Abstract
Diabetic foot ulcers (DFUs) affect one in every three people with diabetes. Imaging plays a vital role in objectively complementing the gold-standard visual yet subjective clinical assessments of DFUs during the wound treatment process. Herein, an overview of the various imaging techniques used to image DFUs is summarized. Conventional imaging modalities (e.g., computed tomography, magnetic resonance imaging, positron emission tomography, single-photon emitted computed tomography, and ultrasound) are used to diagnose infections, impact on the bones, foot deformities, and blood flow in patients with DFUs. Transcutaneous oximetry is a gold standard to assess perfusion in DFU cases with vascular issues. For a wound to heal, an adequate oxygen supply is needed to facilitate reparative processes. Several optical imaging modalities can assess tissue oxygenation changes in and around the wounds apart from perfusion measurements. These include hyperspectral imaging, multispectral imaging, diffuse reflectance spectroscopy, near-infrared (NIR) spectroscopy, laser Doppler flowmetry or imaging, and spatial frequency domain imaging. While perfusion measurements are dynamically monitored at point locations, tissue oxygenation measurements are static two-dimensional spatial maps. Recently, we developed a spatio-temporal NIR-based tissue oxygenation imaging approach to map for the extent of asynchrony in the oxygenation flow patterns in and around DFUs. Researchers also measure other parameters such as thermal maps, bacterial infections (from fluorescence maps), pH, collagen, and trans-epidermal water loss to assess DFUs. A future direction for DFU imaging would ideally be a low-cost, portable, multi-modal imaging platform that can provide a visual and physiological assessment of wounds for comprehensive wound care intervention and management.
Keywords
Introduction
This manuscript aims to present a comprehensive overview, rather than a systematic review, of various imaging modalities employed in the management of diabetic foot ulcers (DFUs) and to discuss potential future developments in this area. Our intention was not to follow PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines or provide an exhaustive analysis of each imaging modality’s application in the study of DFUs. Instead, our focus was to identify, describe, and exemplify each modality’s usage in this field. The references were selected to provide representative examples of each modality, rather than a comprehensive bibliography. Our goal is to present an engaging narrative that delineates the status quo and anticipates future developments in imaging DFUs.
Diabetic Foot Ulcers
Approximately 537 million adults around the world have diabetes mellitus. 1 In the United States, 37 million people have diabetes. This accounts for 11.3% of the country’s population. 2 And an additional 96 million adults have prediabetes, which is 38% of the population. 2 The common complications of diabetes (neuropathy and ischemia) can predispose people to DFU infections. 3 The lifetime incidence rate of DFUs in people with diabetes (PWD) is 19 to 34%. 4 The recurrence rates of DFUs are 40% within a year and 65% within five years. 4 Approximately 20% of moderate or severe diabetic foot infections lead to some level of amputation. 3 Currently, visual inspections in conjunction with measurement of wound size reduction are the gold standard for assessing the healing status of DFUs. 5 Imaging plays a significant role to provide objective assessments of the DFU status and complements the subjective gold-standard visual inspection.
Background on Imaging Modalities and DFUs
Conventional Imaging Modalities to Assess DFUs
Imaging modalities such as magnetic resonance imaging (MRI), computed tomography (CT), ultrasonography (US), positron emission tomography (PET), or hybrid systems such as PET/CT, PET/MRI, and single-photon emitted computed tomography (SPECT)/CT have been used to image DFUs. Based on the imaging modality, they were used to assess the extent of musculoskeletal infections and impact on the underlying bones (osteomyelitis), anatomical foot deformities (e.g., Charcot foot) or dislocations, and peripheral arterial insufficiency. A summary of each of the modalities used as a stand-alone or as a hybrid modality, as well as their applications, advantages, and disadvantages is presented in Table 1. Four reasons why these imaging modalities are not practical for routine outpatients visits are that they: (1) may expose patients to radiation (except MRI or US), (2) may not be portable for a bedside examination, (3) do not offer wide availability, and (4) are expensive.
Conventional Imaging Modalities Used to Assess Musculoskeletal Infections, Bone Loss, or Peripheral Vascular Issues in Diabetic Foot Ulcers (DFUs).
