Abstract
Background:
Glucagon-like peptide 1 (GLP-1) is a hormone that promotes insulin secretion, delays gastric emptying, and inhibits glucagon secretion. The GLP-1 receptor agonists have been developed as adjunctive therapies for type 2 diabetes to improve glucose control. Recently, there has been an interest in introducing GLP-1 receptor agonists as adjunctive therapies in type 1 diabetes alongside automatic insulin delivery systems. The preclinical validation of these systems often relies on mathematical simulators that replicate the glucose dynamics of a person with diabetes. This review aims to explore mathematical models available in the literature to describe GLP-1 effects to be used in a type 1 diabetes simulator.
Methods:
Three databases were examined in the search for GLP-1 mathematical models. More than 1500 works were found after searching for specific keywords that were narrowed down to 39 works for full-text assessment.
Results:
A total of 23 works were selected describing GLP-1 pharmacokinetics and pharmacodynamics. However, none of the found models was designed for type 1 diabetes. An analysis is included of the available models’ features that could be translated into a GLP-1 receptor agonist model for type 1 diabetes.
Conclusion:
There is a gap in research in GLP-1 receptor agonists mathematical models for type 1 diabetes, which could be incorporated into type 1 diabetes simulators, providing a safe and inexpensive tool to carry out preclinical validations using these therapies.
Introduction
Glucagon-like peptide 1 (GLP-1) is a hormone produced in the L-cells in the intestine. It belongs to the incretin hormones group alongside glucose-dependent insulinotropic peptide (GIP), being generated as a response to a meal. 1 The GLP-1 has diverse effects that include promoting insulin secretion, inhibiting glucagon secretion, slowing down gastric emptying, and producing a satiety effect through mediation of the vagus nerve. 2 Endogenous GLP-1 has a short life and is rapidly degraded due to the action of dipeptidyl peptidase-4 (DPP-4). 3 The GLP-1 receptor agonists (GLP-1 RAs) were developed to emulate GLP-1 actions while being protected from degradation by DPP-4, achieving longer half-lives than endogenous GLP-1. 4
The GLP-1 RAs have been designated as adjunctive therapies in the treatment of type 2 diabetes (T2D). Their action has proved effective in reducing HbA1c levels, weight, and insulin requirements. 5 Exenatide, the first GLP-1 RA, was approved by the Food and Drug Administration (FDA) in 2005. Currently, around ten GLP-1 RAs are available in the market, the main difference between the compounds being their action time (ie, requiring weekly or daily administration). However, GLP-1 RAs have shown some waning effect (tachyphylaxis) 2 with degradation of their effect after sustained administration. Also, adverse gastrointestinal (GI) effects have been reported, even leading to gastroparesis. 6 Hence, GLP-1 administration is not trivial.
There has been an interest in the potential of this kind of drug for type 1 diabetes (T1D), where the delay of gastric emptying could potentially ease the management of postprandial hyperglycemia, hence facilitating glucose control and improving weight and HbA1c. 7 Since the development of GLP-1 RAs, the possibility of using them for T1D has been explored.8-10 However, its higher risk of hypoglycemia and ketosis 11 has resulted in a lack of approval for T1D by the FDA. 12 Nevertheless, there is a current interest in these drugs, even if their use is off-label, for patients with T1D. 13 Current T1D treatment often relies on automatic insulin delivery (AID) systems, with a variety of options available in the market. 14
The AID systems administer insulin automatically from a continuous infusion insulin pump based on the readings from a continuous glucose monitor (CGM). One of the main challenges remaining for these systems is the removal of patients’ intervention (ie, becoming completely automatic), eliminating the need for patients to count carbs or announce their meals to the system. Adjunctive therapies such as GLP-1 RAs could facilitate postprandial control, removing meal announcements. Clinical trials have been performed with this aim using pramlintide, an amylin analog, which has similar effects to GLP-1.15,16 Nevertheless, the same way patients need to be aware of the changes in their glucose that these medications are going to introduce, 13 the automatic systems need to be informed of the changes in glucose behavior derived from the administration of GLP-1 RAs to avoid insulin overdosing that could lead to severe hypoglycemic episodes.
