Abstract
Background:
The purpose of this study was to assess adults’ perspectives after using the insulin-only Bionic Pancreas (BP) during a 13-week pivotal trial. Automated insulin delivery (AID) systems show promise in improving glycemic outcomes and reducing disease burden for those with type 1 diabetes mellitus (T1D). Understanding the lived experience of those using the BP can help to inform education and uptake of AID devices.
Methods:
Adults ages 19 to 75 (n = 40) participated in age-specific focus groups (19-25, 26-40, 41-64, and 65+) exploring their experiences, thoughts, and feelings about using the BP. Three authors analyzed the focus group data using a hybrid thematic approach.
Results:
Qualitative analysis of focus groups revealed 14 sub-themes falling into four major themes (diabetes burden, managing glucose levels, daily routine, and user experience). Although participants’ overall experience was positive, some reported struggles related to managing out-of-range glucose levels and challenges with the system responding to unique meal schedules and exercise regimens.
Conclusion:
This study captures patient perspectives regarding their experiences with a new AID system. Patient voice can inform device development and educational approaches for people with T1D. Identifying which patients may benefit the most from wearing this system may facilitate patient/clinician discussions regarding insulin delivery systems that best meet their individualized needs and expectations that may support device uptake and continued use.
Introduction
Advances in diabetes technology include the development of automated insulin delivery (AID) systems. These systems positively impact glycemic outcomes for individuals with type 1 diabetes (T1D), including reduced A1c,1-4 increased time in range,2,3,5,6 and decreased hypoglycemia.3,7
Examining the psychosocial impact of AID systems is crucial to understanding their real-life impact. Despite a robust literature on the metabolic advantages of AID, less research has evaluated the psychosocial outcomes and human factors associated with their use. Current findings suggest that adults generally have a positive impression of AID systems,3,8,9 reporting improvements in treatment satisfaction,3,7,10,11 sleep quality,1,4,5,11 fear of hypoglycemia,4,5,10 diabetes distress,5,6,10 burden, 7 and worry. 8 Of these studies, most use standard questionnaires,6,7,9-11 qualitative interviews or focus groups,4,8 or included no measure of psychosocial health.3,5,7 Although studies show consistent AID benefits, findings related to perceived burdens are less robust. 12
Although these studies offer initial insight into the psychosocial impact of AID systems, additional qualitative research is needed to capture the user’s lived experience.1,7,12-15 It is critical that research captures the complexity of the user’s perceived benefits and burdens1,7,14 for ongoing system development, 16 creating patient education materials, 17 and managing expectations. Assessing the user experience is critical, as the success of AID is predicated upon whether the user finds more benefits than burdens and is, therefore, willing to initiate and continue device use over time.
Most studies have assessed AID over a short period of time (eg, days or weeks), not accounting for the potential long-term benefits or burdens related to device use. Of published studies lasting 12 weeks or longer,4,5,7,8,10,18 evaluation of the psychosocial aspects of AID systems has been limited to and relied on questionnaires to capture psychosocial outcomes.5-7,9-11,19 Studies that included qualitative assessment have begun to provide valuable insights into understanding the user experience.4,18,20 Building on this research, with larger sample sizes, offers a unique opportunity to contextualize psychosocial outcomes by capturing lived experiences.21,22
This study evaluated the lived experience of adults using the insulin-only Bionic Pancreas (BP) System as part of a larger multicenter randomized trial designed to assess the efficacy and safety of the BP. This pivotal trial compared the BP (which is initialized without a warm-up period, using only an individual’s body weight) with standard care over 13 weeks. The BP autonomously determines all insulin doses, and meals are announced without carbohydrate counting. The study design, methods, and results are described elsewhere. 23 The psychosocial outcomes based on validated questionnaires have also been reported. 24 This study aimed to evaluate adult perceptions of the BP using qualitative focus groups.
