Abstract
Artificial pancreas (AP) systems, also called automated insulin delivery systems, have improved the time in range of glucose levels, reduced the daily burden of the user for glucose regulation, and improved their quality of life. Several commercially available AP systems operate in hybrid closed-loop mode that requires manual information from the user for meals and exercise. This article summarizes the progress on mathematical models of glucose-insulin dynamics, continuous glucose monitoring systems, and insulin pumps that form the building blocks of AP systems, the shift from animal studies to in silico clinical trials that accelerated the rate of progress in AP technologies and the efforts for developing the next-generation AP systems, and the fully automated AP that eliminates manual inputs and mitigates the effects of disturbances to glucose homeostasis—meals, physical activities, acute stress, and variations in sleep characteristics. A section is devoted to discuss the unique glycemic management challenges faced by women with diabetes across the lifespan (menstrual cycle, menopause, pregnancy) and summarize progress made to reduce their impact on glycemic management.
Keywords
This article is part of a collection of publications to commemorate the 75th Anniversary of the NIDDK.
The chapter focuses on the development of metabolic models, in silico trials, and control algorithms. The mission of NIDDK is to conduct and support basic, preclinical, and clinical research and to disseminate science-based information on diabetes and other metabolic diseases to improve peoples’ health. In this article, we are celebrating the 75 years of progress enabled by NIDDK to improve treatments and quality of life for people living with type 1 diabetes, focusing on the building blocks that led to the development of the artificial pancreas (AP) system. The commercialization of AP systems was only possible through substantial support from the NIDDK.
Automated insulin delivery (AID) has transformed care for people with T1D, enabling glycosylated hemoglobin (HbA1c) reductions of 0.2% to 0.5% compared with open-loop therapy.1-4 Yet, studies indicate that a majority of people using AID still are not achieving the target of 70% in range (70-180 mg/dL) 5 as recommended by the American Diabetes Association. Multiple possible reasons may cause this, including carbohydrate misestimations, 6 forgetting to dose for meals or dosing after the meal event has occurred, high carbohydrate diets, sedentary, highly active lifestyles, or variations in lifestyles, limited information used by AID systems especially for fully automated AID systems, and shortcomings in AID’s control system logic. This chapter is highlighting the progress made in the journey of AID system development and future directions of progress.
The Early Years: The Building Blocks for Automated Insulin Delivery Systems—Models, Insulin Pumps, and First Continuous Glucose Monitors
Diabetes is a condition known for more than 3000 years. Historical progress of medical knowledge and hypotheses leading to qualitative models 7 provide a strong foundation for recent progress in advanced treatments such as AP (also called AID systems for insulin-only AP systems). Since the discovery of insulin more than a century ago, scientists have tirelessly strived to advance its administration to improve blood glucose concentration (BGC) control in people with diabetes mellitus. The current AP systems represent the culmination of inspired collaboration between physician-scientists, control engineers, and skilled mathematicians over the last 50 years.
The first prototypes of AP systems for closed-loop control (CLC) of BGC, involved recurring intravenous measurements of BGC coupled with intravenous insulin delivery,8-12 resulting in prototypical commercial devices such as the Biostator and the Nikkiso STG-22 systems.13,14 However, these devices, although clearly impractical for daily outpatient use, demonstrated the possibility of closed-loop glucose control.
Advances in technologies to capture information about BGC and insulin pumps were complemented by the development of effective CLC strategies. The emergence of commercially available minimally invasive continuous glucose monitoring (CGM) systems by MiniMed (Medtronic), revolutionized this field that saw the forerunners of subcutaneous-subcutaneous CLC systems for effective glucose control in feasibility studies.15-17
The next set of landmark studies18,19 established the feasibility and efficacy of subcutaneous continuous insulin delivery via insulin pumps in type 1 diabetes (T1D) and paved the way for further research in intravenous glucose sensing coupled with intraperitoneal insulin delivery.20,21 The early real-time CGM devices (Medtronic, Dexcom, Abbott) had high levels of inaccuracies (mean absolute relative differences—MARDs of 15%-21%), 22 especially in the first 24 hours and also in the hypoglycemia ranges. These devices required multiple daily calibrations with fingerstick glucose measurements and experienced glitches with drug interference, most notably, acetaminophen (Tylenol).23,24 In silico simulations determined that a sensor MARD of ≤10% would permit safe, non-adjunctive use of CGM data for insulin dosing. 25 Continuous research and development in the CGM field resulted in factory-calibrated CGMs with MARD <10% that can be used for 10 or 14 days and improved reliability of glucose data from CGM to the CLC systems.