The preclinical validation of AID can be accelerated by simulations, a fast and inexpensive way of assessing the controller’s performance. 17 These validations involve the use of T1D simulators that reproduce the pharmacokinetics (PK) of drugs and their pharmacodynamics (PD) on glucose. However, the most widely used simulators for T1D18-20 do not incorporate the effects of adjunctive therapies such as GLP-1 RA. Some clinical trials have made small studies using GLP-1 alongside AID systems (a review can be found in Furió-Novejarque et al 21 ). Although they improved time in range compared with the insulin-only control arm (by 14% in Renukuntla et al 22 and by 2.8% in Sherr et al 23 ), a proper understanding of these drugs’ effects while designing control algorithms is needed. For this purpose, a simulator that incorporates the effect of these drugs would be required in order to observe the necessary differences in the control in the presence or absence of these effects.
This article aims to conduct a literature search to analyze the available mathematical PK/PD models of GLP-1 RA and to evaluate their viability for use in simulation for T1D. The rest of this article is organized as follows: section “Methods” presents the literature search methods, section “Results” describes the results, section “Discussion” presents a discussion based on the gathered information, and section “Conclusion” lays out the conclusions.
Methods
The purpose of this search was to find GLP-1 PK/PD models that could be incorporated into existing diabetes simulators. Typical models for this purpose consist of compartmental models. The models should include GLP-1 PK, describing the appearance of GLP-1 in the blood after administration, and PD, describing GLP-1 effects. The GLP-1 actions include promoting insulin secretion, inhibiting glucagon secretion, and slowing down gastric emptying. Its satiety effect also helps lower weight over time, which, overall, helps improve glucose control and lower HbA1c.
Three databases were examined in the search for GLP-1 modeling works: Web of Science, PubMed, and Scopus. The search included records published until April 2024. Three different sets of keywords were used to narrow down the search:
“GLP-1 pharmacokinetics pharmacodynamics model.”
“GLP-1 mathematical model.”
“GLP-1 simulation model.”
The search of these keywords resulted in 1530 documents (804 in Web of Science, 503 in PubMed, and 223 in Scopus). The search flow diagram is summarized in Figure 1. An initial selection was performed based on a title and abstract assessment, leaving 80 documents to revise, which became 39 after removing duplicates across databases. The full text of these 39 works was further analyzed, with the addition of three works that were found in the analyzed papers’ references. Papers that focused on other hormones (such as DPP-4) only provided a model for GLP-1 secretion or did not include any GLP-1 effect were discarded. Another exclusion criterion was the lack of use of data for model validation (development should be based on clinical data to support the model behavior). Works considering the secretion of endogenous GLP-1 were included in the selection if a description of its effects were available. For instance, Landersdorfer et al 24 use data from a trial with vildagliptin (a DPP-4 inhibitor), considering the endogenous production of GLP-1, but propose a model on the influence of GLP-1 on insulin secretion. Similarly, Wu and An 3 use linagliptin (another DPP-4 inhibitor) but include descriptions of the GLP-1 effect on insulin secretion and glucagon inhibition. The papers should include the set of equations describing the model dynamics or at least a description of the model in more straightforward proposals.
