Abstract
Background:
Despite rapidly evolving diabetes technology and evidence that early access to technologies improves outcomes, there is disparate use based on socio-demographic factors. We sought to characterize technology uptake in the year following diagnosis of type 1 diabetes in youth based on race/ethnicity, insurance, and household structure.
Methods:
We conducted a retrospective cohort study of 692 youth diagnosed with and treated for type 1 diabetes between 2016 and 2020 at a children’s hospital. Medical record review provided outcomes of interest, including time to initiation of continuous glucose monitors (CGMs), insulin pump therapy, or both. We used cumulative incidence curves and competing risks regression to compare time to initiation by socio-demographic groups.
Results:
There were 692 youth, 59% male, diagnosed at a mean age of 10.8 (±4.2) years, in the sample. The majority (83.2%) were White, English-speaking (94.4%), and privately insured (76.7%), with 71.7% living in two-parent households. Cumulative incidence curves and competing risks regression showed that publicly insured youth had a lower likelihood of starting diabetes technologies in the year following diagnosis than privately insured youth. Black and Hispanic youth were less likely than white youth to start CGM and insulin pumps. Youth from non-intact households similarly exhibited lower rates of technology uptake. Hazard ratios for time to both technologies were comparable to those for time to insulin pump.
Conclusions:
These findings highlight the importance of developing interventions to advance diabetes technology use from onset of type 1 diabetes for all youth.
Keywords
Introduction
Advanced diabetes technologies including continuous glucose monitors (CGMs), insulin pump therapy (continuous subcutaneous insulin infusion [CSII]), and dual technology use with the potential for automated insulin delivery (hybrid closed loops [HCLs]), are part of routine care for many pediatric patients with type 1 diabetes (T1D). Current national 1 and international guidelines2,3 advise early introduction of these devices where available for optimal care for diabetes in youth. Many studies have demonstrated improved biomedical and psychosocial outcomes with the use of new technologies.4,5 Recent literature suggests that these technologies may be both acceptable and desirable at diagnosis or within the first few months of living with T1D and, furthermore, that their early introduction may optimize impact on outcomes.1,6,7
Despite the well-established benefit of diabetes technology for care of T1D in youth, to date, there remain substantial disparities in various subgroups with respect to access to such devices.8 -10 Data from the T1D Quality Improvement Exchange published in 2023 demonstrate that although population rates of CGM, CSII, and HCL use have increased significantly from 2016 to 2022, disparities in technology access on the basis of race/ethnicity and insurance type have persisted, as have outcome disparities as measured by hemoglobin A1c (HbA1c). 11 There are limited published data on the impact of family structure on diabetes device uptake; however, one qualitative study from 2022 cites the impact of family support systems on T1D care and capacity to manage diabetes technology for black adolescents. 12 Despite the growing literature on disparities in access to and use of such devices in certain socio-demographic groups,8 -10 there is a need to evaluate how early in the disease process such disparities develop.
Methods
Electronic medical records (EMRs) of all patients diagnosed with and receiving initial care for T1D at Boston Children’s Hospital (BCH) from January 1, 2016, through December 31, 2020, were systematically reviewed. Inclusion criteria included ages 2-25 years and longitudinal outpatient care at BCH for at least one year following diagnosis. Youth with complex medical conditions, such as cancer, organ transplant, or cystic fibrosis, were excluded. The EMR provided socio-demographic data; youth with “International” or missing insurance data and those not self-identified as “White,” “Black,” or “Hispanic” were excluded.
Systematic EMR review included collection of demographic information, including family structure, obtained from standardized social work evaluation at diagnosis. Determination of the diagnosis and its date of onset were made via manual chart review of clinical documentation to confirm data generated from an EMR data pull of first use of the International Classification of Diseases codes for T1D. Review of clinical care documentation from diagnosis characterized type of initial insulin regimen and review of clinician documentation from diagnosis through one year determined date of initiation of CGM and/or CSII. A device was considered “initiated” if the clinician documented data from the device in the clinical record (ie, CGM time-in-range or CSII settings). Youth were classified as “dual technology users” on the day they initiated their second device.
Summary statistics (mean, SD, frequencies) described the cohort. Chi-square statistics for frequency distributions assessed independence of demographic variables. Analyses included cumulative incidence analysis and competing risks regression models (cmprsk functions cuminc and crr) by subgroup, and differences among groups were assessed with Gray’s Test for cumulative incidence curves and Wald test for subdistribution hazard ratios (HRs).
R (4.4.2) was used for statistical analysis with tidyverse (2.0.0), tidyquant (1.0.7), rstatix (0.7.2.), and cmprsk (2.2-12) packages.