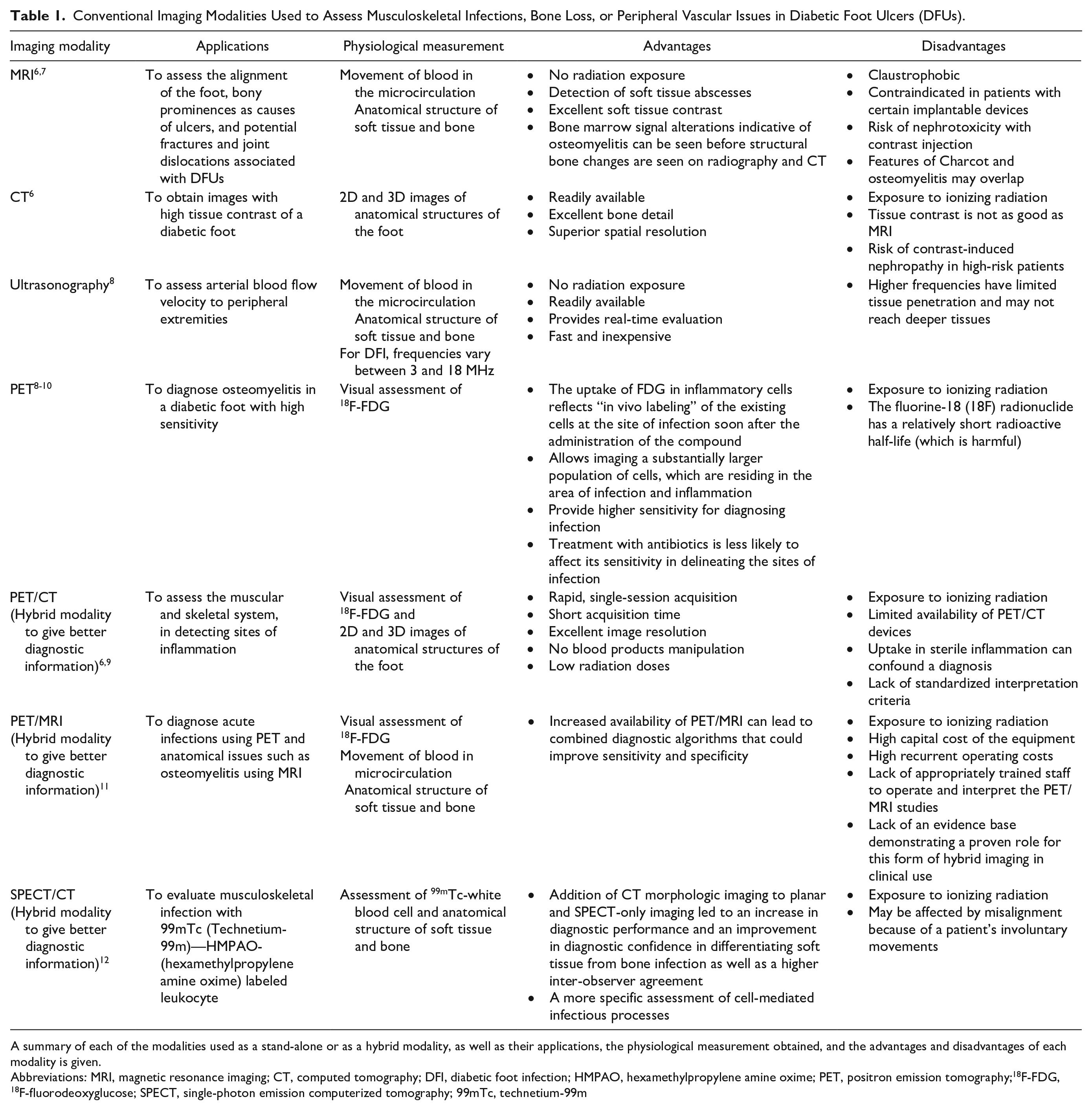
A summary of each of the modalities used as a stand-alone or as a hybrid modality, as well as their applications, the physiological measurement obtained, and the advantages and disadvantages of each modality is given.
Abbreviations: MRI, magnetic resonance imaging; CT, computed tomography; DFI, diabetic foot infection; HMPAO, hexamethylpropylene amine oxime; PET, positron emission tomography; 18 F-FDG, 18 F-fluorodeoxyglucose; SPECT, single-photon emission computerized tomography; 99mTc, technetium-99m
Physiological Assessment of DFUs
Physiological assessment of wounds during periodic wound treatment can aid in observing the underlying changes that typically manifest much earlier than visual apparent changes (the gold-standard wound assessment approach). The current clinical standard of care, however, does not include performing a physiological assessment of wounds as a regular practice. Measurements of tissue oxygen, such as the partial pressure of oxygen (using transcutaneous oximetry (TCOM) methods), in the DFU are clinically monitored in cases with peripheral arterial disease but are not regularly implemented in clinical practice to image all DFUs. 5 Oxygen delivery to the wound site is vital for the wound-healing process, as described in the following section.
Wound-healing process and oxygenation
Wound healing occurs in four sequential phases. These are hemostasis (aka bleeding), inflammation, proliferation, and remodeling. 13 For a wound to heal, an adequate oxygen supply is needed to facilitate reparative processes. 14 Hypoxic conditions from the initial wound onset, coupled with vasoconstriction to prevent blood loss, trigger the wound-healing process to start. The initial hypoxia also triggers the coagulation process associated with the hemostasis phase. 15 In both the inflammation and proliferation stages, increased oxygenation is crucial to healing success. Once hemostasis has been established, blood vessels dilate and capillary permeability increases to initiate the inflammatory phase and increase the wound oxygen supply. 16 The increased oxygen supply is used by macrophages and neutrophils to produce reactive oxygen species to prevent infection. 17 In the proliferation stage, adequate oxygenation is needed to assist with reparative processes such as re-epithelialization, collagen synthesis, cell proliferation, and angiogenesis.14,17 Adequate oxygen supply to the wound is essential for reparative processes such as cell proliferation, bacterial defense, angiogenesis, and collagen synthesis. 14 Without it, wound healing can become stagnant or even reverse, keeping the patient in a loop cycle of treatment. One technique that is used clinically to assess oxygenation in DFUs is TCOM, which is described in detail in the following section.
