Abstract
Introduction:
Diabetes mellitus (DM) is a chronic disease with high morbidity and mortality, and glycemic control is key to avoiding complications. Technological innovations have led to the development of new tools to help patients with DM manage their condition.
Objective:
This consensus assesses the current perspective of physicians on the potential benefits of using smart insulin pens in the glycemic control of patients with type 1 diabetes (DM1) in Spain.
Methods:
The Delphi technique was used by 110 physicians who were experts in managing patients with DM1. The questionnaire consisted of 94 questions.
Results:
The consensus obtained was 95.74%. The experts recommended using the ambulatory glucose profile report and the different time-in-range (TIR) metrics to assess poor glycemic control. Between 31% and 65% of patients had TIR values less than 70% and were diagnosed based on glycosylated hemoglobin values. They believed that less than 10% of patients needed to remember to administer the basal insulin dose and between 10% and 30% needed to remember the prandial insulin dose.
Conclusions:
The perception of physicians in their usual practice leads them to recommend the use of ambulatory glucose profile and time in range for glycemic control. Forgetting to administer insulin is a very common problem and the actual occurrence rate does not correspond with clinicians’ perceptions. Technological improvements and the use of smart insulin pens can increase treatment adherence, strengthen the doctor–patient relationship, and help improve patients’ education and quality of life.
Introduction
Diabetes mellitus (DM) is a group of metabolic disorders that have in common the presence of persistent hyperglycemia. 1 Diabetes mellitus is a chronic disease with high morbidity and mortality due to its complications, which decreases the quality of life and the life expectancy of those affected. It has consequences for the family and community environments.2,3 Although phenotypically distinct, the most common subgroups of diabetes are type 1 diabetes (DM1), in which there is absolute insulin deficiency due to autoimmune destruction of pancreatic beta cells, and type 2 diabetes (DM2), which is fundamentally characterized by the presence of insulin resistance.4,5
The most common type of diabetes in childhood and youth is DM1. The number of cases has increased worldwide, and high incidence rates are found in Europe. An observational study to identify the profile of patients with DM1 in Spain identified the factors that may influence glycemic control: age, motivation, compliance, therapeutic education, and disease management skills, as well as fear of hypoglycemic episodes. To prevent the worsening of patients, glycemic control is the gold standard. 6 Within glycemic control strategies, the detection of glycated hemoglobin (HbA1c) has long been the best test to assess the risk of chronic complications from diabetes. Although the glycemic target must be personalized, the American Diabetes Association (ADA) and other scientific societies recommend, in nonpregnant adults, an HbA1c target of <7% without hypoglycemia.3,7
Continuous glucose monitoring is an increasingly used tool in managing patients with DM1.8,9 Current recommendations support the use of an ambulatory glucose profile (AGP) to measure glycemic control, with a target time in range (TIR) >70% and a time below the range of <4%. 10
Smart insulin pens are among the many technological innovations in managing patients with diabetes. The US Food and Drug Administration (FDA) approved the first reusable smart insulin pen in 2016.11 -13 Among its advantages, the insulin dose reminder stands out, facilitating the calculation of the next bolus.14,15
Real-life studies in Sweden have shown that using smart insulin pens improves glycemic control and insulin management in patients with DM1.16,17
This study aimed to assess the current perspective of physicians on the potential benefits of using smart insulin pens in the glycemic control of patients with DM1 in Spain.
Methods
The Delphi methodology is a structured process that collects information from a group of experts through questionnaires until consensus is reached through rounds.18,19 It is one of the most widely used methods to explore knowledge in areas that are difficult to cover based on evidence, 20 allowing the development of prescription indicators, 21 indicators that reflect the perspectives of patients and physicians, or indicators of care quality. 22
In this study, the Delphi methodology was used through a group of 100 endocrinologists and 10 pediatricians, with endocrinology subspecialty, who were selected according to the following criteria: extensive experience as doctors in managing DM and having a professional practice that was developed in Spain. They were invited to participate in the consensus by e-mail, and the surveys were conducted anonymously online.
The Steering Committee conducted a literature review of the literature in Medline/PubMed to identify evidence gaps from 2012 to 2022, with the following three search strategies: (1) (“clinical reality”) AND (control) AND (DM1 OR “diabetes mellitus type 1”), (2) (technology OR telemedicine OR “digital health” OR “mHealth”) AND (control OR “diabetes management” OR/AND (DM1 OR “diabetes mellitus type 1”), and (3) (“smart insulin pens”) AND (“patient profile” OR “patient characteristics”). The Steering Committee developed the content of the surveys in each round, monitored the responses, and drafted the summaries.