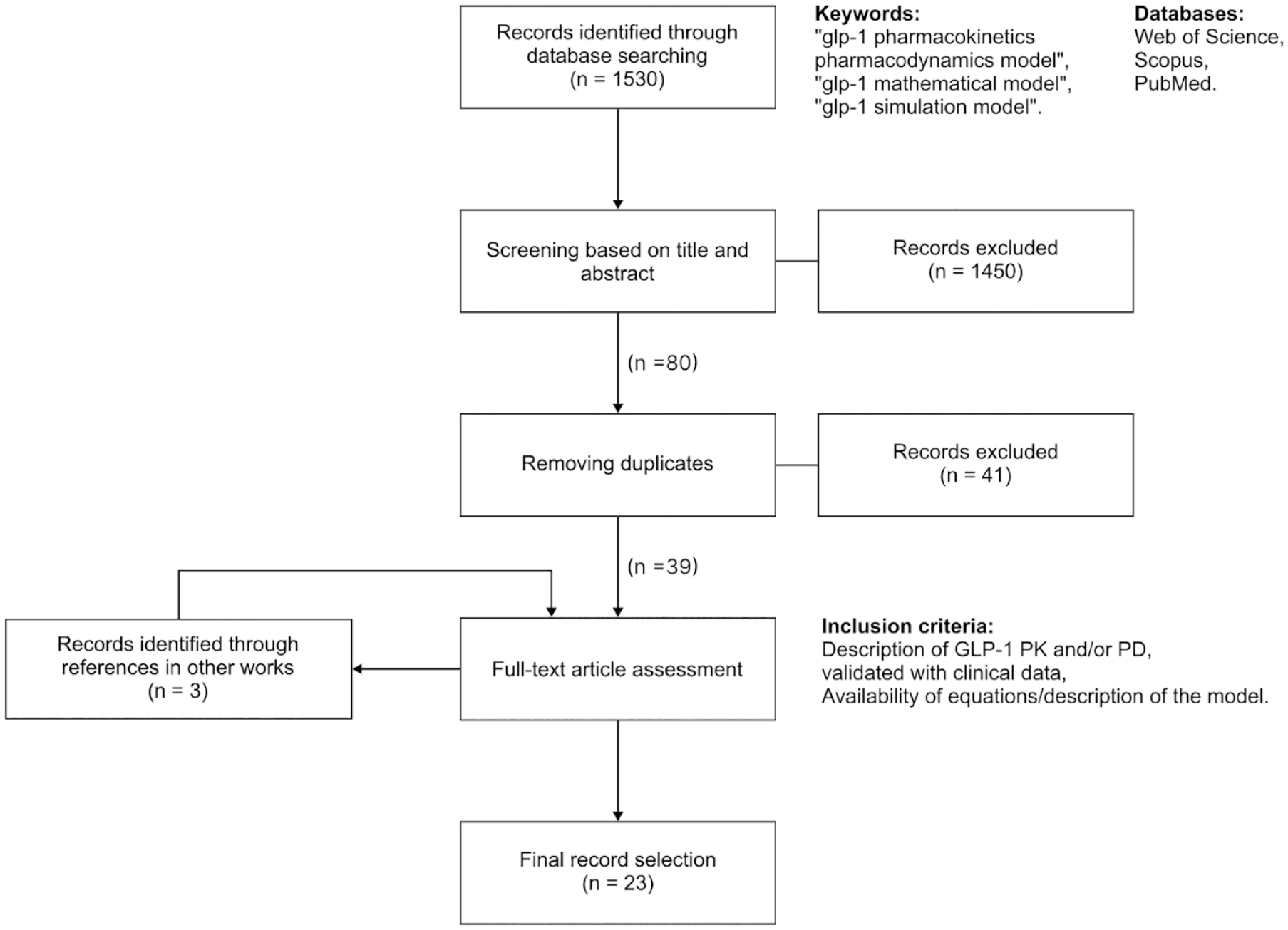
Flow diagram of the literature search.
A total of 23 works were finally selected. They were later classified according to the information they provided, as described in the next section. The information to be collected about each work included what kind of compound was used (ie, the type of GLP-1 RA), what type of data was used for validation of the model (eg, animal models, people with or without diabetes), and what kind of effects are described (increase in insulin secretion, glucagon inhibition, gastric emptying delay, weight loss, or HbA1c reduction).
Results
Table 1 presents a summary of the key information collected in the selected works. For each of the papers, the table indicates the specific GLP-1 analog used (also indicates whether it was endogenous GLP-1 instead), the subjects the validation data were obtained from, and the possible descriptions of the model, meaning whether PK and PD descriptions are present, with a distinction on the type of effects described, namely: promotion of insulin secretion, delay of gastric emptying, or description of long-term effects, such as variations in weight, HbA1c, or fasting plasma glucose.
Summary of the GLP-1 PK/PD models and their characteristics.