Gold-Standard Transcutaneous Oximetry for Perfusion Assessment
TCOM is the gold standard method for assessing perfusion in DFUs. 12 TCOM is contact-based and uses probes positioned in the peri-wound region for point location assessments of oxygenation. The probes contain a heating element to warm tissue to ~44°C and induce vasodilation from the cutaneous capillary loops for maximal oxygen diffusion in the skin. 18 Oxygen (O2) and carbon dioxide (CO2) diffuse from the skin into an O2/CO2 permeable membrane. A Clark electrode within the sensor head uses the permeated O2 to calculate the partial pressure of oxygen as an indirect measure of perfusion to the wound.
Hyperspectral imaging (HSI) and laser Doppler-based approaches are also used for assessment of perfusion, 12 but TCOM remains the most recommended. Laser Doppler-based techniques measure blood flow velocity, rather than oxygenation, which is diffused into the tissue from hemoglobin. In addition, HSI-based approaches can provide spatial oxygenation assessments in the form of oxygenation maps but cannot assess the ability of the tissue to provide oxygen to the wound area. One group recently investigated the ability of TCOM and HSI to predict wound healing and found TCOM to be a better predictor of healing. 19 Overall, TCOM has been shown to be a good indicator of healing across a range of ulcers. 20 It has been shown a good measure of assessing revascularization surgeries in PWD 21 and healing in DFUs.19,22,23
Despite the accolades, TCOM has limitations. TCOM is a contact-based approach that can only measure oxygenation at discrete point locations in the peri-wound; it cannot measure the partial pressure of oxygen in the wound bed.24,25 There is also substantial diversity in the setup of the probe temperature and placements, 26 and the method can require as much as 45 minutes to make a measurement.25,27 In other words, TCOM setup is non-standardized and the time-intensive nature of TCOM can complicate weekly treatments of DFUs. Overall, TCOM is regularly used in clinical practice, both for the initial assessment of peripheral arterial disease and as needed.12,26
Optical Modalities for Oxygenation/Perfusion Measurements
Optics-based imaging techniques (using visible and/or near-infrared [NIR] light) have been used to assess oxygenation and perfusion in tissue. The rationale behind the various attempts has been to improve the wound treatment process.
In short, the fundamental principle behind optical implementations for oxygenation monitoring is that signal attenuation (photon absorption) in tissue is proportional to the concentration of light-absorbing objects—or chromophores. Light in tissue can be either absorbed and/or scattered based on the wavelength. If scattered, then the photon path changes direction and becomes diffused into the tissue media. If not completely absorbed, then the photons eventually backscatter from the illumination surface (or diffusely reflect) or diffusely transmit out of the tissue. If diffusely reflected or transmitted out of the media, then the measured signal contains information about the chromophore concentrations (e.g., oxy- and deoxyhemoglobin) in the tissue. While transcutaneous oxygenation measurements using TCOM are direct measures of tissue oxygen, oxygen saturation and related hemoglobin measurements of tissue oxygenation are indirect measures of tissue oxygen. Several optical imaging modalities can assess tissue oxygenation changes (in terms of oxygen saturation and related hemoglobin parameters).
Table 2 provides an overview of various optical modalities that have been applied to DFUs. The fundamental principle of each of the modalities used for DFU imaging, their key findings, and their limitations are described below.
A List of Non-invasive Optical Imaging Modalities That Have Been Used for Imaging DFUs Is Provided.
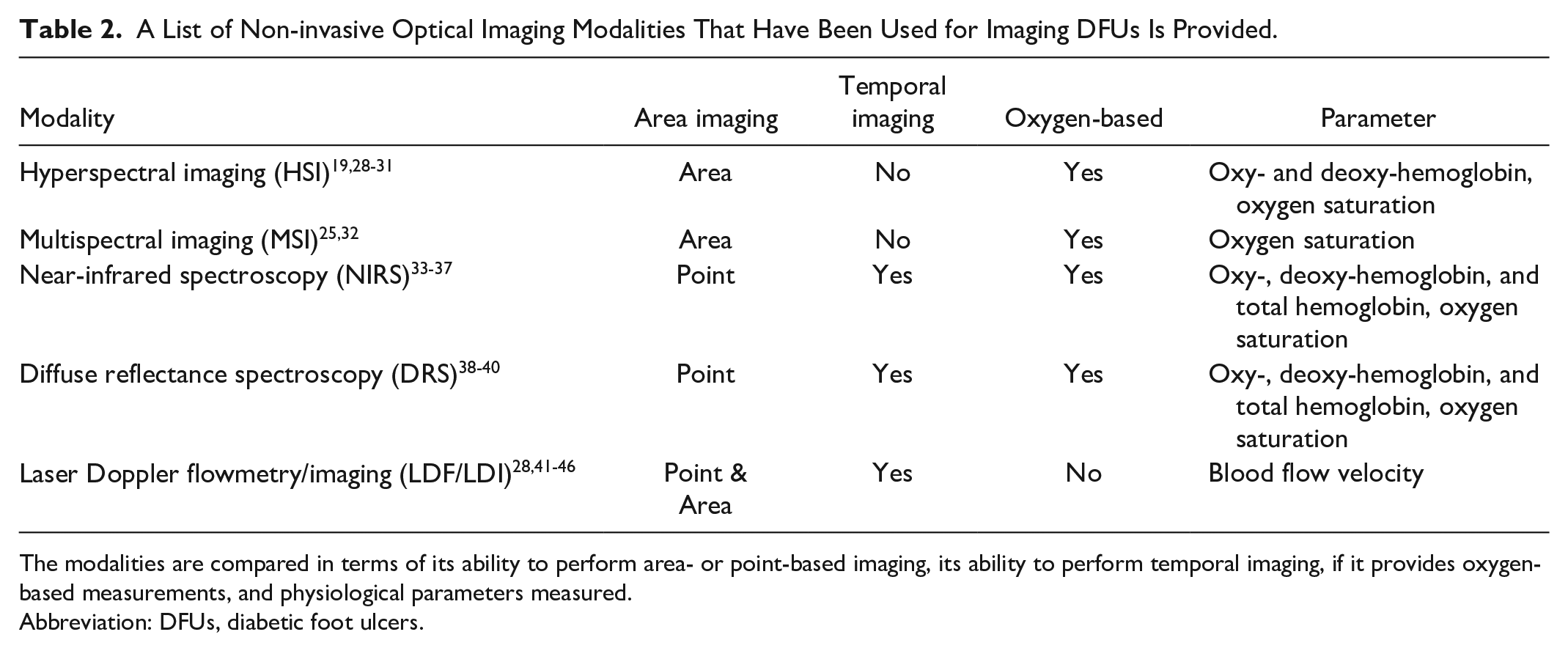
The modalities are compared in terms of its ability to perform area- or point-based imaging, its ability to perform temporal imaging, if it provides oxygen-based measurements, and physiological parameters measured.