Results
Study Population and Time to Initiation of Diabetes Devices
The cohort included 692 youth (41.0% female) with mean age at diagnosis of 10.8 (±4.2) years (Table 1). The cohort was primarily White (83.2%), English-speaking (94.4%), and privately insured (76.7%). Public and private insurance rates differed among race subgroups with 85% of White youth covered by private insurance compared to 36% of Black youth and 38% of Hispanic youth (chi-squared = 123, df = 2, P < .01). The majority of these newly diagnosed youth with T1D lived with two parents in one household (71.7%). Among youth in intact households, 84% were privately insured compared with 56% in non-intact households (chi-squared = 51, df = 1, P < .01). There were significantly more non-White youth living in non-intact households (chi-squared = 47.9, df = 2, P < .01) (see Table 2).
Baseline Characteristics of Diagnosis Cohort.

HbA1c: hemoglobin A1c; DKA: diabetic ketoacidosis.
Continuous variables are summarized as mean (standard deviation). Categorical variables are summarized as % within category.
“Unknown” designates that the field was completed but designated as “unknown.”
“Diverse” refers to a combined group of “Hispanic” and “Black” subjects.
If there was not an EMR HbA1c value within 90 days of diagnosis, it was characterized as missing. Nine subjects (1.3%) did not have HbA1c data.
“Unspecified” designates that the chart was reviewed for the categorical data, but the data were not present in the available documentation.
Individuals with “Unspecified” family structure were removed from the binary categorization.
Baseline Characteristics of Diagnosis Cohort by Subgroup.
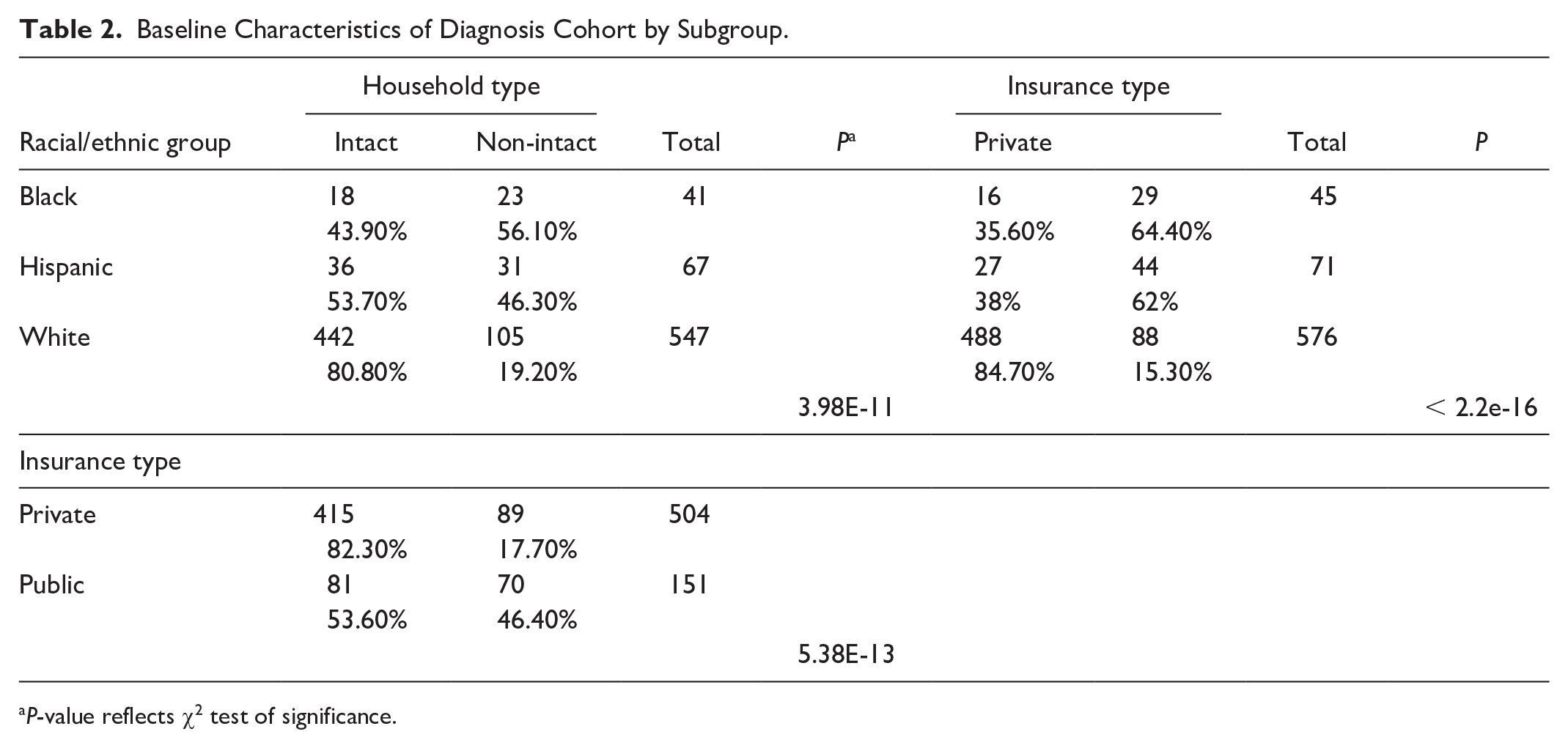
P-value reflects χ2 test of significance.