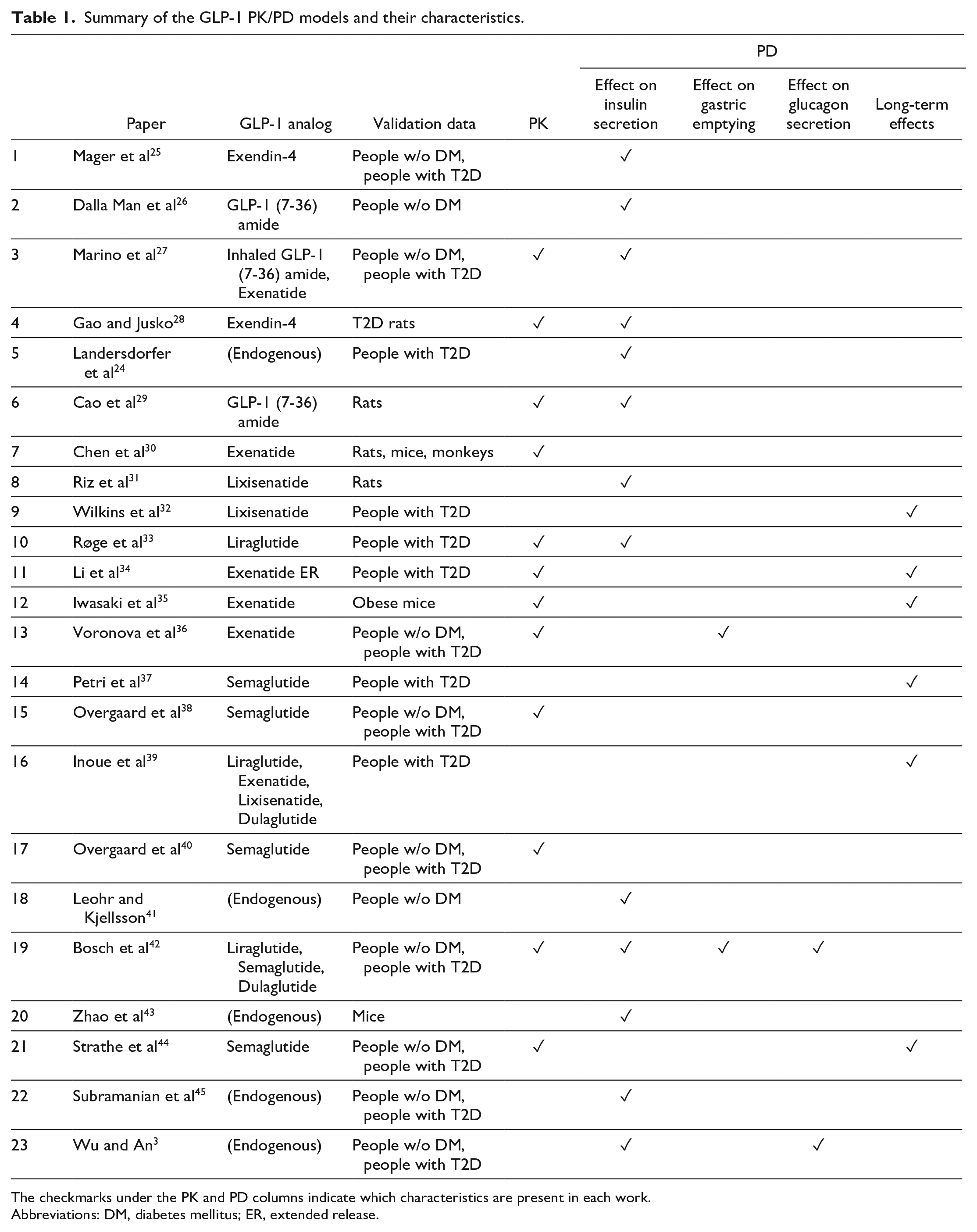
The checkmarks under the PK and PD columns indicate which characteristics are present in each work.
Abbreviations: DM, diabetes mellitus; ER, extended release.
Of the 23 selected records, three described GLP-1 PK only,30,38,40 11 of them described only GLP-1 PD effects (they may consider secretion of endogenous GLP-1), and nine of them described both PK and PD. The effect of GLP-1 on insulin secretion is the most present in the selected works (13 of the 20 works that describe some GLP-1 PD). Two of them describe the influence of GLP-1 on gastric emptying,36,42 the inhibition of glucagon secretion is described in two,3,42 and long-term effects are included in six. Most of the developed models (17 works) are based on human data (usually a combination of people with and without diabetes), whereas the rest use mostly rats or mice data.