Early insulin pumps included the “Autosyringe” of Dean Kamen, the Mill-Hill pump of John Pickup, and Minimed of Al Mann. The “Backpack” of Arnold Kadish in 1963, provided dual-hormone (insulin/glucagon) delivery. Two pumps: Minimed in the United States and Disetronic (later Ypsomed) in Europe offered pumps with tubes for a number of years. Mann Foundation and Medtronic proposed pumps in peritoneal cavity. A patch pump, OmniPod, was introduced by Insulet. 26
Since 1970s, there has been interest in developing quantitative models of glucose-insulin dynamics. 27 These models were generally detailed physiological models focused on predicting a population average from clinical trial data. They enabled the early work (Figure 1) on the development of model predictive control (MPC) algorithms for AID systems.28,29 Bergman and Cobelli reported the minimal model, with two quasi-linear differential equations: one representing insulin kinetics in plasma and a second representing the effects of insulin and glucose itself on restoration of the glucose after perturbation by intravenous injection. 30
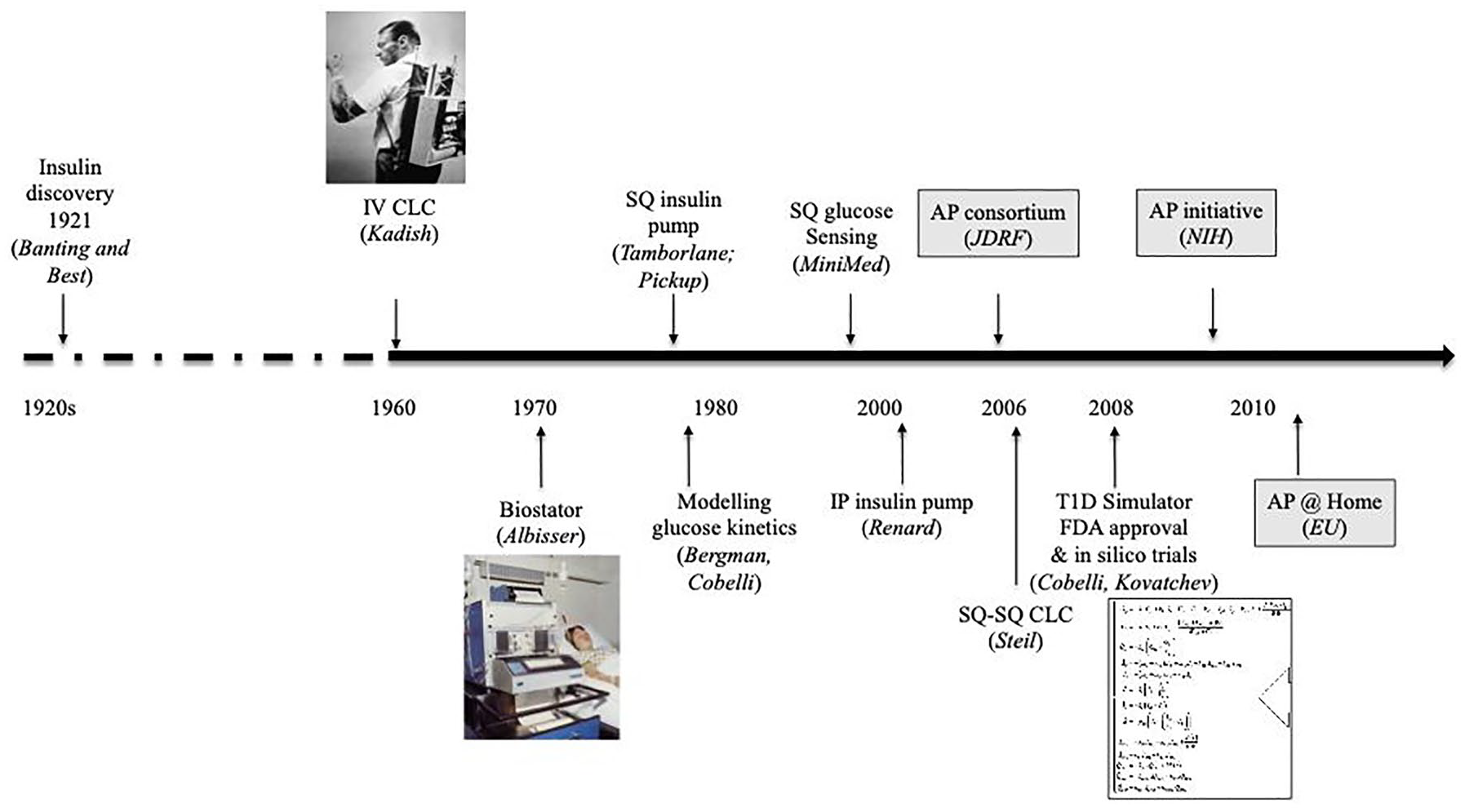
Milestones for AP development, progress, and funding initiatives.
A milestone achievement was the development of the University of Virginia-University of Padova (UVa-Padova) T1D Simulator,31-34 accepted by the U.S. Food and Drug Administration (FDA) in 2008, that was underpinned by extensive dynamic profiling of carbohydrate metabolism in over 200 adults at Mayo Clinic. 35 This enabled rapid in silico testing of CLC algorithms’ performance on virtual patients with different metabolic characteristics, eliminating the need for preclinical animal trials of CLC systems.
The critical aspects of progress in AP systems include iterative refinements of the essential components of the AP system—CGM, controller platform often on a smartphone and the insulin pump—and their connectivity and communication with each other. Early systems were larger and multicomponent (eg, Dexcom Sevenplus CGM; DiAs platform; 36 Medtronic or Tandem t-slim pump) with suboptimal performance and frequent connectivity issues.37,38 Significant progress continues to be made in all these aspects. Recent advances in this century including AP use in pregnancy,39,40 during menstrual cycle41,42 and inpatient management 43 will be highlighted in this commemorative venture.
