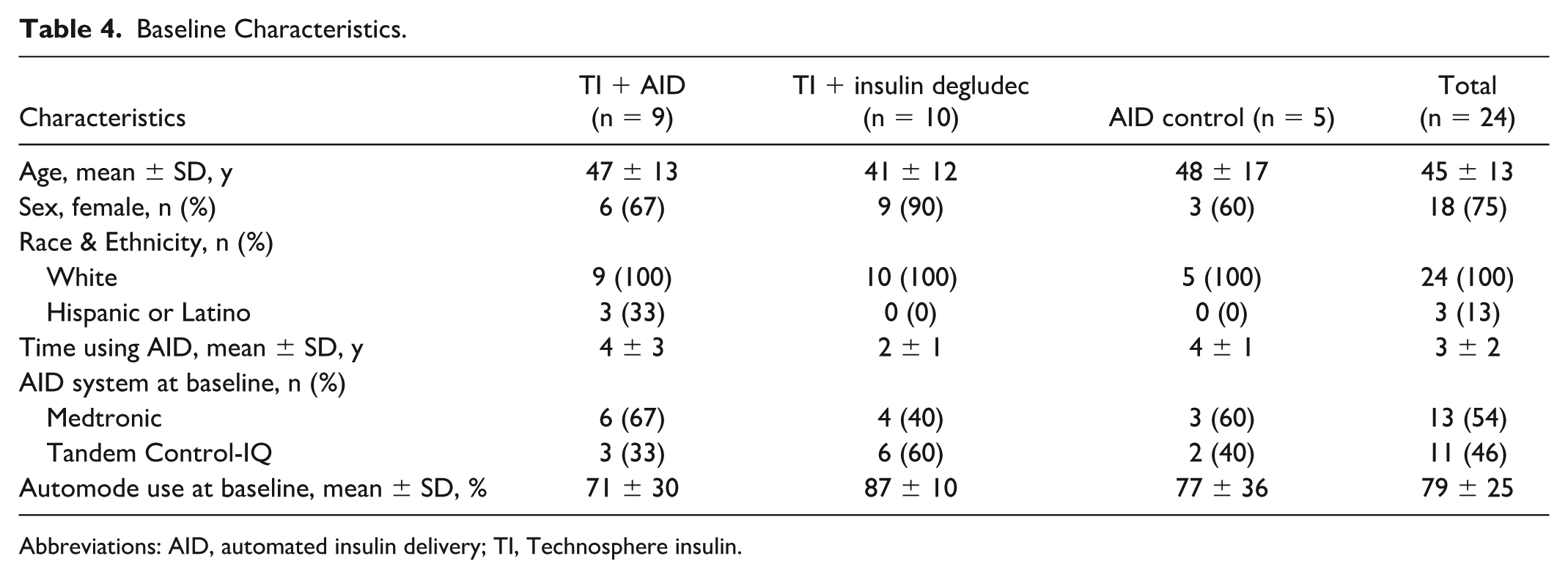
Abbreviations: AID, automated insulin delivery; TI, Technosphere insulin.
HbA1c
All groups demonstrated comparable decline in HbA1c from baseline to end of study (Figure 2). Mean ± SEM change from baseline was −0.3 ± 0.18 for TI + AID and −0.4 ± 0.18 for TI + insulin degludec vs −0.5 ± 0.13 for the AID control group (
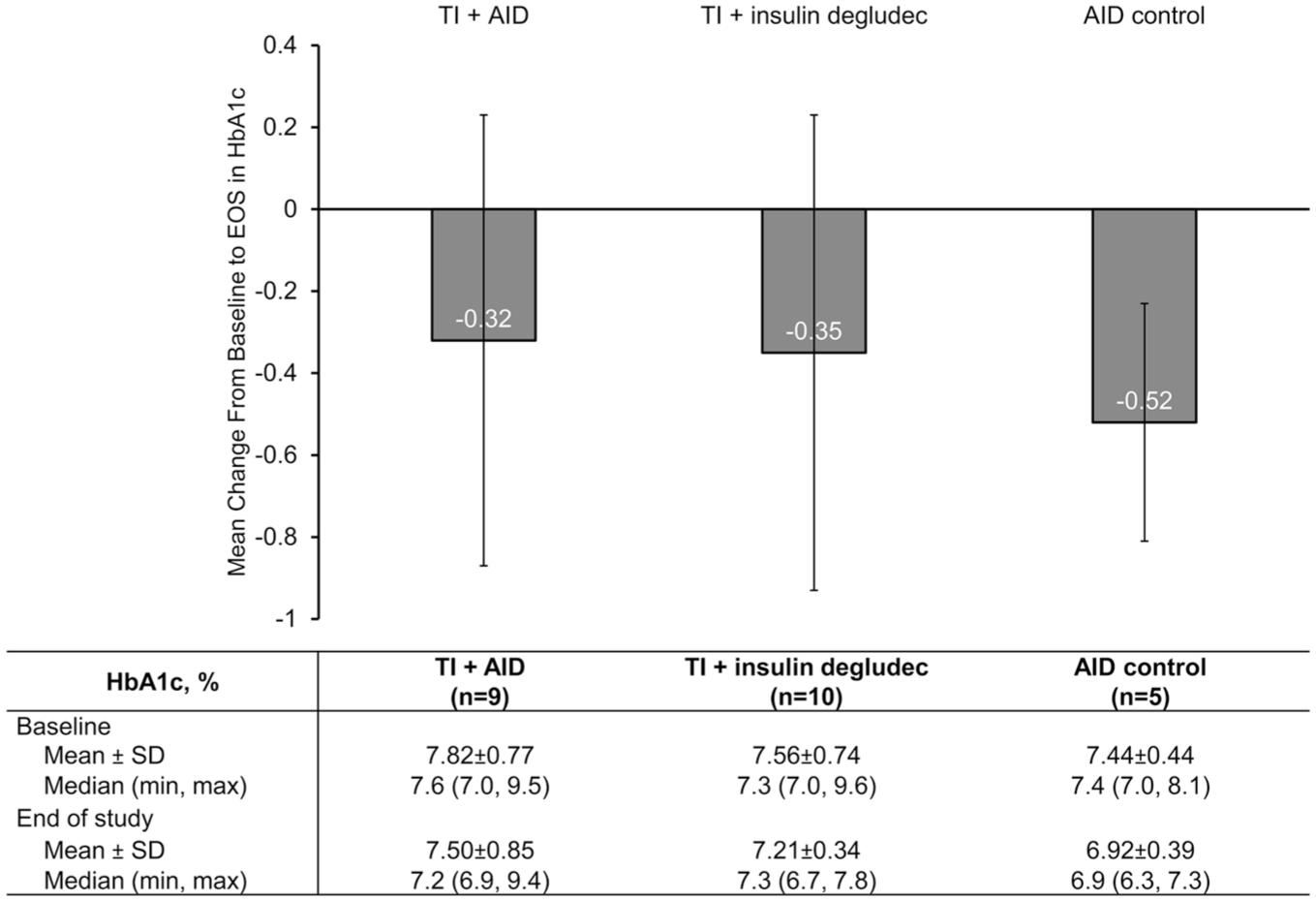
Mean change in HbA1c from baseline to EOS (90 days). Error bars are SEM.
TI Dosing in Patients Switching From AID to TI
In the TI + insulin degludec group, the median percent of bolus insulin over total insulin (bolus + basal) was estimated to be ≈59% (equivalent to ≈1.5:1 bolus-to-basal insulin) and ≈60% (≈1.5:1 bolus-to-basal insulin) at the start and end of study, respectively. The end-of-study minimum and maximum percent of bolus (in TI units) to basal insulin was ≈41% (≈0.7:1 bolus-to-basal insulin) and ≈82% (≈4.4:1 bolus-to-basal insulin), respectively. Supplemental Table 1 shows insulin dosing information at baseline and end of study.
Lung Function
The TI + AID group demonstrated a mean ± SD decrease in percent predicted FEV1 of 2% ± 5%, while the TI + insulin degludec presented a slight increase of 2% ± 6%. The AID control group had a mean decrease of 2% ± 6%. No statistically significant differences were observed from baseline to end of study within or between groups (Table 5, Figure 3a). Comparing all patients treated with TI with those in the AID control group revealed no statistically significant difference in change from baseline to end of study (Figure 3b).
Percent Predicted FEV1 by Group.