Research Design and Methods
Participants
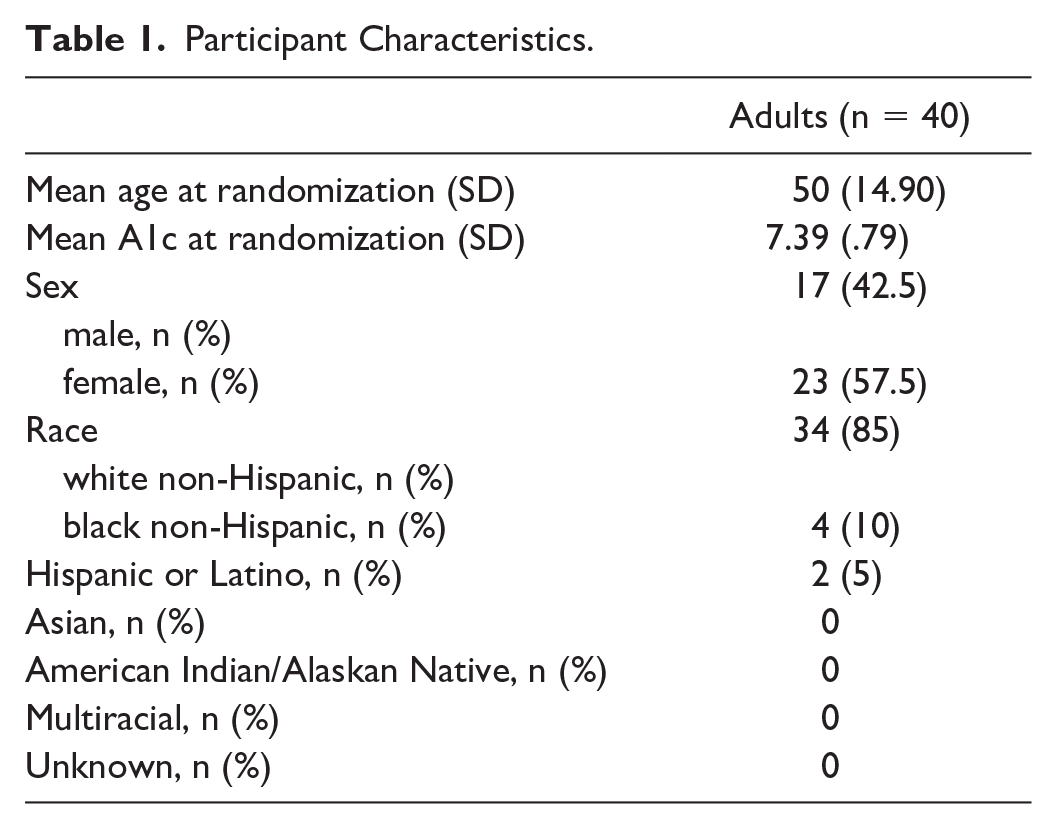
Adults participating in this pivotal trial were invited via email at the end of the 13-week trial to participate in focus groups to share their experiences with the BP. A total of 217 adults randomized to the BP completed the pivotal trial and were invited to participate. Of these, 40 (18%) participated in a focus group or interview. Eight focus groups and one semi-structured interview were held with adults ages 25 to 75 (n = 40). Participants were grouped by age: 26 to 40 years old (two groups; n = 12), 41 to 64 years old (four groups; n = 19), 65 to 75 years old (two groups, n = 8), and one interview (age range = 19-25). Participant characteristics can be found in Table 1. Although all participants in the 19 to 25 age category were invited to participate in focus groups, only one individual signed up, so an individual interview was completed. Because the same questions were asked during this individual interview as were asked during the focus groups, we chose to include this individual’s responses in the data presented. Focus group participants were similar to participants in the overall trial in terms of race and ethnicity. Focus group participants were older than the average pivotal trial participants, and the average baseline A1c was 0.41% lower.
Participant Characteristics.
Procedures
Focus groups were held virtually using Health Insurance Portability and Accountability Act (HIPPA)-compliant teleconferencing software and were conducted by one licensed psychologist and one psychology fellow who followed a focus group guide to maintain fidelity. Focus group questions were developed by the study team’s psychologist, endocrinologists, and nurse educators. Questions were grounded by the Unified Theory of Acceptance and Use of Technology (UTAU), 25 which focuses on four areas of technology acceptance: performance expectancy, effort expectancy, social influence, and facilitating conditions.25,26 The focus group guide assessed expectations of the BP, experiences in different settings (eg, home, work, and sports), trusting the BP, benefits and challenges of its use, and experience with BP-specific features. The 90- to 120-minute discussions were recorded and transcribed by Medikin, a medical transcription company. Study protocols were approved by the Jaeb Center for Health Research Institutional Review Board. Transcriptions of the discussions and coding and analysis took place after completing all focus groups.