The survey consisted of 94 questions that were distributed in the following blocks of knowledge: block 1 (38 questions): the assessment of the clinical reality of the control of DM1 in the consultation; block 2 (40 questions): the advantages of the use of technology to improve the control of DM; and block 3 (16 questions): the profile of the candidate patient for smart insulin pens.
Two rounds were carried out with the experts: the first between February 25 and April 25, 2022, and the second between May 23 and June 27, 2022. For the second round, the questions on which there was no consensus in the first round were reformulated by the steering committee.
Statistical Analysis
Questions with discrete quantitative responses to each item were evaluated using a Likert scale from 0 to 11 points (0 = completely disagree; 10 = completely agree). The consensus criteria used were as follows: for agreement, a median ≥8 and an interquartile range (IQR) ≤0.4; for disagreement, a median ≤2 and an IQR ≤0.4. Questions with categorical answers were evaluated using the distribution of frequencies and percentages. The consensus criterion for the agreement for the questions with categorical nominal answers was that one of the answers reached ≥50% of the total number of mentions. The questions with ordered responses were evaluated using the frequency distribution, percentage, mean order, standard deviation, and coefficient of variation. The consensus criterion for the agreement was when the ranking positions obtained a coefficient of variation ≤0.3.
The data were analyzed using Gandia Barbwin® (Tesi S.L., Valencia, Spain) software Version 7.0.2110.5 and XLSTAT Version 21.04 of Microsoft Excel® (Microsoft Corporation, Washington, USA).
Results
In the first round, 100 endocrinologists and 10 pediatricians (with endocrinology subspecialty) participated, and 75% of the participants (90% of pediatricians) continued the study in the second round. The consensus obtained after completing the study was 95.74%. The experts’ most relevant recommendations in each section are shown in Figures 1 and 2.
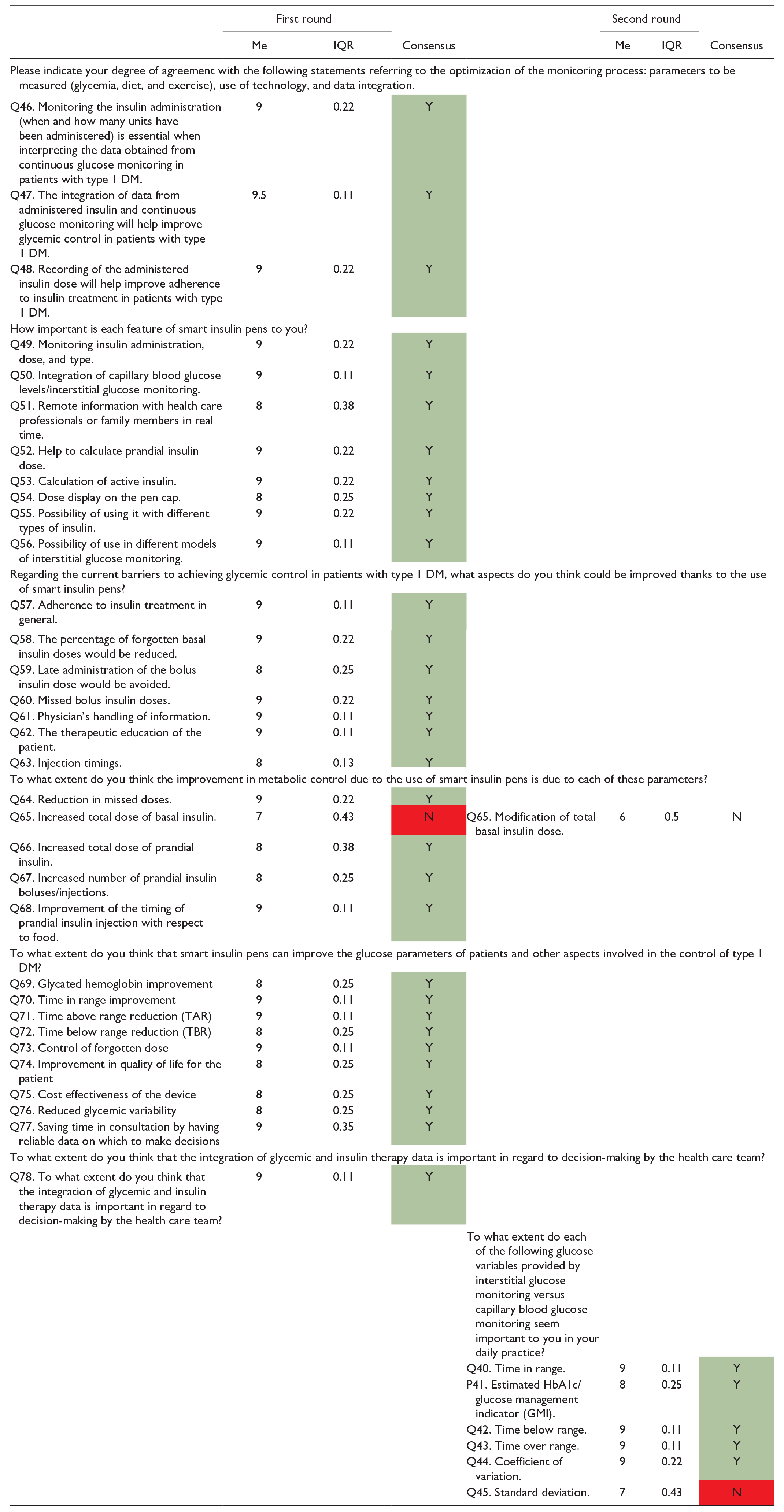
Most relevant recommendations are provided by the experts on assessing the clinical reality of the control of DM1 in consultations and the advantages of using technology to improve DM control.