None of the research and progress in the AP arena could have been possible without the unprecedented commitment of the researchers and equally importantly, funding support from various agencies—National Institutes of Health, Juvenile Diabetes Research Foundation, European Commission, Helmsley Charitable Trust and Industry partners—whose combined forces made safe and effective CLC insulin delivery an everyday reality in the United States, Europe, and expanding to other parts of the world.
Animals Replaced by In Silico Trials: The University of Virginia-University of Padova Simulator of the Human Metabolic System in Type 1 Diabetes
Given the large observed inter-individual variability, an average model cannot describe the spectrum of individual responses to diabetes treatment. A cohort of in silico “subjects” that spans the inter-individual variability observed in the general population of people with diabetes would enable a more realistic assessment of the performance of a proposed treatment.
Data from 204 individuals without diabetes 35 who underwent a triple tracer meal protocol provided plasma glucose and insulin concentrations, and model-independent estimates of fundamental fluxes of the glucose system enabled the development of a large-scale glucose-insulin model. The model had 18 differential equations with 42 parameters (33 free and 9 derived from steady-state constraints). Estimates of the model parameters of the 204 subjects enabled the generation of any number of virtual subjects by using the joint multivariate probability distribution of the model parameters.
As the AP development research accelerated with the JDRF initiated Artificial Pancreas Project in 2006 and significant investment by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), the demonstration of the safety and feasibility of AP systems in animals, before any testing in humans slowed the progress. A simulator of type 1 diabetes mellitus (T1DM) having virtual patients with realistic inter-subject variability could enable fast and extensive testing of AP control algorithms and be more representative than small-size animal trials. Researchers at the UVa-Padova collaborated to modify the model by substituting exogenous insulin delivery in place of endogenous insulin secretion subsystem and addressed inter-person variability, yielding a T1DM simulation model with 13 differential equations and 35 parameters (26 free and 9 derived from steady-state constraints) (Figure 2). Model validity was tested using T1DM data sets including adults, adolescents, and children, resulting in the UVA-Padova simulator with 100 adult, 100 adolescent, and 100 children virtual subjects. The simulator included CGM and insulin pump models, enabling testing of any meal and insulin delivery scenario in silico. The simulator was accepted by the FDA as a substitute to animal trials for the preclinical testing of control strategies in AP studies in January 2008.
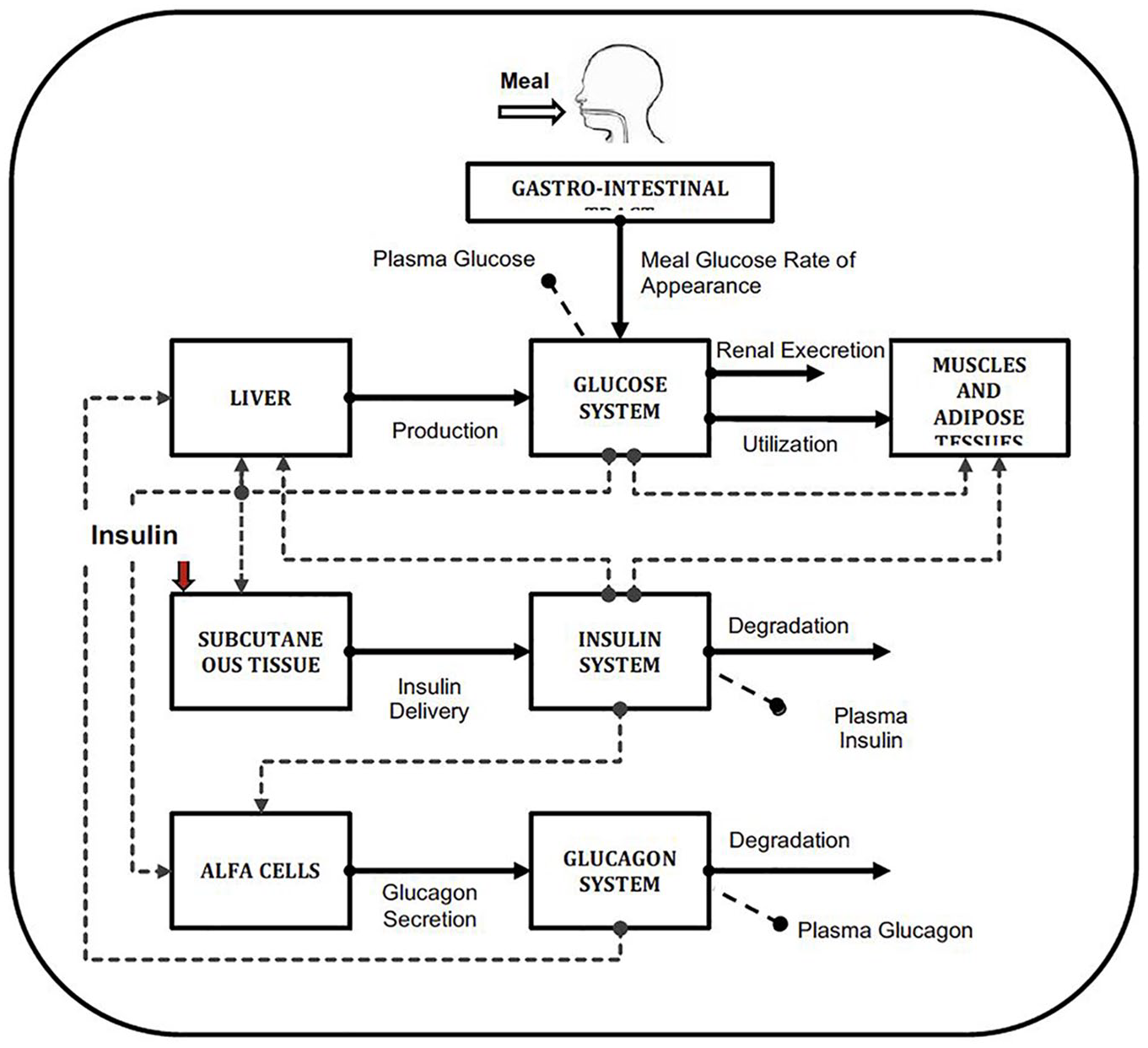
Scheme of the glucose metabolism model in the UVa-Padova T1DM simulator (45).