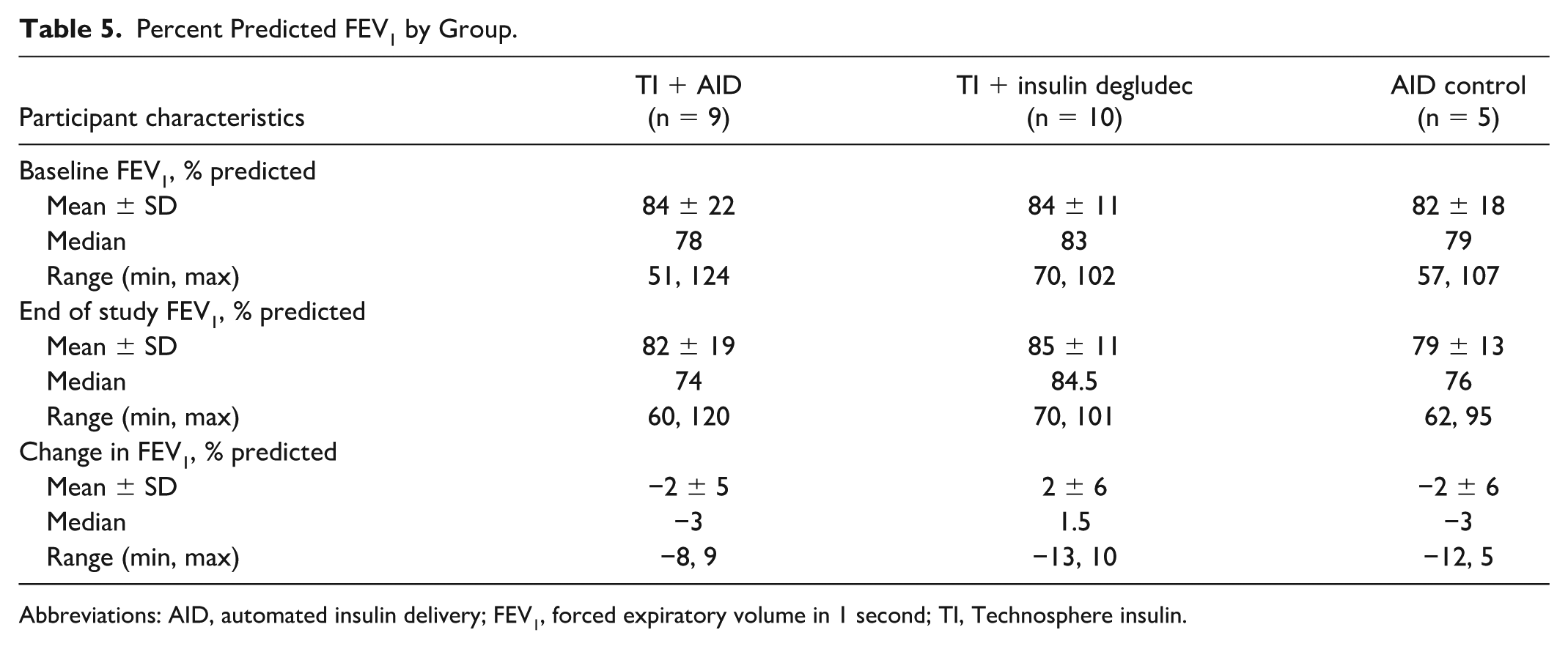
Abbreviations: AID, automated insulin delivery; FEV1, forced expiratory volume in 1 second; TI, Technosphere insulin.
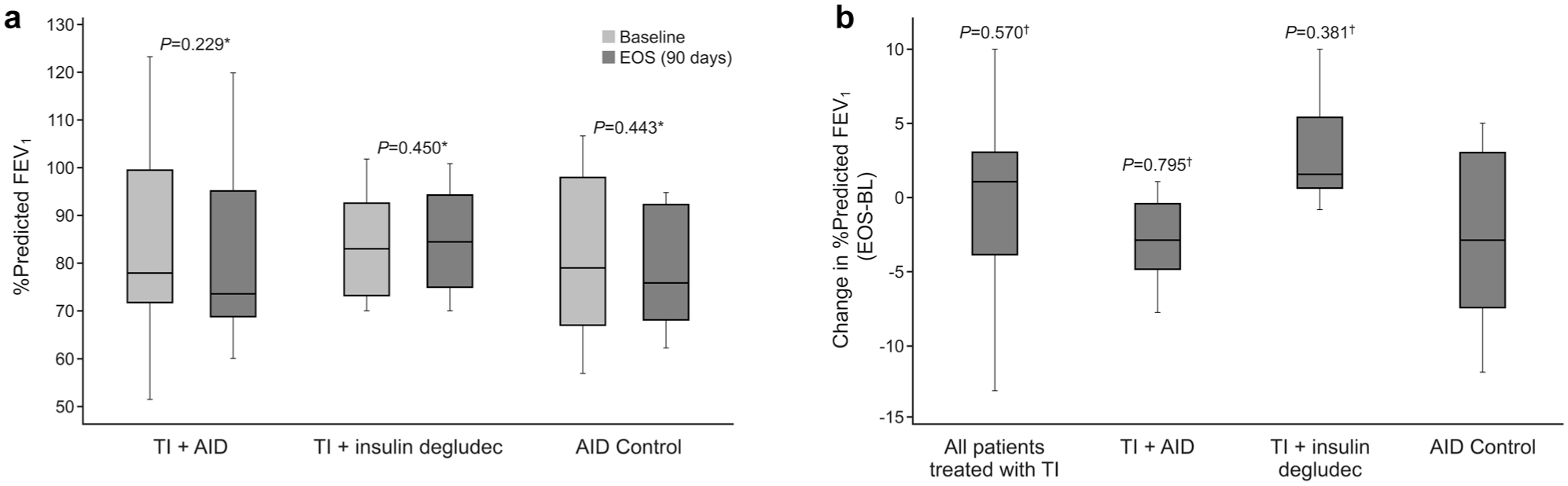
Boxplots of % predicted FEV1 at BL and EOS (90 days) (a) and change in % predicted FEV1 from BL to EOS (90 days) (b). *Paired
Hypoglycemia Events
Mean incidence of hypoglycemic events <70 mg/dL ranged between 13 and 20 across the 3 groups. Mean event rate ranged between 13 and 21% (Table 6). For hypoglycemic events <54 mg/dL, mean incidence ranged between 3 and 5, with a mean event rate of 3 to 6%. There were no statistically significant differences between groups in incidence or event rate of hypoglycemia <70 mg/dL or <54 mg/dL based on SMBG measurements (all
Hypoglycemia Incidence and Event Rate.