The coding team first read all the transcripts to familiarize themselves with the data. The coding team (K.R.H., J.W.-B., and K.P.G.) consisted of a psychologist and a psychology post-doctoral fellow who conducted the focus groups and a cultural anthropologist who did not lead any focus groups. This team’s make-up helped ensure coding was deeply rooted in the data and not biased by outside experiences. Following this initial read, a review of two transcripts by the entire coding team was conducted using a hybrid thematic analysis approach27,28 incorporating both inductive and deductive coding which allowed coders to gather unique themes and constructs that emerge from the data.29,30 A priori codes were established, guided by the theoretical construct and the research questions, to capture expectations. The initial codebook included constructs such as trust, wearability, day vs nighttime use, and food management. After reviewing, the coding team met to discuss transcripts, resolve disagreements, and correct all codes. This process informed the development of a codebook incorporating changes and additional constructs (eg, social activities, work challenges, and intimacy). Transcripts were uploaded to NVivo 12.0. 31 To maintain rigor in the coding process, two coding team members independently analyzed two to four transcripts each week until all transcripts were coded. All coders met weekly to discuss the transcript content, discrepancies, nuances, and memos of additional constructs. Modifications were made to the codebook and transcripts as needed. Once coding was complete, and all coders agreed with the results, the team sorted the coded data into major themes and used sub-themes to capture nuances and relationships between the data within and among the overall themes. 32
Results
Content analysis of focus group data resulted in four major themes: Diabetes Burden, Managing Glucose, Daily Routine, and User Experience. Fourteen sub-themes were identified within the four major themes (see Table 2). Themes reflected the daily experience of diabetes management using the BP and the perceived benefits and burdens of the system. Themes were consistent, regardless of respondents’ age and are therefore presented collapsed by age.
Summary of Themes and Sub-Themes.

Themes and Sub-Themes
Diabetes burden
Overall, adults reported the BP reduced diabetes-related burden, including improvements in the emotional toll of diabetes and the time spent managing and thinking about diabetes. However, adults also reported increased burden related to managing hypoglycemic events.
Emotional burden
Adults reported a reduced need to pay close attention to their glucose levels. One participant described: “a feeling of this is how normal people live . . . [I almost forgot] I had diabetes because the pump actually . . . corrected my blood sugars.” Another described using the BP as: I think it’s hard to get used to not being able to decide what you need to do. But . . . that’s a positive of the product, it’s doing it for you. So even if . . . I had to worry about diabetes 50% less a day, that’s still a huge thing.
Adults described being more open about their diabetes without feeling responsible or guilty about numbers. One adult shared: “I could be more transparent about what was going on because I felt less like I messed up.” Another adult said: “It [helped with] stigma . . . I was . . . able to travel and be with family . . . snacks everywhere . . . and there were definitely fewer questions.”
Time burden
Some reported spending less time managing diabetes because the BP: “did a lot of [managing my glucose levels] automatically.” However, for those experiencing ongoing issues with hypoglycemia, the time burden was worse, spending: “way more time correcting lows.”
Cognitive burden
Adults expressed feeling relief in not thinking about diabetes all the time. One participant shared: “it was thinking for me a lot more than I had to do. It was a big help [with] the highs and the lows.” The reduced need to count carbohydrates also reduced the cognitive burden associated with T1D: I don’t think that I had a clear sense of how much carb counting I do . . . until I didn’t need to carb count. And I found that to be . . . an incredibly mentally freeing experience.
Managing glucose
Adults commented on their experiences with how the BP manages glucose variability.
A1c and time in range
Many perceived improved glycemic outcomes, exemplified by one adult’s comment: “It definitely lowered my A1c . . . I texted my mom as soon as I got those results because I haven’t been in that good control since I was probably . . . 7 years old!” Another participant shared: “It was a little hard on my ego because it is actually substantially better controlling my blood sugar.” However, some found it did not improve time in range, exemplified by: “I stayed at both ends of the spectrum all day.”
Managing out of range glucose levels
Some had positive experiences with the BP responding to out-of-range numbers: “it dealt with high blood sugars faster than I expected it to.” Others had negative experiences: “It sometimes [would] overcorrect and cause a lot more lows.”