The UVa-Padova simulator focused on the effects of meals on glucose dynamics. As new clinical data were collected from many clinical studies, its features were enhanced over the years 33 and simulation period increased. 34 Physiological inputs, based on carefully conducted physiology studies in T1D44-47 enabled the incorporation of factors such as circadian changes to insulin sensitivity and dawn phenomenon into the simulator.
Several research-grade simulators have been developed with capabilities to simulate the effects of physical activities and generate additional signals such as energy expenditure from the model as inputs to control algorithms48-50 and dual-hormone AIDs. 51
Designing the Algorithms: Major Problems and Innovative Solutions
The heart of the AP system is the control algorithm that links the input—subcutaneous interstitial glucose measurements—with the output—subcutaneous insulin infusion rates—via the insulin pump. The CLC algorithms need to account for several physiological delays involved in glucose transport from the vascular to the interstitial compartment; insulin absorption and transport from the subcutaneous to the venous compartment; arterial insulin delivery to end organs such as liver, muscle, and fat tissue from the venous compartment; and insulin action on responsive tissues viz, muscle and liver. These algorithms also need to accommodate rapid and dynamic changes to the metabolic state that occur frequently in free-living—meals and physical activity. Early efforts in CLC algorithm design used proportional-integral-derivative (PID) control that relied on the difference between the CGM-reported GC and the target GC. It was supplemented by estimators that used mathematical models describing the dynamics of glucose metabolism.52,53 Over time, powerful GC control algorithms based on MPC (Figure 3) and adaptive control were developed.38,15,54-61 This generation of AID systems was called hybrid CLC because of their dependency on user-provided meal and exercise information.
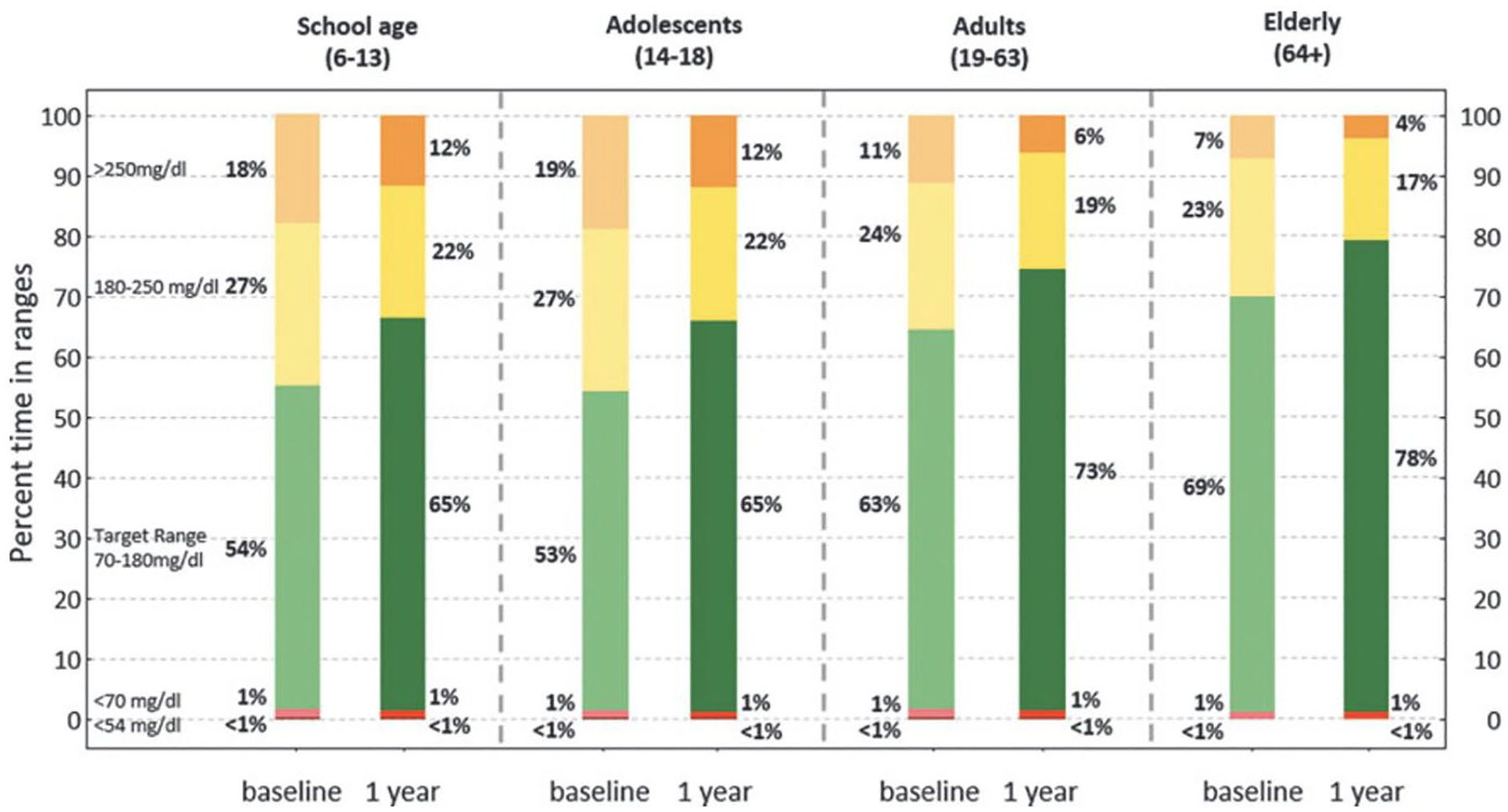
Consensus CGM outcomes for baseline and 12 months Control-IQ use broken down by age groups. 54
Models used in simulators and in CLC algorithms have different tasks and characteristics. Simulator models must have a high level of accuracy. Physiology-based compartment models have been used for this purpose.15,62-65 Their parameters were fitted to data from individual subjects, to create a pool of values for each model parameter. These sets of values enabled the development of statistical distributions for each parameter for use in generating many virtual patients for the simulators. Recently, deep neural networks (DNNs) were also considered to develop models, but they require significantly more data. Physiology-based DNNs can merge both approaches. Models used in AP systems are more compact for computational efficiency. Both physiology-based models and data-driven models are used to build the state-space models that form the core of MPC algorithms.
Two physiology-based models have been used in developing most MPC systems: Hovorka model 15 and UVa-Padova model.63,64,66 In the early stages of MPC algorithm development, fixed-parameter linear state-space models were used. Parameter adaptation was introduced to improve CLC performance. In most algorithms, adaptation was made infrequently, mostly once a day. Data-driven models based on time series model structure provide more compact state-space models and permit higher frequencies of adaptation.67-69 The rationale for frequent adaptation (every 5 minutes when new CGM data are provided) is to adjust the model to rapidly changing metabolic dynamics caused by food consumption or physical activities. To reduce the computational burden and frequent adjustments of insulin infusion rates, zone MPC algorithms have been proposed.70,71