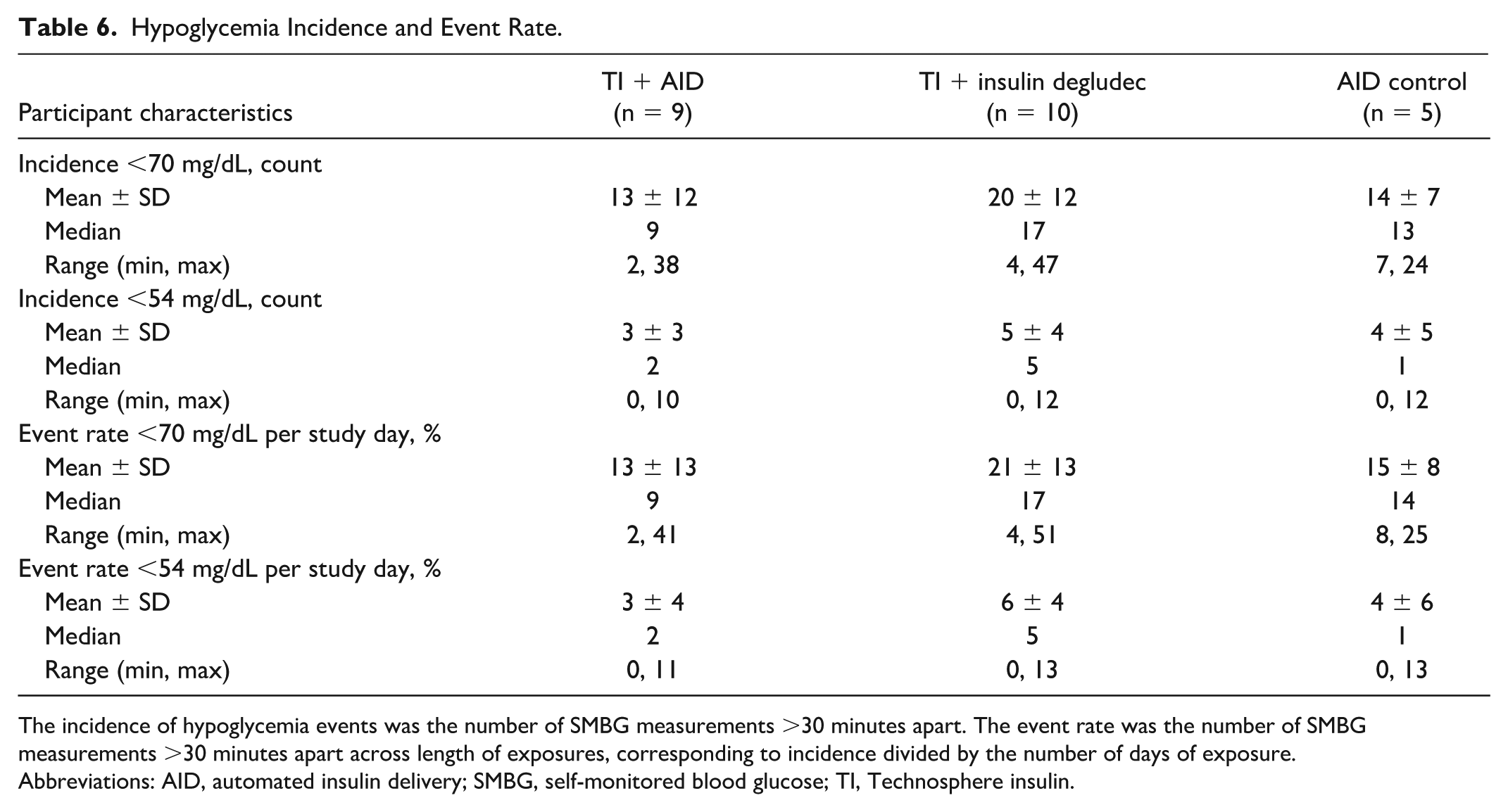
The incidence of hypoglycemia events was the number of SMBG measurements >30 minutes apart. The event rate was the number of SMBG measurements >30 minutes apart across length of exposures, corresponding to incidence divided by the number of days of exposure.