Most useful time of day
Most reported their glucose levels were most consistent during the night. For example: “overnight it’s a peace of mind that should not be overlooked and cannot be overstated”; “I found . . . even if I went to bed a little bit high . . . I’d wake up at 110 . . . to have that benefit to me [was] a wonderful thing.”
Algorithm learning my body
Many reported that the BP took longer to learn their body than expected. For example: The first month . . . my blood sugar . . . wasn’t in . . . range as much . . . it wasn’t till the first month and a half . . . that it started just being in range almost all the time.
Others experienced frustrations with waiting for the system to manage out-of-range numbers: [The nurses and doctors] would say don’t panic about [200s], but you . . . want to do something about it. You don’t want to ignore it. So, they said . . . just relax. It’ll come down . . . but that took a few months . . . [the BP] definitely changes and learns and adjusts.
Adults reported that once the algorithms learned their patterns, they felt greater freedom from daily diabetes tasks. For example: “I was completely blown away with how [the BP] learned . . . the algorithms of me . . . and my insulin resistance . . . I think also that’s where some of the freedom came in.”
Some shared concern that the BP was not able to accommodate frequent changes in schedules, such as working evening or swing shifts that require changes in mealtimes. Participants wished there was a way to build flexible schedules into the system expressing a desire to “easily mark . . . when I wake up in the morning saying this is a workday and then it . . . [would learn] that mode, [which is] different than my day-to-day life when I’m at home.”
Daily routine
Participants shared both the benefits and the burdens of the BP in their daily lives.
Meals
Many were pleased with how well the BP managed meals. For example, one participant shared their “testing” of their system: I’m gonna see how the BP handles Mexican food and . . . pizza, [which shoot] me . . . sky high . . . I ate my pizza and I started to rise a couple of hours later and it dropped me back down, and I was like . . . you’re my best friend.
However, some expressed frustration with how the system responded to snacking or unique mealtimes. For example, I [am] a grazer, I like to have snacks. And the BP doesn’t accommodate that at all. It’s breakfast, lunch, dinner, small, medium, large. So that was really kind of like tricky . . . I had to adjust my lifestyle to fit the BP, which I don’t want to have to do with diabetes technology.
Exercise
Many shared challenges with exercise, feeling the system did not permit spontaneity in exercise or regulate glucose levels as expected. One adult shared: “I . . . didn’t really exercise how I normally would. Because again, what’s the point? If I’m just going to have to come right back in 20 minutes and have a snack again.”
Interpersonal relationships
Responses to the impact of the BP on relationships were mixed. For example, one participant shared how their spouse was pleased with the BP as she was “happier with the lack of alarms and less lows.” However, some believed the BP burdened their spouse through glycemic variability, alarms, or alerts: “My wife was much more aware of my diabetes . . . than she normally is . . . especially just because of the lows.” Another stated, For me, it was worrying that the pump would start beeping in the middle of the night, and I had no control over it, and could I get out of the bedroom fast enough so it wouldn’t wake up my wife [which] made me anxious.
User experience
Participants discussed whether their expectations of the BP were met. They also shared their experiences with alarms, learning to give up control, and provided overall thoughts on their daily experiences.
Expectations
Most reported their expectations were met. For example: “Overall, I think it really met my expectations . . . my A1c improved, my blood sugars improved . . . I had a handful of days that were perfect days in range, which [are rare].” Another said, “it really did exceed my expectations because I was in range so much more of the time . . . and I didn’t have to monitor it all night, which was way better than I expected.” Another mentioned, “this device has kept me in a very safe range both for long term complications and short term, seizure concerns . . . it blew my expectations out of the water.”
However, some reported disappointment regarding the automation or the time required to manage the system. For example: “I anticipated [the BP] to be more automated and not need as much human thought . . . I was a little bit disappointed that I had to give it more thought.” Another adult shared, [I thought] I was gonna be able to set it and forget it . . . And I quickly learned that maybe [even though the study team] did warn me I was going to be at higher blood sugar levels at the beginning than I’d like, I also had expected that it would correct those higher blood sugars faster.
Alarms
Adults shared varied experiences with the alarms. One person commented: “The most helpful was the alarm to change the infusion set.” But another person added their frustration with the alarms going off at a time that was not critical, the alarm “would go off to change the insulin it woke me up and drove me nuts [especially when I had plenty to get me through the night].”