By the end of one year following diagnosis, 69% of youth were using at least a CGM, 29% at least CSII, and 26% were using a CGM and CSII (referred to as dual technology). Among those who had started technologies within the year following diagnosis, median time to CGM initiation was 110 (interquartile range [IQR] = 55-169) days, while median time to CSII start was 218 (173-285) days.
Cumulative incidence analyses demonstrated longer time to CGM, CSII, and dual technology initiation among publicly vs privately insured youth, Black or Hispanic vs White youth, and those living in non-intact (ie, two parents living in two separate houses, single parent, or a non-parent primary caregiver) vs intact households.
Odds of Starting Diabetes Devices in the First Following Diagnosis by Insurance Status, Race/Ethnicity, and Family Structure
Insurance
Publicly insured youth were 27% less likely to start CGM than privately insured youth (HR = 0.73; 95% confidence interval [CI] = 0.60, 0.91; P = .004) (Figure 1a). The likelihood of initiating pump therapy and dual CGM/pump therapy was 61% and 65%, less for publicly versus privately insured youth, respectively (HR = 0.39; 95% CI = 0.25-0.59; P < .001 for pump therapy and HR = 0.36; 95% CI = 0.23-0.57; P < .0001 for dual CGM/pump therapy) (Figure 1a). By the end of the year following diagnosis, only 12.4% of publicly insured youth were dual technology users (HR = 0.36; 95% CI = 0.23-0.57; P < .0001) compared with 30.7% of privately insured youth.
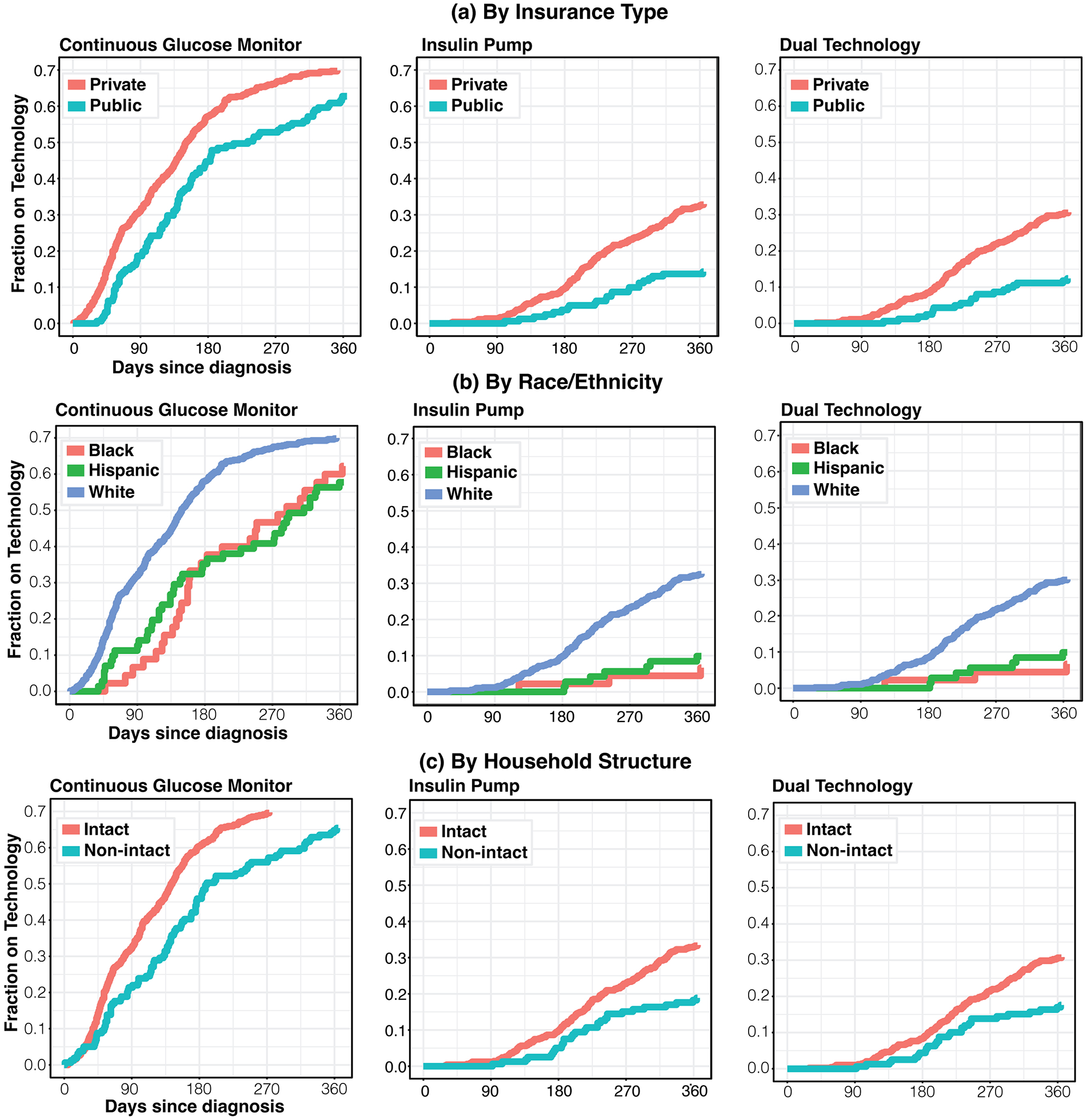
Cumulative curves demonstrate that significant differences in time to technology start in the year following diagnosis are present among subgroups. Privately insured youth, White youth, and those from intact families receive technology earlier in their disease course than their counterparts.
Race/ethnicity
Black (HR = 0.64; 95% CI = 0.46-0.88; P = .0062) and Hispanic (0.62; 95% CI = 0.46-0.83; P = .0013) youth were 36% and 38%, respectively, less likely to start CGM in the year following diagnosis than their White counterparts (Figure 1b). In addition, black (HR = 0.17; 95% CI = 0.056-0.54; P = .003) and Hispanic (HR = 0.26; 95% CI = 0.12-0.55; P = .0004) youth were 83% and 74%, respectively, less likely to start CSII within year one after diagnosis (Figure 1b). Only 6.7% of Black (HR = 0.29; 95% CI = 0.14-0.61; P < .01) and 9.9% of Hispanic (HR = 0.19; 95% CI = 0.06-0.60; P < .01) youth were dual technology users at the end of the first year compared with 30.0% of White youth (Figure 1b).
Household structure