Abbreviations: AID, automated insulin delivery; SMBG, self-monitored blood glucose; TI, Technosphere insulin.
CGM Metrics.
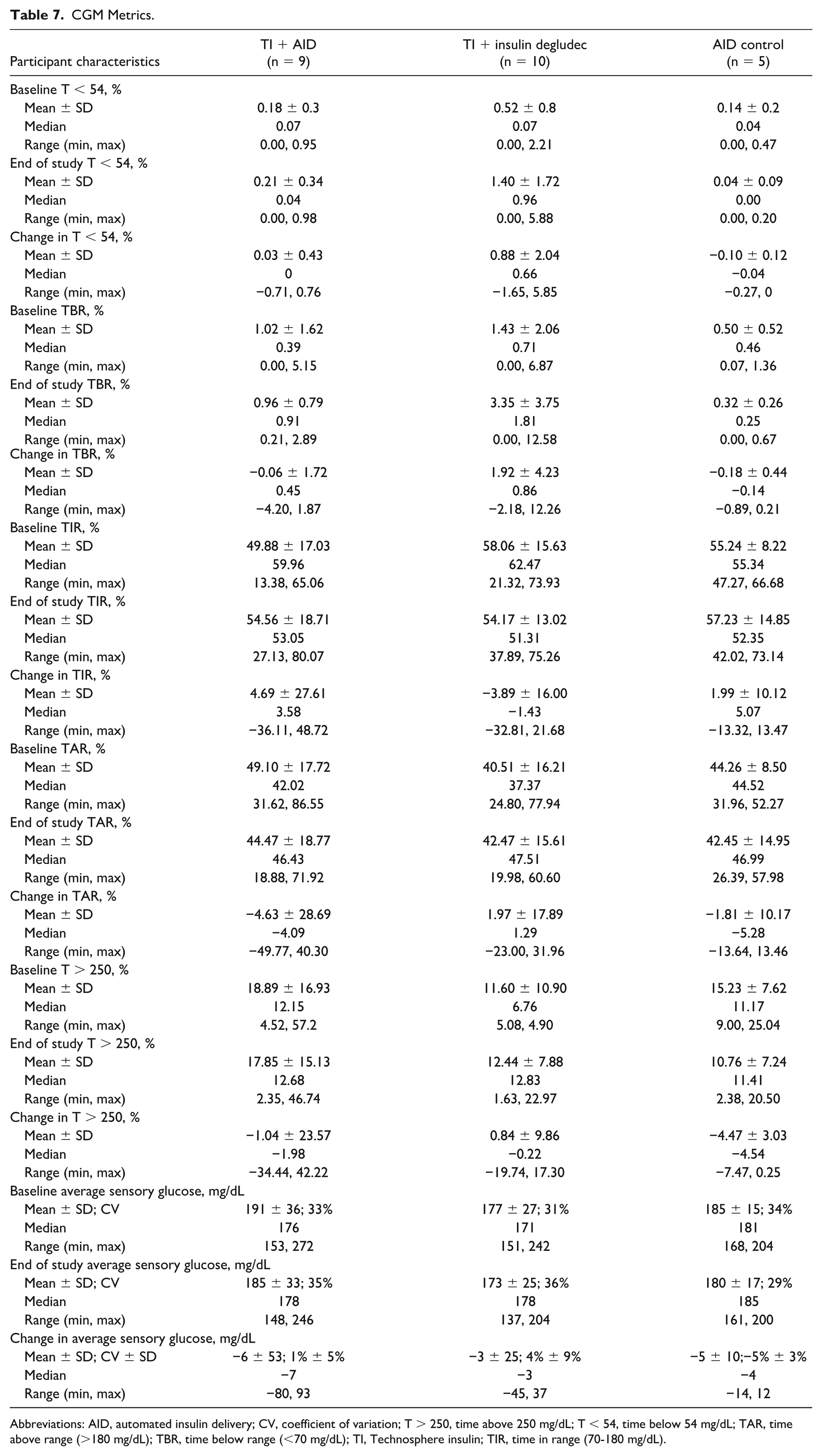
Abbreviations: AID, automated insulin delivery; CV, coefficient of variation; T > 250, time above 250 mg/dL; T < 54, time below 54 mg/dL; TAR, time above range (>180 mg/dL); TBR, time below range (<70 mg/dL); TI, Technosphere insulin; TIR, time in range (70-180 mg/dL).