Giving up control
Adults had varying responses to the inability to control insulin dosing and delivery. Many expressed the uneasiness of giving up control after managing their insulin delivery needs for so many years. One adult stated, “giving up that control is very hard . . . I would have little waves of panic getting used to it.” But other adults expressed the relief of giving up control, “I really relinquished control and it was delightful.”
Overall experience
Overall, most were pleased with their experience, looking forward to the BP being approved for commercial use. Comments included: “I was just elated. I can’t say enough good things about my experience”; “It was nice to be on this because it was small, it was easy, and it felt pretty safe. It was good”; “I think it’s a marvelous device”; “I didn’t want to give it up”; “I had a handful of days that were perfect days in range . . . to see that in one month’s time . . . really met my expectations.”
Others provided critiques on different system elements that made the BP less user-friendly. For example, one participant disliked “the cartridge, all the steps involved, and then the screen timing out so much. It was a pain in the butt really.” Others expressed frustration with the frequent need for cartridge changes: “The size of the reservoir [was small and I was] actually forced to replace it every two to three days . . . I didn’t love it.”
Discussion
This study assessed user experiences among adults using the insulin-only BP, a new and advanced AID system that requires only limited user input. A qualitative analysis of focus group discussions revealed four major themes: diabetes burden, managing glucose levels, daily routines, and user experience.
This study adds to the growing literature on adult experiences with AID systems and answers the call1,7,12-15 to include the human factor in diabetes technology research. Although the features and management of AID systems vary greatly, the BP results were consistent with prior AID research3,5-11 in that participants reported several perceived benefits and challenges (ie, exercise continues to be an area for refinement). 33 Specifically, participants reported improvements in their overall perceived burden of living with diabetes, including a reduced emotional toll, reduced time spent managing diabetes, and reduced time thinking about diabetes. Improvements in glycemic outcomes were also noted, especially at night. Benefits related to managing difficult meals/foods were noted as well. Many reported that it exceeded their expectations, and they would want the system once it became commercially available.
Participants also reported several perceived burdens related to the BP. Some experienced increased time managing hypoglycemia, either due to over-correction or exercise. Others expressed frustration with the time it took for the system to learn their body and respond to out-of-range glucose levels. Participants also reported dissatisfaction with some aspects of the system design (eg, insulin capacity). Finally, although some participants were quite happy to give up control over insulin dosing, others struggled. These findings should be noted by both system developers and clinicians, as they prescribe different technologies and can inform future research exploring patient profiles that may guide system selection and education.
There are limitations to this study. Adult perspectives on the BP were only assessed during the post-trial period. Understanding the expectations of individuals at baseline would provide awareness of how expectations change, and this data could be used to develop educational guidelines related to realistic expectation-setting. These data could also facilitate device uptake and continued use. In addition, only one person in the emerging adult age group (19-25 years) participated. This specific age group may have a different experience than what is captured in this study, and efforts to capture the perceptions of individuals in this age group will be important. The development of a brief questionnaire, through a thorough review of the existing qualitative research on AID systems, could benefit these efforts as a larger sample of AID users could be reached. The perspectives of a broader range of AID users could strengthen our understanding of the lived experience and help inform further development and refinement of these systems to enhance uptake and diversify users.
Conclusion
As patients adopt new technologies, it is necessary to understand the long-term experiences of device wearers. Incorporating patient lived experiences will inform not only device development but also help clinicians feel comfortable teaching patients. In addition, identifying which patients may benefit the most from wearing these systems may inform future device developments and assist clinicians helping patients identify the systems that would best meet their individualized needs and expectations.
Footnotes
Acknowledgements
The authors thank all of the families who participated in the study. The authors also thank all of the research team members. Fast-acting insulin aspart and insulin aspart were provided by Novo Nordisk and insulin lispro was provided by Eli Lilly. Blood glucose meters and test strips were provided by Ascensia Diabetes Care. Continuous glucose monitor sensors and transmitters were purchased from Dexcom, Inc, at a discounted price. J.W.-B. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Abbreviations
AID, automated insulin delivery; BP, bionic pancreas; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.W.-B. reports consulting payments from Beta Bionics. M.F., K.R.H. and K.P.G. report no declaration of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases (grant no. 1UC4DK108612-01), by an Investigator-Initiated Study award from Novo Nordisk, and by Beta Bionics, Inc, which also provided the bionic pancreas devices used in the study.