Five works in the list assume an endogenous secretion of GLP-1 and the remaining make use of GLP-1 RAs. Regarding the type of GLP-1 RA, exenatide is present in five works; lixisenatide appears in three works, semaglutide in five, liraglutide in three, and dulaglutide in two. The main difference among these compounds is that exenatide, lixisenatide, and liraglutide require a daily injection, whereas semaglutide and dulaglutide use weekly administrations. Exendin-4 is a natural analog of GLP-1, 46 and GLP-1 (7-36) amide is equivalent to the endogenous peptide. 47
Of note, the work by Overgaard et al 40 is based on the model presented in Overgaard et al, 38 where the authors propose some modifications to the original model and perform an additional validation with a different data set. Similarly, the work of Bosch et al 48 carries out a new validation of the model proposed in Bosch et al, 42 but no modifications to the model are proposed. Overgaard et al 49 perform a validation of the models proposed by Petri et al 37 using semaglutide, exenatide ER, and dulaglutide data.
Discussion
The literature review carried out in this work provides an overview of the available GLP-1 models in the literature, with a general description of their features. However, our interest lies in bringing these simulation models to T1D, and none of the models found have been validated with data from people with T1D. Moreover, most of the models focus on GLP-1’s influence on insulin secretion, which makes sense when used in T2D, but it would only be useful for T1D at the onset of the disease.
One feature that could be translated to T1D simulators almost directly is the description of GLP-1 RAs PK. In the works included in this review, most of the PK models share a common structure as most of them consist of one compartment, or two compartments with first-order absorption and first-order elimination rates, regardless of the compound used. The only exception is the model by Li et al, 34 where they model exenatide ER and their proposal uses a total of seven compartments for the PK subsystem. Nevertheless, it could be inferred that a readjustment of transfer rates would allow them to be adapted to any GLP-1 RA compound.
The effects of interest for T1D would be the delay in gastric emptying and the inhibition of glucagon production. Glucagon response after meals is one of the reasons for hyperglycemia in T1D after meals. 50 However, the only models to include inhibition of glucagon secretion by GLP-1 in this selection are the proposals of Bosch et al 42 and Wu and An. 3 In the Bosch model, the glucagon compartment includes a modulation term on glucagon concentration that depends on the concentration of GLP-1:
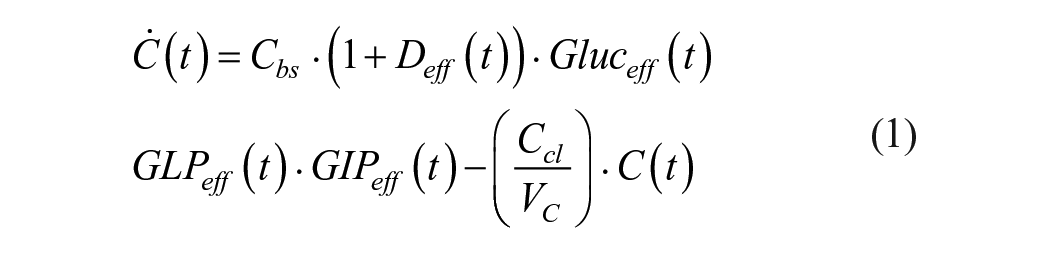
The notation used in Equation (1) has been adapted from the original expression in Bosch et al
42
[Equation (17) in that paper] for simplicity. In the above expression,
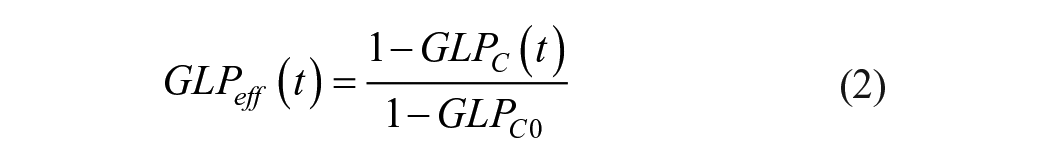
This expression is based on Equation (21) in the work by Bosch et al.