Household structure was dichotomized as “intact” (two parents, one house) vs non-intact. Youth living in “Unspecified” house structure (37) were removed from this analysis. Youth with T1D living in non-intact households were 28% less likely to start CGM (HR = 0.72; 95% CI = 0.58-0.89; P = .002) and 48% less likely to start CSII (HR = 0.52; 95% CI = 0.35-0.77; P < .001) than their counterparts in intact households (Figure 1c). Nearly one third (30.8%) of youth in intact households were dual technology users by the end of year one compared with 17.6% in non-intact households (HR = 0.53; 95% CI = 0.35-0.79; P = .002) (Figure 1c).
Competing Risks Regression Model: Subgroup Analysis
In a combined model incorporating insurance at diagnosis, race/ethnicity group, and household structure, insurance and household structure were not significant predictors of time to CGM initiation (public insurance HR = 0.91; 95% CI = 0.72-1.15, P = .40; non-intact household HR = 0.81; 95% CI = 0.65-1.02, P = .069), but race/ethnicity group remained significant (Hispanic HR = 0.68; 95% CI = 0.50-0.93; Black HR = 0.71; 95% CI = 0.50-1.01; P = .019). In an analogous model for time to pump initiation, both insurance type and race/ethnicity group maintained significance with P = .031 and .003, respectively, while family structure was not significant (P = .088). The same was true for the regression model assessing subdistribution hazards for time to dual technology use (insurance type P = .014, race/ethnicity group = .013, and family structure = .13).
Discussion
In our retrospective cohort of 692 youth diagnosed with T1D between 2016 and 2020 and cared for at a tertiary care children’s hospital, there were significant disparities in time to initiation of current standard-of-care diabetes technologies, including CGM and CSII, according to socio-demographic factors. Publicly insured and non-White youth were significantly less likely to initiate either device. Furthermore, 2.5 times more youth with private insurance compared with those with public insurance were using both CGM and CSII within the first year of diabetes diagnosis, and nearly 3.5 times more non-Hispanic White youth vs Black or Hispanic youth were dual technology users within that same time frame. Youth with T1D who lived in intact households were also more likely to initiate CGM, CSII, and the combination of both technologies than those in non-intact households. Combined models confirmed that race/ethnicity was a consistent and potent predictor of time to any technology initiation; notably, insurance type was not associated with time to CGM start when adjusting for race/ethnicity group and family structure.