Large disturbances to glucose homeostasis can be caused by food consumption, physical activities, acute psychological stress, irregularities in sleep patterns, and metabolic variations in the female body caused by factors such as pregnancy and menstrual cycle. Recent work focused on the detection of meals and physical activities, the estimation of their characteristics, and forecasting their potential effects on GC in the quest for fully automated AP systems.
A major challenge to the performance of all control algorithms is the delays caused by inferring BGC from subcutaneous CG and the delay of the insulin infused to subcutaneous tissue to affect the glucose metabolism. These delays can be accommodated under manual control by providing insulin boluses before consumption of a meal or adjustment of target glucose values and basal insulin levels before starting an exercise. However, an AP does not know about these future activities. The hybrid CLC system required such manual inputs at the expense of increasing the daily burden on the user. Eliminating the need for manual user inputs to achieve fully automated AP systems continues to be an active research and development area.
Several approaches have been proposed to eliminate manual meal information entry:
(1) Determining meals times and characteristics from historical data to predict the time and dose of insulin boluses.72,73 Large amounts of individual historical data are needed to improve accuracy, and precautions must be added to modify bolusing if the user’s pattern changes on a specific day;
(2) Extending the historical data-based approach to capture the various patterns of behavior of an individual and use these patterns to select the current behavior pattern of this individual. Use this information to provide a sequence of miniboluses as evidence is developing over time on the occurrence of a meal.74,75
(3) Detecting the occurrence of a meal from current CGM data, the estimation of meal characteristics from recent CGM data and suggesting insulin miniboluses based on dynamic estimation of meal characteristics.76,77
(4) Leveraging AI by developing Bayesian networks to estimate current and future meals from historical and current data, and adjusting insulin infusions accordingly. 78
(5) Leveraging accelerometer data from a smartwatch, eating and drinking can be detected based on hand gestures in real-time.79,80
A combination of these techniques can detect meals as soon as they begin, estimate carbohydrate content based on CGM data in real-time, affirm by comparing to individual behavior patterns.
Some approaches for automating the detection of physical activities and adjustment of insulin-dosing/target glucose levels relied on strategies similar to meal detection from historical data.81,82 Others approaches used information such as heart rate or energy expenditure variations collected in real-time.37,71,83-87 Relying exclusively on heart rate signals poses a challenge since psychological stress can also increase heart rate yet will have opposite effects on BGC.88,89 With the recent increase in attention to anxieties and stress, it is foreseeable to expect additional modules in AP algorithms to detect, discriminate, and quantify stress.89,90
The control algorithms have many additional features and constraints for reducing the possibility of aggressive control actions that may lead to hypoglycemia or hyperglycemia, and for minimizing the daily total insulin dose. They are developed as a modular architecture to enable optimization for improved efficiency60,91 and incorporate of algorithms for fault detection,92,93 diagnosis, and mitigation of their effects. Pressure-induced sensor attenuation, 94 imputation of missing sensor data, 95 pump and insulin delivery faults,96,97 and errors in control logic have been addressed.