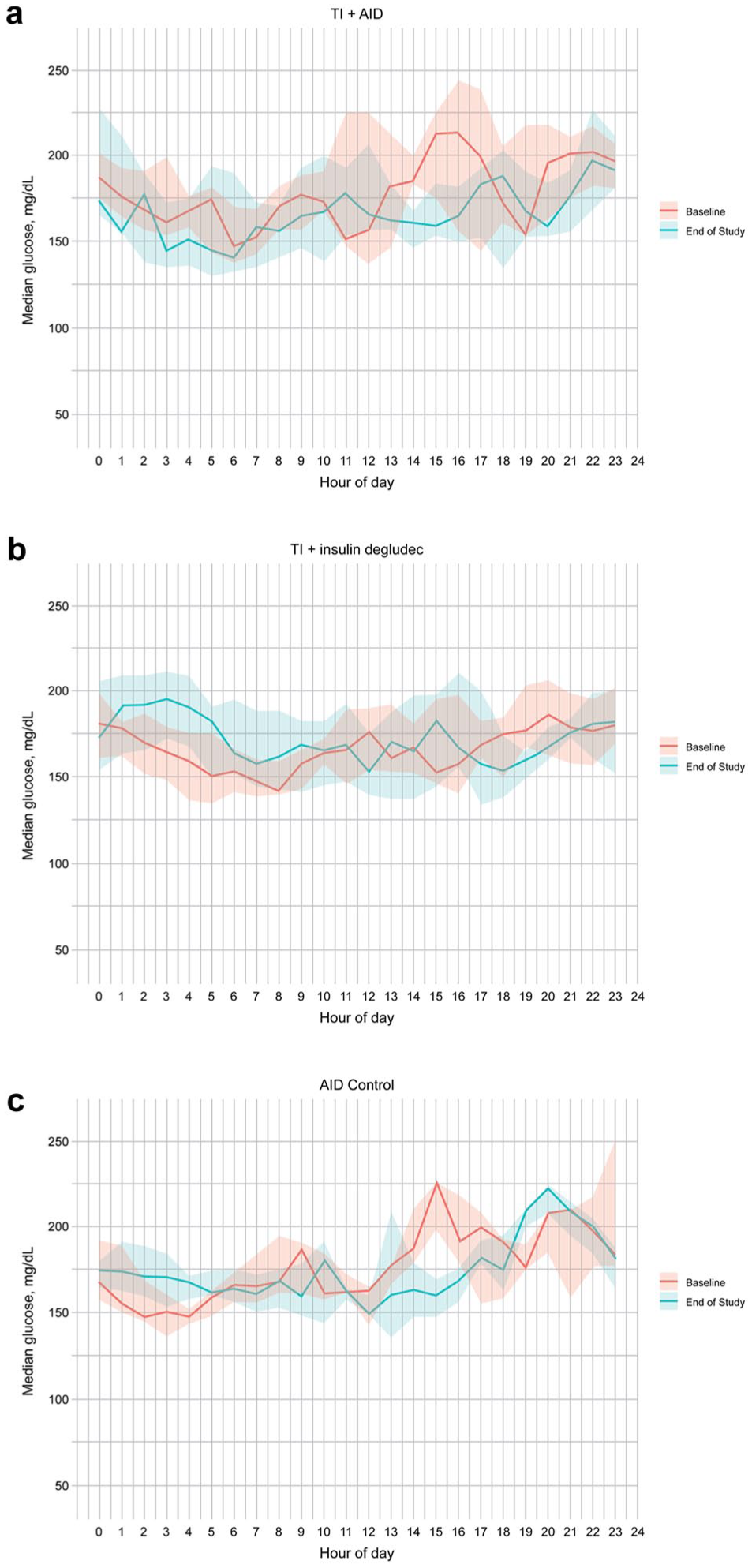
Aggregated median 24-hour CGM data over 10 days at baseline (red) and end of study (blue) by treatment group (a) TI + AID, n = 9; (b) TI + insulin degludec, n = 10; and (c) AID control, n = 5. Shaded regions represent the interquartile range.
AEs
A total of 48 AEs were reported, including 3 SAEs and 3 AESIs (Table 8). Of these, 4 participants had AE(s) deemed related to TI (total of 7 events), which included cough (2 participants), sore throat, decline in pulmonary function, throat pain, abnormal breathing sensation, and wheezing. Two of these events, clinically relevant decline in pulmonary function and wheezing, were classified as AESIs and resulted in study withdrawal. A third AESI was a DKA event considered unrelated to TI as the participant had not been taking TI for several days ahead of its onset. No SAEs were deemed related to TI. An AE of wheezing was reported as an AESI by a site investigator that did not fit the predefined study definition for an AESI.
Summary of AEs.