Abbreviation: DFUs, diabetic foot ulcers.
Diffuse reflectance spectroscopy
Diffuse reflectance spectroscopy (DRS) typically employs broadband sources that produce light in the 400-800 nanometer (nm) range. DRS measurements can be reported in terms of hemoglobin-based parameters such as oxy-, deoxy-, and total hemoglobin and oxygen saturation. DRS-based techniques can be used for continuous data acquisition but are typically contact-based for point location assessments of tissue. 47
Calluses, ulcers, and healthy skin in DFUs can be discriminated via DRS. 48 In general, however, DRS-based studies have been conducted toward preventing the development of DFUs and monitoring DFU healing. 49 proposed a method through DRS to predict hypoxic conditions in non-ulcerated diabetic feet, and 50 imaged controls and prediabetic subjects and found a difference in the reflectance spectra of the two groups. A few research groups converted the DRS data into hemoglobin-based concentration data when applied to subjects with or without diabetes. One group 38 found that, on average, PWD, compared to people without diabetes, had lower levels of total hemoglobin in the foot. Two other research groups Rajbhandari et al 39 and Anand et al 40 found that, in healing DFUs, oxyhemoglobin concentrations decreased prior to ulcer closure. The findings of Rajbhandari et al 39 and Anand et al 40 are in agreement with near-infrared spectroscopy (NIRS)-based reports described further below.
Hyperspectral imaging
HSI is a prominent non-contact optical modality that has been used to assess DFUs. HSI utilizes a broadband source of visible and NIR wavelengths and a series of optical filters to capture reflectance data at discrete wavelength ranges. By imaging narrow spectral bands over a continuous spectrum, the reflectance spectra can be obtained, by pixel, and used to calculate oxygenation parameters.51,47 While HSI devices typically capture images, HSI can be used for point-based assessments of blood flow in PWD. 52
One study reported that calluses were associated with decreased oxyhemoglobin prior to ulceration, 53 but in general, the bulk of the work has been directed toward developing predictive healing indices through HSI-based approaches. Using hemoglobin-based oxygenation measurements from the oxygenation maps, various teams were able to develop predictive healing indices with high sensitivity (80-95%) and specificity (71-86%) from one to six months’ worth of data.19,28-31 However, there were caveats. For example, some groups found that, between healed and non-healed DFUs, healed DFUs had higher oxygenation levels at baseline.28,29 However, other groups reported the opposite.31,52 Still others did not comment on the oxygenation changes between baseline and later dates. 19 It would appear that spatial oxygenation mapping alone is not enough to completely characterize DFUs.
Multispectral imaging
MSI techniques are similar in principle to HSI but utilize fewer spectral ranges. What defines a multispectral modality, however, is not standardized. While it is accepted that MSI-based techniques acquire electromagnetic signals with fewer spectral bands, there is no definition of how many bands an MSI device uses nor the range of each band. 54 Regardless, by utilizing fewer wavelengths, calculated parameters are less prone to artifacts because less complex modeling approaches are used. 47 MSI-based devices have been applied to wounds.32,55-57 However, studies using MSI that specifically recruited subjects with DFUs are limited. Basiri et al 32 applied MSI-based imaging to wounds, including DFUs. The group reported that, by the final week of imaging, the wound size as well as the oxygen saturation in the wound decreased when compared to the initial week of imaging, 32 possibly from coagulation and scar formation during the wound-healing process. 55 One recent pilot study showed that measurements of partial pressures of oxygen from TCOM measurements and non-contact NIRS imaging were correlated. 25 Non-contact oxygen saturation measurements could be converted into partial pressures of oxygen using the Severinghaus oxygen dissociation curve. They found that both measurements had a 75% correlation with each other.