A significant body of literature has demonstrated that the year following diagnosis with T1D sets the trajectory for a person’s life with diabetes. The HbA1c trends are established, 13 as are disparities in metabolic management based on social disadvantage. 14 Differing experiences with diabetes technology7,15,16 likely play a major role in these disparities, and multiple studies have identified the potential of CSII to improve metabolic management across the lifetime when initiated soon after diagnosis with T1D. 17 American Diabetes Association Standard of Care Guidelines for 2025 include considering initiation of CGM at or shortly after diagnosis. 1 The CGM initiation at diagnosis has become standard of care at many pediatric centers, along with the recommendation to offer CSII and/or HCL to all youth and families able to use such technologies. Recent studies suggest that early implementation results in durable use of technology and improved glycemic outcomes.18 -20
While prior studies have identified similar disparities in the implementation of CSII among socially disadvantaged youth with T1D9,15,21 and the persistence of these disparities over time, 10 our study adds to this literature by demonstrating that publicly insured, non-White, and youth from non-intact households continued to display lower rates of device initiation within the year following diagnosis, while overall rates of technology use have increased. 22 Although insurance type in a combined model no longer predicted CGM use, most youth in this study were Massachusetts residents, where public insurance has historically had relatively generous coverage of CGM devices for youth with T1D.23,24
Furthermore, with increased use of HCL systems, it is important to consider not only implementation of CGM or CSII but also the concomitant use of both technologies. As HCL systems have become more available and impactful in the pediatric population in the years since these data were generated, it is imperative that individuals are able to adopt both technology types (CGM and CSII) to experience the benefit of an integrated system. Our study provides a detailed analysis of time to dual technology use among different demographic groups and demonstrates that disparities described in prior studies in CGM and CSII use are recapitulated and often more extreme for dual technology use. Further research is needed to better understand what drives differences in prescribing patterns to youth from different socio-demographic groups and which aspects of social disadvantage may be most effectively targeted to enhance equity. Persistence of significant differences in access to technology among different racial/ethnic groups when controlling for insurance status suggests that programs to enhance equitable technology uptake must expand beyond simply ensuring that both private and public insurers cover diabetes devices for youth.
Limitations of this study include the small sample size of non-White youth; however, the study demographics are largely reflective of population level pediatric data of T1D in the United States. An additional limitation of this study is that it reflects data from a large academic center, which may not be generalizable to all youth with T1D. Future studies should examine technology use at centers that serve more diverse patient populations and at smaller, non-academic programs in other regions of the country. Lack of data regarding reasons why youth/families did not initiate CGM or CSII is another limitation of this study; future qualitative studies among patients and families and prescribers can help capture reasons to explain the observed disparities. Strengths of this study include the large sample size in general, individual-level detail about family structure, and inclusion of multiple types of advanced diabetes technologies.
Conclusions
In conclusion, given the need for both CGM and CSII for automated insulin delivery systems, and their value in optimizing glycemic control while reducing self-care burden, there is an imperative to identify opportunities to increase uptake of these devices across all segments of the pediatric population with T1D as soon as possible after diagnosis to preserve health and reduce future complications of the disease. To mitigate potential biases and the risk of perpetuating disparities in access to life-altering diabetes technologies, this may be most meaningfully achieved via policies that systematically introduce CGM and CSII to all youth with T1D within three to six months of diagnosis.
Footnotes
Acknowledgements
The authors acknowledge research assistants and data manager Sarah Clemons/Radhika Joshi and James Zavadoski, as well as Dr. Grant Tremblay for figure design. Boston Children’s Hospital’s High-Performance Computing Resources Enkefalos 2, installed and configured by BioGrids (eLife 2013;2: e01456, Collaboration gets the most out of software). REDCap secure web platform for data management. Mentors: Drs. Katherine Garvey and Christina Astley, Boston Children’s Hospital.
Abbreviations
BCH, Boston Children’s Hospital; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; CI, confidence interval; EMR, electronic medical record; HbA1c, hemoglobin A1c; HCL, hybrid closed loop; HR, hazard ratio; IQR, interquartile range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health under Award Numbers K12 DK094721 (EST) and K23DK133685 (EST), as well as a Boston Children’s Hospital Health Equity grant (EST).
