Abstract

A continuous and standardized postmarket assessment of blood glucose monitoring systems (BGMS) is essential from both a clinical and a legal perspective. As a European Union’s In Vitro Diagnostics Regulation mandate, it ensures proper performance and patient safety. Consequently, dissemination and availability of studies on a device’s performance (ie, precision and accuracy) have become critical factors guiding evidence-based procurement choices for distributors and users. Comprehensible, meaningful and robust performance indices are thus becoming increasingly important. A comparative assessment of 3 BGMS (Contour Care, Ascensia, GL22 and GL60, both Beurer) was conducted in accordance with the protocols and requirements outlined in DIN EN ISO 15197:2015 1 and a new assessment metric is proposed.
Commercially available BGMS were provided by their respective manufacturers/distributors. Glucose measurements of native and manipulated samples (glycolysis at 37°C or supplementation of a 40% glucose solution) from fingertip capillary whole blood were taken using the BGM in conjunction with 1 of 2 reference methods using whole blood or plasma (Table 1). Whole blood measurements were converted to plasma equivalents using a hematocrit-based conversion method, where necessary. Accuracy and precision of the BGMS and reference assays were validated using manufacturer-issued control solutions and certified standards. For each system, 100 subjects with diabetes, type II, were included, amounting to a total of 1800 assessable measurements based on the inclusion and exclusion criteria established by the manufacturers and Supplemental Appendix A of DIN EN ISO 15197:2015.
Results of System Accuracy Evaluation.
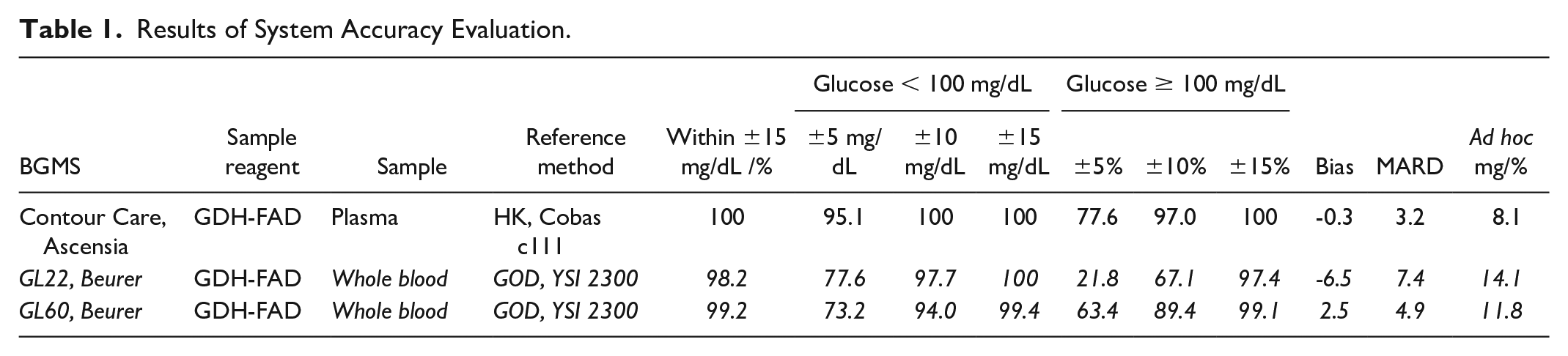
All systems fulfilled the acceptance criteria. All measurements of each system lie in zones A and B of the Parkes error grid. 2 Across systems, between 98.2% (GL22) and 100% (Contour Care) of measurements showed deviations of less than ±15 mg/dL and ±15% for glucose concentrations <100 mg/dL and ≥100 mg/dL, respectively (Table 1). Bias (systematic deviation) and MARD (mean absolute relative difference) vary considerably but inconclusively between the systems (see Table 1).
An ad hoc analysis to determine the narrowest error margin in which at least 95% of meter inaccuracies fall was calculated at 8.1 mg/dL/% (Contour Care), 11.8 mg/dL/% (GL60), and 14.1 mg/dL/% (GL22), highlighting the variations not reflected in the standard assessment. A 2-factor analysis of variance revealed significant effects of both reference method and sample method (P < .05) on the deviation of BGM to the reference as a performance indicator.
Accuracy and precision of BGMS are 2 of the key elements for achieving optimal glycemic control in a self-management regimen. Only the precise detection of dysglycemic episodes can ultimately reduce the risk of diabetes-associated complications. Variations in performance can be introduced by several factors including sample type, reference method, technical aspects of BGMS, and each system’s susceptibility to interferents.1,3 Although considerable deviations between established reference systems have been demonstrated,4,5 which might obscure comparability of results and complicate data interpretation, a Tukey post hoc test showed YSI calibrated systems and whole blood sampling resulted in significant (P < .05), yet still clinically acceptable deviations from the reference method, maintaining regulatory compliance. Based on the criteria outlined in DIN EN ISO 15197:2015, our evaluation revealed that all systems tested met and surpassed the minimum system accuracy requirements.
However, differences in key performance metrics usually considered (ie, BIAS, MARD) suggest that adherence to standards is better understood as a spectrum along which quality can be defined further, especially considering the limitations and shortcomings of these parameters. In a clinical context, therapeutic decisions should always be founded on measurements that are truly reflective of the actual glycemic status. The ad hoc confidence metric presents a promising approach for a robust and comprehensive performance indicator.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968241289958 – Supplemental material for Comparative System Accuracy of Blood Glucose Monitoring Systems—Advocacy for a New Accuracy Assessment Metric
Supplemental material, sj-pdf-1-dst-10.1177_19322968241289958 for Comparative System Accuracy of Blood Glucose Monitoring Systems—Advocacy for a New Accuracy Assessment Metric by Matthes Kenning, Anselm Puchert and Eckhard Salzsieder in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors wish to express their sincere appreciation to the personnel of the Institute for Diabetes Karlsburg GmbH for providing technical help, intellectual input, and feedback in conducting the studies. The sponsors were permitted to review and comment on the manuscript, final decision on content was retained by the authors.
Abbreviations
BGM, blood glucose monitor, BGMS, blood glucose monitoring systems, MARD, mean absolute relative difference
Author Contributions
All authors contributed equally to the manuscript in terms of research, writing, and editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institute for Diabetes Karlsburg GmbH, Germany, which carries out studies evaluating blood glucose meter systems on behalf of various clients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