Laser Doppler flowmetry and imaging
The laser Doppler principle states that when photons strike a moving object (such as a circulating blood cell), the photon undergoes a change in wavelength and frequency (Doppler shift). Impact with a static object does not cause a Doppler shift. A laser is used to illuminate tissue and the backscattered light can be used to calculate the blood flow. 47 Based on the setup, the technique can be used for point laser Doppler flow (LDF) and area-based (laser Doppler imaging or LDI) measurements. Laser Doppler-based techniques, however, cannot be used to calculate hemoglobin-based oxygenation but can calculate perfusion-based measurements of blood flow.
Laser Doppler-based techniques have been used extensively to assess perfusion in PWD both with and without foot wounds. Using LDF-based approaches on PWD without wounds, it was found that the microvascular perfusion (and skin blood flow) was overall lower than that in control subjects (i.e., patients without diabetes).58,59 However, in response to a heating stimulus, the PWD had a greater increase in perfusion in comparison to the controls. LDF-based approaches found that people with type 2 diabetes without wounds, compared with control subjects, had an increased blood flow response to pressure offset 60 and a lower vascular response to heating. 61
In DFUs, LDF has frequently been used to study how different therapies affect DFU healing. One such therapy is carbon dioxide (CO2) therapy, where CO2 is transcutaneously or sub-cutaneously administered for therapeutic effects on both microcirculation and tissue oxygenation. It has been reported that both CO2 therapy, 41 local heating, and electric therapy 42 improved cutaneous blood flow and improving healing rates. One study reported that skin blood flow was higher in healed DFUs as opposed to DFUs that did not heal. 43 However, another study reported that the skin blood flow in healed compared to non-healing wounds was not different. 28 It would appear, it seems, that, similar to HSI, blood flow monitoring alone is not conclusive.
LDI has also been used to study microvascular blood flow, but not as frequently as LDF. Two studies found that dermal replacements increased microvascular blood flow in DFUs.44,45 In on mixed population wound study where DFUs were imaged, it was found that: (1) DFUs with greater blood flow tended to heal faster and had higher capillary density 46 and (2) electrical stimulation increased blood flow to the skin. 42
Near-infrared spectroscopy
NIRS utilizes NIR light and capitalizes on the biological optical window within the range of 650-900 nm to obtain physiological information about the imaged tissue. 62 Light in this wavelength range is minimally absorbed and preferentially scattered, allowing its deeper penetration to identify the differences in the extent of absorption of the major tissue chromophores within the biological tissues. Multiple chromophores exist in skin, but the main chromophores of skin at the NIR region are oxy- and deoxyhemoglobin, water, and melanin. 63
NIRS has typically been used for point source measurements to assess tissue oxygenation of DFUs. NIRS has a well-established background in assessing DFU healing. Several NIRS-based studies have reported that, as DFUs healed and approached closure, a reduction in oxyhemoglobin concentration was observed.33,34,64 During the inflammatory phase of wound healing, oxyhemoglobin concentration increases because of angiogenesis. This angiogenesis stops during the late proliferation phase, causing a decrease in the supply of oxygenated blood. Thus, the oxy- and total hemoglobin tend to decrease with healing. An additional study also reported that oxy- and total hemoglobin decreased in healing wounds toward concentrations similar to the background tissue. 35 One group applied NIRS to study the effects of low-level light therapy on DFUs. 65 They found that low-level light therapy increased total hemoglobin in the feet of patients with DFUs but not the feet of control subjects. A notable advance in NIRS-based technology has been the development of portable NIRS devices. Portable NIRS-based devices are being developed for optical brain imaging 66 and also for DFUs assessment. One group has determined a relationship between healing and Buerger exercises (non-invasive physical therapy to promote perfusion in lower extremities, which includes leg elevation, feet flexion/extension then pronation/supination, and laying down) using a custom, portable, NIRS-based device for point location assessment. They have reported that Buerger’s exercises increased oxy- and total hemoglobin and that most of the DFUs of the recruited subjects had improved toward healing.36,37 In more recent work, the same research group has reported that healing and non-healing patients with and without peripheral arterial disease had notable differences in hemoglobin-based parameter concentrations pre- and post-exercise. 67 As previously stated, however, these technologies are being applied as point-based assessments, instead of providing a spatial map of the entire wound region.
Spatial frequency domain imaging
Spatial frequency domain imaging (SFDI) employs patterned visible and NIR illumination to non-invasively determine hemoglobin (superficial and subsurface total hemoglobin concentrations) and oxygen saturation from deeper tissues (3-4 mm) and over a wide field of view. In a recent study on DFUs, SFDI 68 has been used to assess the microcirculation in DFU tissues and identify patients with the highest risk of ulceration and predict the ulcer onset. From a study on 252 subjects, the total hemoglobin in the papillary dermis and tissue scattering-based SFDI biomarkers predicted the onset of new ulcers with a sensitivity/specificity of 68.8/64.8 and 75.0/69.1%, respectively. SFDI is an emerging technology and has demonstrated the potential to develop a microcirculation-based biomarker for DFU predictions. However, the challenge in this technology is the precision required in performing imaging studies. The technique is very sensitive to movement when using laser-based patterned illumination that assesses scattering, apart from absorption features of the foot.