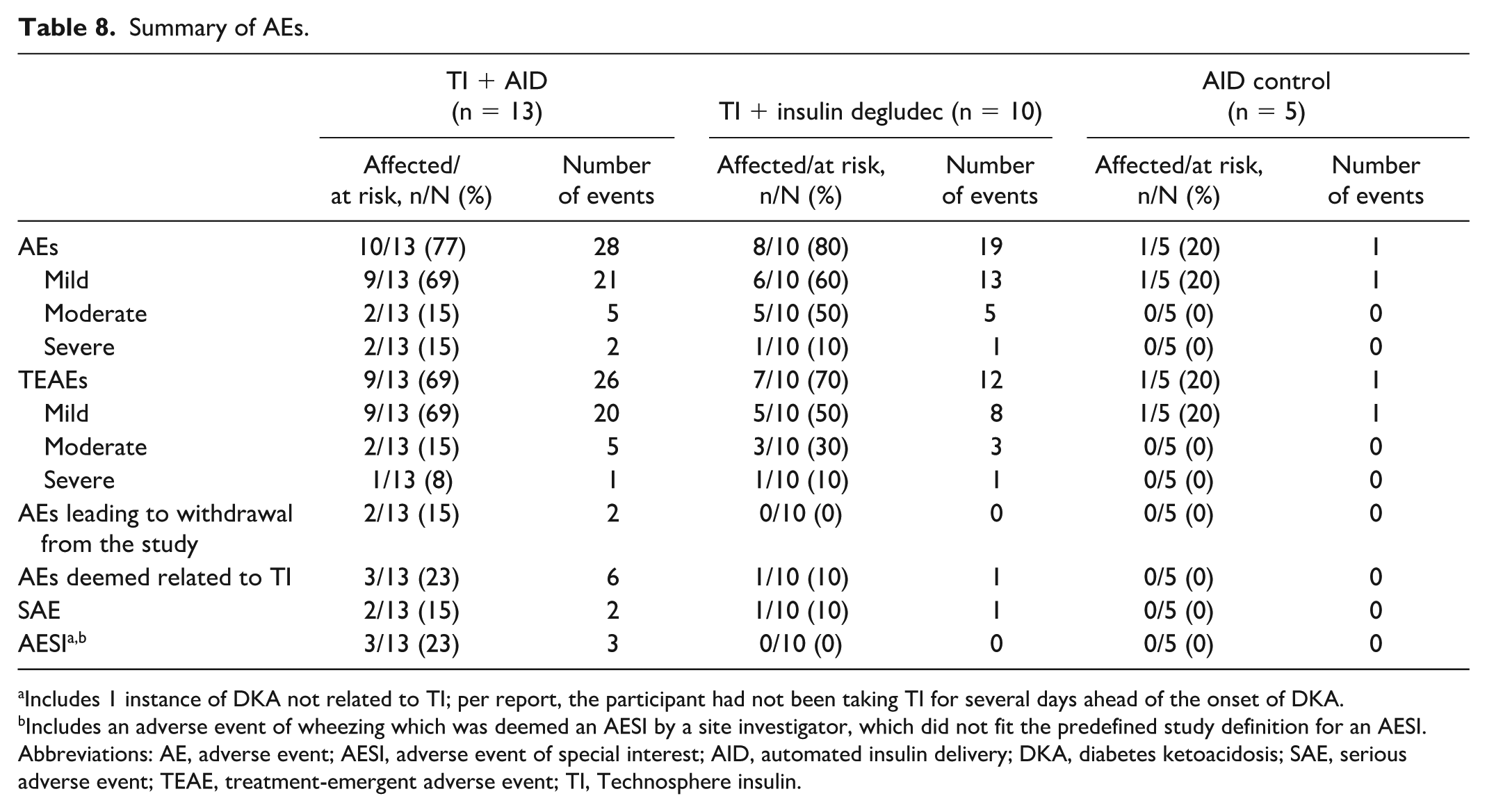
Includes 1 instance of DKA not related to TI; per report, the participant had not been taking TI for several days ahead of the onset of DKA.
Includes an adverse event of wheezing which was deemed an AESI by a site investigator, which did not fit the predefined study definition for an AESI.
Abbreviations: AE, adverse event; AESI, adverse event of special interest; AID, automated insulin delivery; DKA, diabetes ketoacidosis; SAE, serious adverse event; TEAE, treatment-emergent adverse event; TI, Technosphere insulin.
Discussion
This study demonstrates that patients taking TI + insulin degludec or TI + AID, at doses calculated using the higher modified conversion shown in Table 3 and titrated to glycemic effect, achieved glucose-lowering effects comparable with patients who continued using their AID systems, yet only the AID group was significant in lowering HbA1c. Although hypoglycemia risk was higher with TI + insulin degludec, this was primarily driven by a single patient. These findings suggest that the lower conversion dose recommended in the current USPI is likely insufficient for achieving glycemic control. Lung function changes were similar between groups, and observed mean decreases align with expectations for this study population.