42
A different model by Wu and An 3 focuses on the PK/PD of sitagliptin, a DPP-4 inhibitor, and they include a model for endogenous GLP-1 secretion and its effect on glucagon inhibition. The glucagon concentration is defined as:
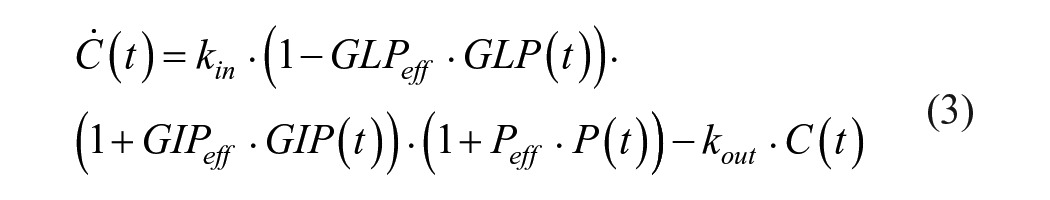
Similar to Equation (1), the notation has been simplified compared with the equation in the original work [Equation (10) in Wu and An
3
]. The parameters
Another effect of interest for T1D simulators would be the descriptions of gastric emptying. However, GLP-1 RAs’ influence on gastric emptying is a matter of discussion. The work by Røge et al 33 did not model the effect of liraglutide on gastric emptying because such an effect was not observed in their data. The data were obtained from the trials carried out by Flint et al 51 and Hermansen et al. 52 Both trials administered three liraglutide doses (0.6, 1.2, and 1.8 mg) for one week each and measured gastric emptying at the clinic using the paracetamol test. The conclusions regarding liraglutide’s influence on gastric emptying were unexpected as the 0.6 and 1.8 mg doses were ineffective in the first trial. Likewise, no difference in gastric emptying was observed in the second trial between the 1.8 mg liraglutide dose and placebo. In Hermansen et al, 52 gastric emptying was assessed with both the paracetamol test and the 13C-octanoate breath test. Nevertheless, the authors mention in the discussion that the high-fat content of the meal used in the trial could have masked the effect of the GLP-1 RA as fatty meals present a slower emptying from the stomach. In addition, the paracetamol test has been questioned as an effective way to measure gastric emptying as it reflects the apparition of liquids from the stomach in plasma, but it is not useful to assess solid gastric emptying. 53 Scintigraphy, the “gold standard” technique to measure gastric emptying, should be used instead, although this technique is burdensome and costly. 12 The work of Marino et al 27 also measured gastric emptying after the administration of inhaled GLP-1, measuring the absorption of 13C-octanoate from the meal. However, there was no apparent influence of inhaled GLP-1 on gastric emptying. Consequently, it was not described in their model either.
The models proposed by Voronova et al 36 and Bosch et al 42 provide descriptions of GLP-1 RAs on gastric emptying. The first was based on various data sets that measured gastric emptying using the paracetamol test, whereas the second based its results on glucose data.
In the model by Voronova et al,
36
the GLP-1 RA affects the gastric emptying rate for liquid food via a Michaelis-Menten function in the compartment describing the exit of liquid food (
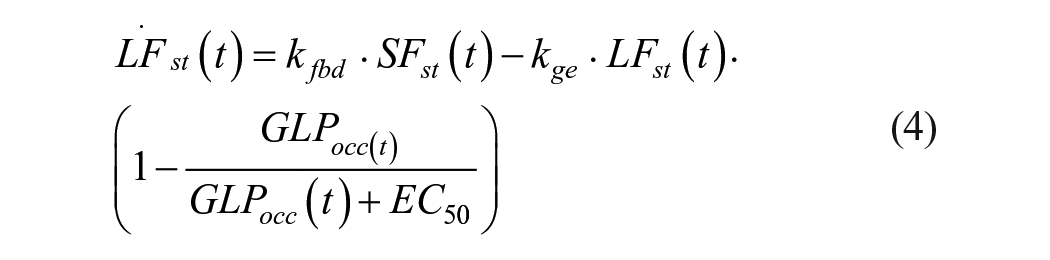
where
The model by Bosch et al
42
also describes the effect of GLP-1 on the gastric emptying rate as a modulation of their nominal absorption rate constant
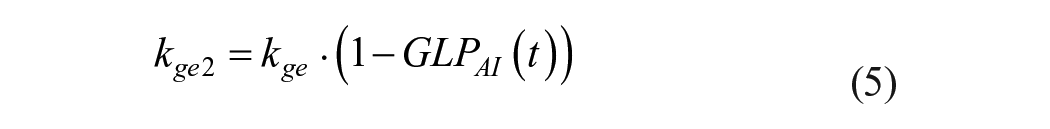
The term
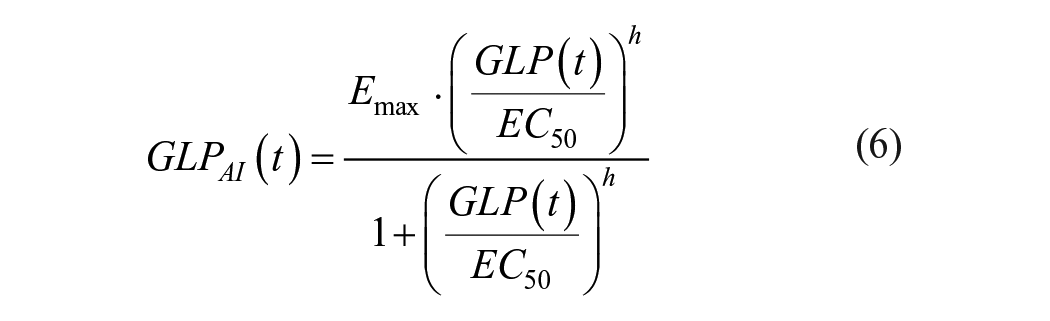
Equations (5) and (6) correspond to Equations (1) and (2) in Bosch et al 42 (some variable names from the original expressions have been modified for brevity).