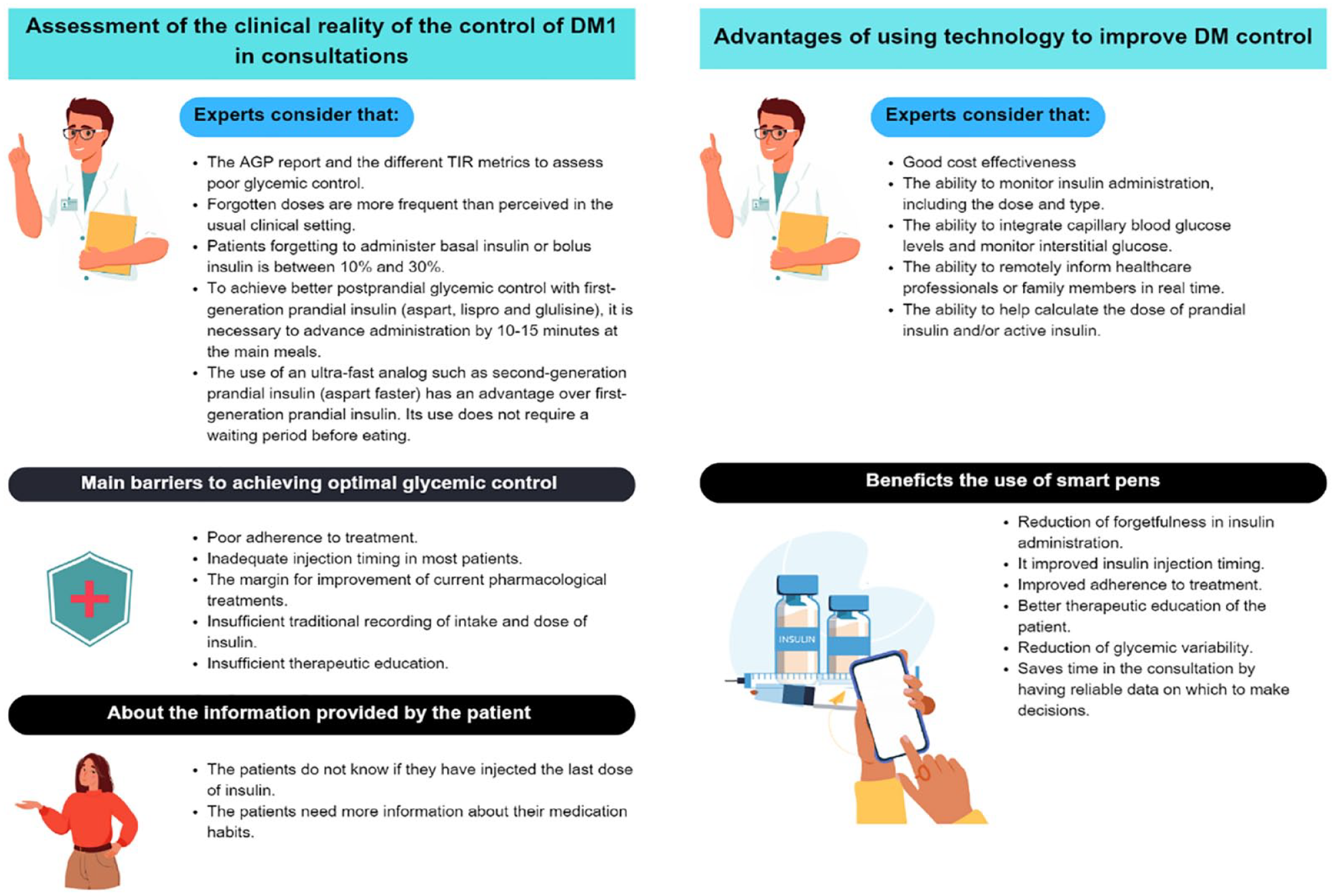
Most relevant recommendations are provided by the experts on the profile of the patient candidate for smart insulin pens.