Despite advances in glucose monitoring and insulin delivery systems, achieving HbA1c goals and managing the risk of late postprandial hypoglycemia remain key challenges with SC RAAs. 6 A prior phase 3 study of TI in participants with T1D (N = 375) demonstrated significantly lower rates of hypoglycemic events 2 to 6 hours after meals compared with SC mealtime insulin. 11 The fast onset of action and rapid elimination of TI, combined with the higher modified conversion dosing used in this study, may be a promising approach to mitigate the risk of late postprandial hypoglycemia associated with SC RAA.
Consistent with previous studies,13,21 TI had no significant effect on lung function. AEs experienced by the participants were mild and expected, and, as anticipated, 12 cough was one of the most common AEs. However, based on clinical experience, cough does not usually deter patients from using TI, and practical guidance from clinicians may help minimize cough. One participant experienced DKA deemed unrelated to TI; however, it should be noted that when using AID, any insulin dosed independent to the pump is not accounted for by the algorithm and may impact glucose control.
Overall, supplementing AID with TI was successful. However, certain nuances related to pump algorithms, such as whether the pump system used total daily dose in its AID algorithm (Medtronic 770G system) or not (Tandem Control-IQ), were observed during the study that should be considered in future research or clinical use. In the TI + AID group, total daily dose values decreased because TI doses, being external to the pump, were not included in the total daily dose calculation. Consequently, when these participants began covering their meals with TI, they may not have received adequate levels of basal and automated correction bolus coverage. Therefore, future studies should focus on comparing TI plus long-acting insulin to AID systems, in addition to examining TI + AID, until AID algorithms can incorporate the time-action profile of inhaled insulin.
Replacing AID with TI in the TI + insulin degludec group resulted in a median bolus insulin percentage of ≈60% of total insulin (bolus + basal) at the end of the study, equivalent to a bolus-to-basal ratio of ≈1.5:1. The observed bolus-to-basal range was ≈41% to ≈82%, with a maximum recorded ratio of ≈4:1. In contrast to injectable insulin, where typical bolus-to-basal insulin ratio in well-controlled patients is 1:1,1,22 a higher ratio is expected for patients using TI (≈2:1 to 4:1). Of note, the percentage of bolus-to-basal insulin may vary, but clinical studies have shown that many patients require basal insulin be <50% of their total daily dose.23,24
Limitations of this proof-of-concept study include small sample size and absence of formal sample size calculations. Due to the limited sample size, any nonprotocol lifestyle behaviors (eg, change from a sedentary routine to a more active lifestyle) may have had a larger effect on the study results, potentially presenting as the control group numerically outperforming the TI groups. Further studies with larger populations and longer durations are warranted to build on these pilot results.
Conclusions
This proof-of-concept study demonstrated efficacy and safety in patients using TI added to an AID system for mealtime control or combined with daily SC basal injection for glycemic control. No new safety signals were identified for TI. Considering that a higher modified dose conversion was used for the initial TI dose in this study, these proof-of-concept data also demonstrate that the higher conversion dose with titration may be needed for achieving glycemic control.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251388128 – Supplemental material for Replacing or Supplementing Automated Insulin Delivery With Inhaled Insulin: A 90-Day Randomized Controlled Trial
Supplemental material, sj-docx-1-dst-10.1177_19322968251388128 for Replacing or Supplementing Automated Insulin Delivery With Inhaled Insulin: A 90-Day Randomized Controlled Trial by Kevin B. Kaiserman, Johanna Ulloa, Jennifer Pleitez, Joseph Sylvan, Kevin Codorniz, Scott Lee, Christopher Jacobson and Thomas Blevins in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
Medical writing support was provided by Sanjula Wickramasinghe, PhD, from The Curry Rockefeller Group, LLC, a Citrus Health Group, Inc., company (Chicago, Illinois), and was funded by MannKind Corporation (Danbury, Connecticut).
Abbreviations
AE, adverse event; AESI, adverse event of special interest; AID, automated insulin delivery; CGM, continuous glucose monitor; DKA, diabetic ketoacidosis; FDA, US Food and Drug Administration; FEV1, forced expiratory volume in 1 second; G6 Pro, Dexcom G6 Professional; HbA1c, glycated hemoglobin; RAA, rapid-acting insulin analog; SAE, serious adverse event; SC, subcutaneous; SMBG, self-monitored blood glucose; TI, Technosphere insulin; USPI, United States prescribing information.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KBK, JP, and JS are employees and stockholders of MannKind Corporation. JU was an employee and stockholder of MannKind Corporation at the time the research was conducted. KC and SL have no relevant disclosures. CJ served as an advisory board consultant for MannKind Corporation (2022) and is currently an advisory board consultant and on a speakers bureau for Medtronic. TB serves as an advisory board consultant for MannKind and Insulet and on speakers bureaus for Lilly, Novo Nordisk, Boehringer Ingelheim, Sanofi, and AstraZeneca and receives clinical research support from Lilly, Novo Nordisk, Insulet, Medtronic, MannKind, Tandem, and Boehringer Ingelheim.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by MannKind Corporation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