In summary, indirect assessments of perfusion via TCOM-based oxygenation measurements remain the gold standard for DFU assessment. However, TCOM cannot provide full insight into the oxygenation across a two-dimensional (2D) area and this method is time-consuming. DRS, NIRS, MSI, and HSI-based techniques have demonstrated that, as DFUs heal, the oxy- and total hemoglobin decrease with wound closure. Oxygenation monitoring from point locations (NIRS/DRS) and across an area (HSI/MSI/SFDI) can be used to predict healing. Healing assessments can be from periodic oxygenation assessments ranging from weeks to months. Hence, they cannot provide an immediate assessment. Laser Doppler-based techniques can provide immediate insight into the cutaneous blood flow of a tissue region, via blood flow velocity, but not tissue oxygenation changes. Temporal hemoglobin-based tissue oxygenation assessments can provide quantitative insight into the availability of oxygen to the imaged tissue, as well as how well oxygenated the blood flow is to a region. Hence, the ability to assess the variations in oxygenated flow along with spatial oxygen distribution across a 2D area can provide a comprehensive insight into the extent of oxygenation and its related flow to the entire wound and its surrounding area and not just at point locations in and around the wound.
An Oxygenation Flow-Based Imaging Approach Using NIRS
Recently, we developed a NIRS-based optical imaging device, or a near-infrared optical scanner (NIROS), that can perform spatio-temporal oxygenation mapping of tissues. 69 This non-contact imaging device can obtain 2D spatial and dynamic maps of tissue oxygenation (in terms of oxy-, deoxy-, and total hemoglobin and oxygen saturation). Tissue oxygenation flow changes are measured by utilizing an innovative breath-hold (BH) stimulus that induces vasoconstriction and determines the differences in the flow patterns in and around the tissues. 70 The BH-induced oxygenation changes can be used as a stimulus to assess the ability of peripheral vasculature to respond to an oxygenation demand in diseased tissue models such as DFUs. 70 Thus, the measurement of tissue oxygenation using our approach is like that of TCOM which also assesses how the vasculature responds in DFUs. The added advantage of our NIRS technique (using NIROS) is its ability to image the entire wound region, unlike discrete point measurements by TCOM. Additionally, the overall imaging time using our imaging approach is reduced to <2 minutes in comparison to the 30+ minute procedure using the gold-standard TCOM.
Unlike the spatial distribution of oxygen saturation measured by various researchers to assess healing, our innovative approach uses the temporally changing spatial maps of oxygenation saturation to determine the extent of synchrony or asynchrony in the oxygenation flow patterns. These variations in oxygenation flow patterns were quantified into a novel oxygenation flow index (OFI), ranging from -100% to +100%, and were used to differentiate the non-healing from healing DFUs. Preliminary studies on control subjects and subjects with DFUs (healing and non-healing) showed distinct differences in OFI across the subject groups. A preliminary threshold (OFI < 28%) could differentiate non-healing and complicated DFUs from healing DFUs. The overall oxygenation flow pattern was less synchronous (or the OFI value was reduced) in the non-wound areas of the feet that were non-healing. In other words, the reduced OFI value (<28%) in the entire foot excluding the wound region is a possible indicator that the wound may not heal. 71
Effect of skin tone on oxygenation flow-based measurements
NIR light is significantly attenuated with an increase in melanin concentration (which occurs with darker skin colors) in the epidermis. It is essential to account for this melanin-related attenuation in the epidermis to evaluate the changes in tissue oxygenation in the dermis and lower layers of the skin. Most of the optical imaging studies (using HSI, MSI, NIRS, etc.) studies do not account for the effect of melanin in their tissue oxygenation calculations. Hence, the hemoglobin-concentration profile plots are inherently influenced by the presence of melanin, limiting the application of the optical imaging to lighter skin color (that have less effect of melanin on the tissue oxygenation measurements). Unlike tissue oxygenation-based measurements, the temporal flow-based measurements are not impacted by melanin and only related to variations in hemoglobin parameters in response to the BH stimulus, based on our preliminary studies on a few DFU cases. 72 Thus, in a temporal setting the contribution of melanin to the measured hemoglobin-based signal can be disregarded. This may allow our non-contact spatio-temporal-based oxygenation flow-based monitoring method to be applicable across different skin colors, so that this method can be effective for various races, including African Americans and Hispanic/Latinos. Currently, extensive Monte-Carlo-based simulation studies are being carried out systematically to determine the effect of melanin on the oxygenation flow changes and determine the limits of the technology and the smallest flow pattern change that can be captured in the darkest skin-colored tissue.
Various imaging modalities for physiological assessments of DFUs
While the optical imaging modalities are primarily intended to measure oxygenation or blood flow to the wound site when assessing DFUs, there are various other physiological measurements using different imaging modalities that have been used by researchers to assess the healing status of DFUs. These parameters include thermal measurements, auto-fluorescence measurements for bacterial load, trans-epidermal water loss (TEWL) measurements to determine water content, pH measurements, and collagen assessment to determine structural changes during healing. All these measurements are obtained at the local wound site and are not focused on measurements obtained from a systemic response of the body to the presence of DFUs. These parameters (other than tissue oxygenation or related perfusion) that are measured in DFUs to monitor their healing status are summarized in Table 3 (the references are selective examples for each parameter and not a complete review).
Various Imaging Modalities for Physiological Assessment of DFUs, Along With Their Principle, Parameters Measured, and Major Observations Made in Wound Monitoring.