A recent initiative is to replace the MPC that requires real-time optimization at each sampling time with a neural network trained to mimic the MPC, eliminating the time and resource consumption by the repetitive optimization computations. 98
Dual-hormone AP systems that automate glucagon infusions have been an attractive design that would reduce the potential of hypoglycemia episodes.99-104 The lack of long-term-stable glucagon, user concerns to carry two pumps, having two cannulas for insulin and glucagon, and potential interactions between insulin and glucagon controllers are being addressed by various research teams and companies 105 and clinical trials are being conducted to assess the performance of dual-hormone AP systems.106,107
Digital Twins Technology to Optimize and Personalize Closed-Loop Therapy
A major reason why many people with T1D may fail to achieve clinically recommended time-in-range targets is the lack of personalization of AID control algorithms. Historically, they have been designed to perform well for an entire population of people with T1D, rather than being tailored to individual needs. As a result, an AID control algorithm may be effective for one person but not for others. One way to address personalization is the use of recursively updated models in MPC algorithms.87,108 Digital twins (DTs) offer an alternative by enabling simulations with a digital clone of an individual to determine the appropriate insulin therapy for that individual.
A digital twin (DT) is a mathematical representation of a physical system. They have been utilized in automation, 109 manufacturing, 110 and aerospace applications 111 to optimize processes through simulations, in personalized medicine112,113 and in diabetes and obesity management.114,115 In diabetes technology, a DT refers to a metabolic mathematical model of a single individual that describes how glucose dynamics are impacted by carbohydrates, other nutrients, exercise, insulin, and other hormones such as glucagon and pramlintide. The models can be represented by differential equations15,33,116 or neural networks.114,117
The CGM, insulin, and nutrition data can be used to identify each person’s DT, estimating the parameters of the metabolic model. Figure 4 shows real-world CGM, insulin, carbohydrate, and exercise data collected from the T1D Exercise Initiative 118 that are fit to several DTs. 116 Once the closest DT is identified, it can then be used to compare the person’s current insulin-dosing therapy with new therapies by replaying various alternative therapies based on the person’s individual nutrition, exercise, and insulin-dosing choices. Digital twins have been successfully used to identify optimal changes to insulin-dosing strategies by replaying insulin-dosing settings such carbohydrate ratios,119,120 making suggestions on changes to insulin dosing or carbohydrate intake around exercise to avoid hypoglycemia, 121 or to develop new algorithms for decision support system 122 outcomes based on nutrition.
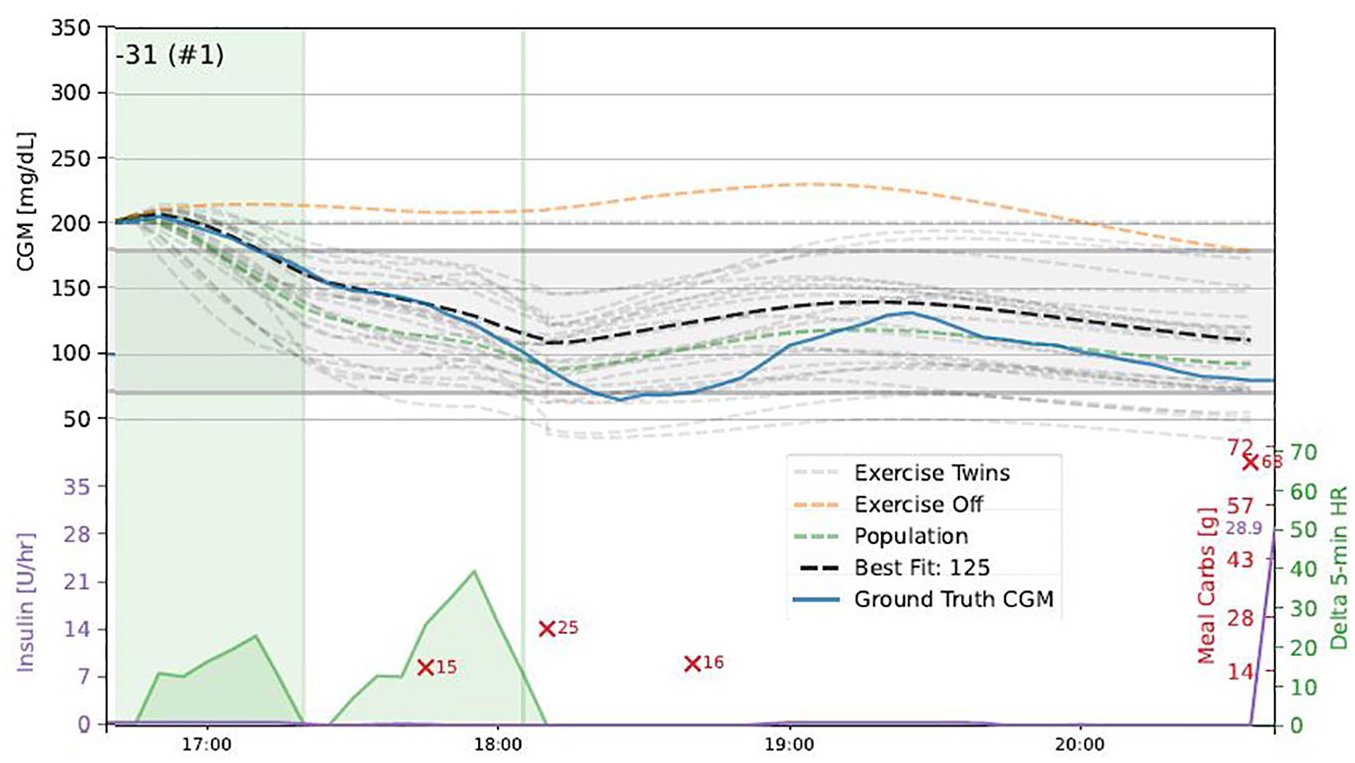
Example of a DT fit during aerobic exercise (shaded green area on the right). Top panel is CGM. Blue trace is real-world data from a T1Dexi participant and hypoglycemia occurred post-exercise. Predictions of DT candidates are light gray–dashed lines. The black-dashed line indicates the best DT most closely matching real-world CGM. The bottom trace shows heart rate changes during and after the exercise event, and carbohydrate intake shown as red X’s with the amount of carbs in grams (124).