Among the models in this selection, the two examples that include a modification of the gastric emptying rate use a similar approach, modulating said rate based on a Michaelis-Menten or Hill equation that depends on GLP-1 concentration (or active GLP-1). The main difference lies in the maximum allowed inhibition since the first model
36
allows for complete gastric emptying inhibition (
Model structures inspired by these examples could be incorporated into T1D simulators, identifying its parameters with data from people with T1D to be adapted to the simulator.
Other effects to be included in T1D simulators would be the influence of GLP-1 on fasting plasma glucose values, which would affect HbA1c measurements. Estimations of long-term weight variations are not usually included in this type of simulator, but it would be an informative outcome for both health care professionals and algorithm developers, who would be the primary users of the simulators.
The use of these drugs is also associated with several GI side effects. 54 A simulation tool aware of these consequences could help adjust the dosing of these treatments when patients are first introduced.
Incorporating GLP-1 models into T1D simulators will allow the development of closed-loop algorithms that include GLP-1 as a control action or that are aware of the potential changes in glucose dynamics introduced by these treatments. The AID systems should be able to deal with both functioning modes without requiring major interventions from the patients (eg, just activating or deactivating a configuration option). For instance, a patient could start a GLP-1 RA treatment and discontinue it temporarily due to an adverse reaction. The closed-loop algorithm should be able to function and regulate glucose in both situations.
Conclusion
This work has presented a review of available GLP-1 models in the literature, bringing to light that there are no available models that have been validated with data from people with T1D. Given the recently regained interest in the use of these therapies alongside AID systems, simulators including GLP-1 RAs mathematical models would provide a safe and inexpensive tool to carry out preclinical validations of closed-loop algorithms taking into account the effects of adjunctive therapies. This article also has provided an overview of the potential elements of the existing models that could be used to develop PK/PD models for T1D. Nevertheless, experimental data on the use of these drugs on people with T1D will be needed to validate future model proposals.
Footnotes
Acknowledgements
None.
Abbreviations
GLP-1, glucagon-like peptide 1; GIP, glucose-dependent insulinotropic peptide; DPP-4, dipeptidyl peptidase-4; GLP-1 RA, GLP-1 receptor analog; T1D, type 1 diabetes; FDA, Food and Drug Administration; AID, automatic insulin delivery; CGM, continuous glucose monitor; PK, pharmacokinetics; PD, pharmacodynamics
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by PID2019-107722RB-C21 funded by MCIN/AEI/10.13039/501100011033 and grant CIPROM/2021/012 funded by Conselleria de Innovacion, Universidades, Ciencia y Sociedad Digital from Generalitat Valenciana.