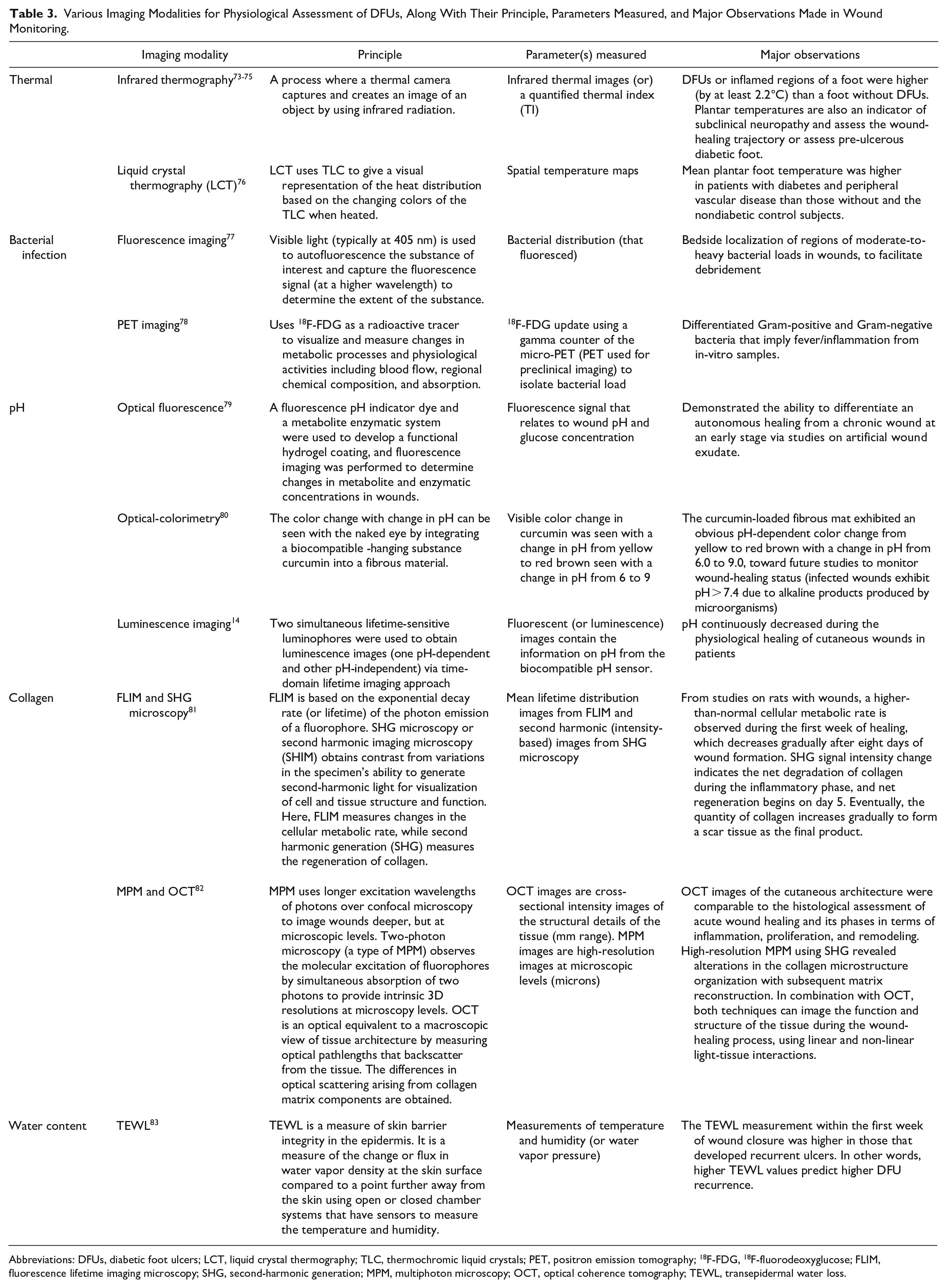
Abbreviations: DFUs, diabetic foot ulcers; LCT, liquid crystal thermography; TLC, thermochromic liquid crystals; PET, positron emission tomography; 18F-FDG, 18F-fluorodeoxyglucose; FLIM, fluorescence lifetime imaging microscopy; SHG, second-harmonic generation; MPM, multiphoton microscopy; OCT, optical coherence tomography; TEWL, transepidermal water loss.
Future Direction of DFU Imaging to Assess Healing
The conventional imaging modalities presented in Table 1 cannot be implemented as a routine inexpensive approach to assess healing during periodic outpatient visits by DFU patients to the clinic or hospital. This barrier to technology can be overcome by using low-cost bedside imaging techniques that measure multiple physiological parameters using optical and other inexpensive imaging techniques.
Each technique focuses on a single parameter of interest to assess healing. Wound healing is a complex process and multiple physiological changes occur simultaneously. There is a need to measure more than one physiological parameter to monitor the wound-healing status or predict its onset or recurrence. This approach can require the use of several low-cost devices sequentially during patients’ clinical visits, although this approach can lengthen the time of stay and can increase the overall costs of wound management. Hence, there is a need to develop a multi-modal assessment approach where more than one physiological parameter can be determined simultaneously via a single device.
Several smart sensors are currently developed as bandages to measure multiple physiological parameters such as pH, temperature, pressure, and/or oxygen saturation. However, these are point-based measurements at a single location and cannot assess the changes across the entire wound and its surroundings. Obtaining the physiological pattern across an entire wound region is vital to determine the true problematic regions for where interventions would be.