Finally, DTs allow patients to experiment with their own data using interactive computer simulations and thereby get insights into what would happen if they change their parameters of their AID system. 123 This latter application was first tested in a pilot clinical study, refined, and deployed in a recently completed larger 6-month randomized clinical study (NCT05610111).
A challenge of the DTs is the necessity to update their parameters as the individuals’ metabolism and lifestyle may change over time. The current DTs are reliable for short periods, but their parameters need to be optimized periodically for longer-duration simulations.
As the DT technology matures, DTs may be used to personalize settings within AIDs to optimize the dosing strategy for each individual. We anticipate that advancements in DT approaches, enhanced by AI and driven by progress in cloud and edge computing, will enable DTs to operate efficiently on devices with limited computational power such as insulin pumps and smartphones.
Extension of Models to Women-Specific Characteristics (Menstrual Cycle, Menopause, Pregnancy)
Women with diabetes (WD) face unique glycemic management challenges across the lifespan. Physiological events such as menstrual cycles, pregnancy, and menopause drastically impact insulin requirements and can exacerbate disease burden, worsen glycemic variability, and increase the risk of complications. Optimizing insulin-dosing algorithms (IDAs) for WD requires addressing sex-specific physiological and lifestyle factors that impact glucose regulation.
The menstrual cycle influences insulin’s glucodynamic action with reduced glucose-lowering action during luteal phase (LP) compared to follicular phase (FP). Data derived from insulin pharmacokinetic and pharmacodynamic studies allowed to quantify the variability in insulin glucodynamic action across menstrual cycle phases in women with T1D. When introduced in a simulator of glucose metabolism in T1D, this variability pattern led to increased time spent >180 and >250 mg/dL during LP vs FP, suggesting that current AID may benefit from informing the IDA with menstrual cycle-related insulin action changes.41,42 Building on this preliminary in silico evidence, there is ongoing work to design and test innovative IDA to optimize insulin treatment across the menstrual cycle in women with T1D.
The WD have an increased risk of pregnancy complications, including pregnancy loss, pre-eclampsia and large-for-gestational-age neonates. Intensification of insulin treatment to achieve glycemic targets is required starting from the pregestational period. There is emerging data regarding the use of machine learning methods for early identification of women with a high risk of developing gestational diabetes. Adverse pregnancy outcomes despite contemporary treatment methods are still inexcusably high for WD. Incremental improvements as small as a 5-unit increase in TIR at 12 weeks reduce the risk of preeclampsia and large for gestational age infants by ~50%. 124 Currently, there is only one AID system that is approved for use in pregnancy, the CamAPS FX (CamDiab Ltd, Cambridge, UK), with a treat-to-target adaptive MPC algorithm. The CamAPS FX use in pregnant women with T1D resulted in a significant increase in time in range (TIR) of 10.5%. 125 The Longitudinal Observation of Insulin Use and Glucose Sensor Metrics in Pregnant Women with Type 1 Diabetes Using Continuous Glucose Monitors and Insulin Pumps (LOIS-P) consortium’s zone-MPC algorithm customized for use in pregnancy, allowing for lower glycemic targets during the day (80-110 mg/dL) and during the night (80-100 mg/dL), yielded favorable glycemic outcomes in a small-scale study. 126 There are evolving research efforts to characterize insulin sensitivity fluctuations across pregnancy trimesters to inform metabolic models and in silico studies for more advanced AID.
Adverse metabolic changes and reduced insulin sensitivity associated with menopause might exacerbate dysglycemia in WD. Data regarding the effect of menopause on insulin action and AID are scarce. Conducting studies to determine the effect of menopause on insulin action would be key to model menopause-associated metabolic changes and customize AID treatment strategies for WD during menopause.
Metabolic models, in silico trials, and advanced IDA have been revolutionizing diabetes care for people with diabetes. These tools must be designed with sex-specific considerations and validated extensively. Most of the current AID systems are often trained on generalized data sets. Expanding data sets to include diverse female populations and tailoring models to help optimize insulin dosing based on sex-specific metabolic characteristics represent a crucial step toward achieving personalized diabetes care.