For instance, we envisage the near-term use of fluorescence-guided debridement of wounds to reduce chronic inhibitory bacterial load. This could be extended to a closed-loop system in the future with advances in drug delivery or other wound optimization techniques. These multi-modal devices should be bedside, portable, and easy to use and provide objective physiological assessments to complement subjective clinician expertise. 84
Some of the more advanced parameters that provide an insight into wound-healing status are temperature, bacterial load, tissue oxygenation, and perfusion (as shown in Figure 1).85,86 A multi-modal imaging device that measures several physiological parameters across an entire wound region should be our focus as we move forward to improve the quality of wound care management in DFUs. An effort to combine one or more of these parameters into a single device is our ongoing effort in the field of DFU wound monitoring.
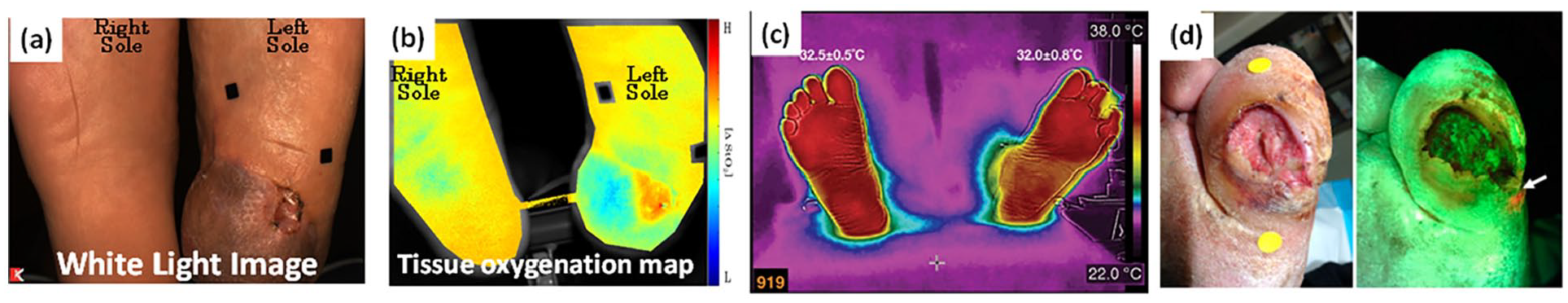
Assessment of foot ulcers as (a) white light digital color images of left foot with foot ulcer and right foot without foot ulcer (non-diabetic case). 85 (b) Tissue oxygenation map in terms of oxygen saturation (∆StO2) obtained using our hand-held near-infrared optical scanner of the left foot with foot ulcer and right foot without foot ulcer—a collaborative study with Univ of Miami Wound Care Center (Kirsner and team). 85 (c) Thermal maps of feet with recurrent ulcers in the left foot in a patient with diabetes 86 (Figure 1c reproduced under the terms of the Creative Commons Attribution License [http://creativecommons.org/licenses/by/4.0/]). (d) White light digital color image and the corresponding fluorescence maps from heavy growth of bacteria in a DFU case 87 (Figure 1d reproduced under the terms of the Creative Commons Attribution License [http://creativecommons.org/licenses/by/4.0/]). The square black stickers in (a) and (b) and the circular yellow stickers seen in the wounds are for the device’s wound measurement software. Abbreviation: DFU, diabetic foot ulcer.
Footnotes
Acknowledgements
The authors would like to thank the members of the Optical Imaging Laboratory, collaborating clinicians, and staff at the University of Miami Wound Care Center (our clinical imaging site).
Abbreviations
CT, computed tomography; DFI, diabetic foot infection; DFU, diabetic foot ulcer; DRS, diffuse reflectance spectroscopy; FDG, fluorodeoxyglucose; FLIM, fluorescence lifetime imaging microscopy; HMPAO, hexamethylpropylene amine oxime; HSI, hyperspectral imaging; LCT, liquid crystal thermography; LDF, laser Doppler flowmetry; LDI, laser Doppler imaging; MPM, multiphoton microscopy; MRI, magnetic resonance imaging; MSI, multispectral imaging; NIR, near-infrared; NIRS, near-infrared spectroscopy; OCT, optical coherence tomography; OFI, oxygenation flow index; PET, positron emission tomography; PRISMA, preferred reporting items for systematic reviews and meta-analyses; PWD, people with diabetes; SFDI, spatial frequency domain imaging; SHG, second-harmonic generation; SPECT, single-photon emission computerized tomography; TCOM, transcutaneous oximetry; TEWL, trans-epidermal water loss; US, ultrasonography; 99mTc, technetium-99m
Declaration of Conflicting Interests
All authors (except Dr. Klonoff) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr. Klonoff is a consultant to Better Therapeutics, Eoflow, Integrity, Lifecare, Nevro, Novo, Sanofi, and Thirdwayv.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is partially supported by NIH-NIDDK (F31DK125153), NIH-NIBIB (1R01EB033413-01), NIH-NIDDK’s DiaComp Pilot Grant (5U24DK115255-02), and FIU-BME Coulter Seed Funds and CURE (Coulter Undergraduate Researh Experience) funding to Godavarty and team. This study is partially supported by NIH-NIDDK 1R01124789-01A1 and NSF Center for Stream Healthcare in Place (#C2SHIP) CNS Award No. 2052578 to Armstrong and team.