Conclusions
Many of the advances in technology contributing to the transformation of diabetes care have been supported by the NIDDK over the last 75 years. This chapter illustrated the advances in technology, the development of mathematical models, in silico trial systems, and novel control algorithms. It also highlighted current research activities that will enhance the performance of the AP systems (AID and dual hormone), serve the needs of various groups of people with diabetes that have specific challenges. The trend toward eliminating manual information provided by users, accommodation of the effects of meals, physical activities, stress, variations in sleep, and seasonal changes will improve the TIR of glucose levels and the quality of life of people with diabetes. The NIDDK will continue to play a major role in contributing to and enabling these multifaceted advances.
Footnotes
Abbreviations
ADA, American Diabetes Association; AID, automatic insulin delivery system; AP, artificial pancreas; BCG, blood glucose concentration; CGM, continuous glucose monitoring; CLC, closed-loop control; GC, glucose concentration; FP, follicular phase; FDA, U.S. Food and Drug Administration; HbA1c, glycosylated hemoglobin (%); HCL, hybrid closed-loop system; IDA, insulin-dosing algorithm; LP, luteal phase; MARD, mean absolute relative difference; MPC, model predictive control; PID, proportional-integral-derivative control; T1D, type 1 diabetes; T1DM, type 1 diabetes mellitus; WD, women with diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AB received research funding from NIDDK and Medtronic. MDB reports research grants handled by the University of Virginia from the National Institutes of Health, Novo Nordisk, and Dexcom, and nonfinancial research support from Tandem Diabetes Care, outside of the submitted study. In addition, MDB has patents with royalties licensed to Dexcom, Sanofi, Tandem Diabetes Care, and Novo Nordisk. MDB also reports consulting/speakership activities with Dexcom, Tandem Diabetes Care, Roche, Portal Insulin LLC, BoydSense, and Vertex. BB has received research support from NIDDK, NIH, Breakthrough T1D, Helmsley Charitable Trust, Medtronic Diabetes, Tandem, Insulet, Bigfoot, Dexcom, Abbott, Convatec, and Lilly. Advisory Boards for Medtronic, Tandem, Animas, Sanofi, Roche, Novo Nordisk, Lilly, Convatec, BD, Profusa, and Glysense. AC reports research grants handled by Illinois Institute of Technology from the National Institutes of Health (NIDDK), National Science Foundation, Walder Foundation and Breakthrough T1D (JDRF); research device support from Dexcom; and honoraria from Eli Lilly, Medtronic and American Diabetes Association. EC is scientific advisory board member of Novo Nordisk, Eli Lilly, MannKind, Arecor, Portal Insulin, Tandem, and Ypsomed. FJD reports funding over the time course described in this paper from the Showalter Foundation, Roche Diagnostics Corporation, the Juvenile Diabetes Research Foundation, the Helmsley Charitable Trust, and the National Institutes of Health (NIDDK). FJD also reports royalty payments from patents in a portfolio that have been licensed to Insulet, Roche, and Dexcom. PGJ receives research support from Dexcom and Eli Lilly and is a co-founder and shareholder in Pacific Diabetes Technologies. ED has received personal fees from Roche and Eli Lilly and Company; holds patents on artificial pancreas technology; and has received product support from Insulet Corporation, Tandem Diabetes Care, Roche, and Dexcom, Inc. The work presented in this article was performed as part of his academic appointment and is independent of his employment with Eli Lilly and Company. CF reports receiving research support from Novo Nordisk, Dexcom, and Tandem Diabetes Care handled by the University of Virginia, and patent royalties from Novo Nordisk and Dexcom handled by the University of Virginia’s Licensing and Ventures Group. IBH reports Research funding: NIDDK, Tandem, Dexcom, and MannKind and consulting with Abbott, Roche, and Hagar. PGJ receives research support from Dexcom and Eli Lilly and is a co-founder and shareholder in Pacific Diabetes Technologies. BPK reports research grants handled by the University of Virginia from the National Institutes of Health, Novo Nordisk, Dexcom, and Tandem Diabetes Care. In addition, BPK has patents with royalties licensed to Dexcom and Novo Nordisk. CC, CDM, AF, and LQ have no disclosures relevant to this work. JSS has been an advisor to 4Immune, AbbVie, Abvance, ActoBiotics, Adocia, Aerami/Dance Biopharma, AiTA, Applied Therapuetics, Arecor, AstraZeneca, Avotres, Bayer, Biomea Fusion, COUR Pharmaceuticals, Dexcom, Eli Lilly, Kriya Therapeutics, Levicure, Novo Nordisk, Oramed, Provention Bio, PolTREG, Quell Therapeutics, RegCell, Remedy Plan Inc., Respond Health, Sanofi, Shoreline Biosciences, Signos, Vertex, vTv Therapeutics, and WiNK. He is a member of the Board of Directors of Applied Therapeutics and of SAB Biotherapeutics. He is Chair of the Strategic Advisory Board of the EU EDENT1FI consortium. JSS has equity in: 4Immune, Abvance, AiTA, Applied Therapeutics, Avotres, Dexcom, Immunomolecular Therapeutics, Oramed, SAB Biotherapeutics, Signos, vTv Therapeutics, and WiNK.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