Assessment of the Clinical Reality of the Control of DM1 in Consultations
The experts reached a consensus of 86.84% agreement in the first round and 94.74% in the second round. The results are shown in Tables 1 and 2. The experts answered the assessments using their experience in managing patients with DM1.
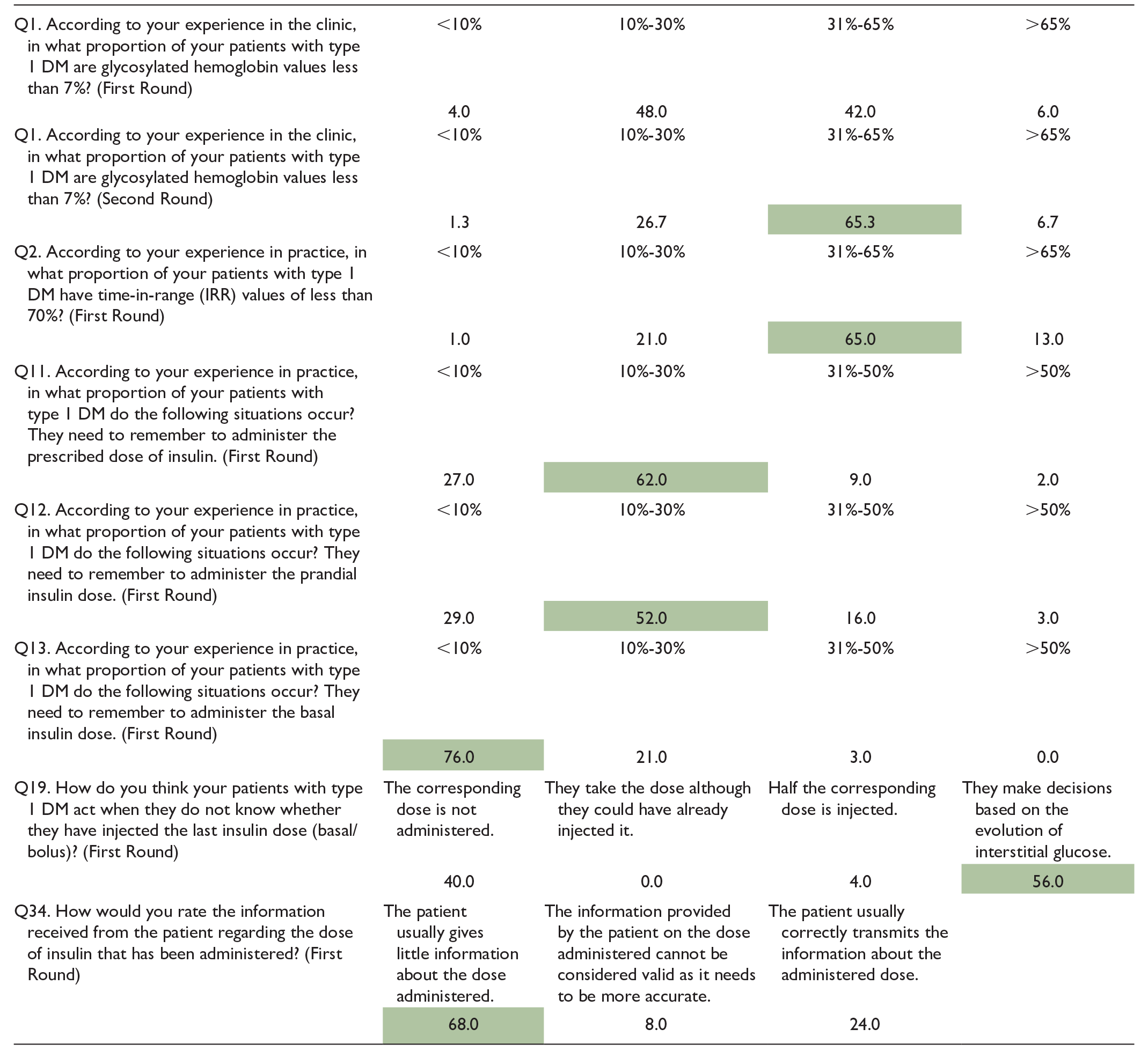
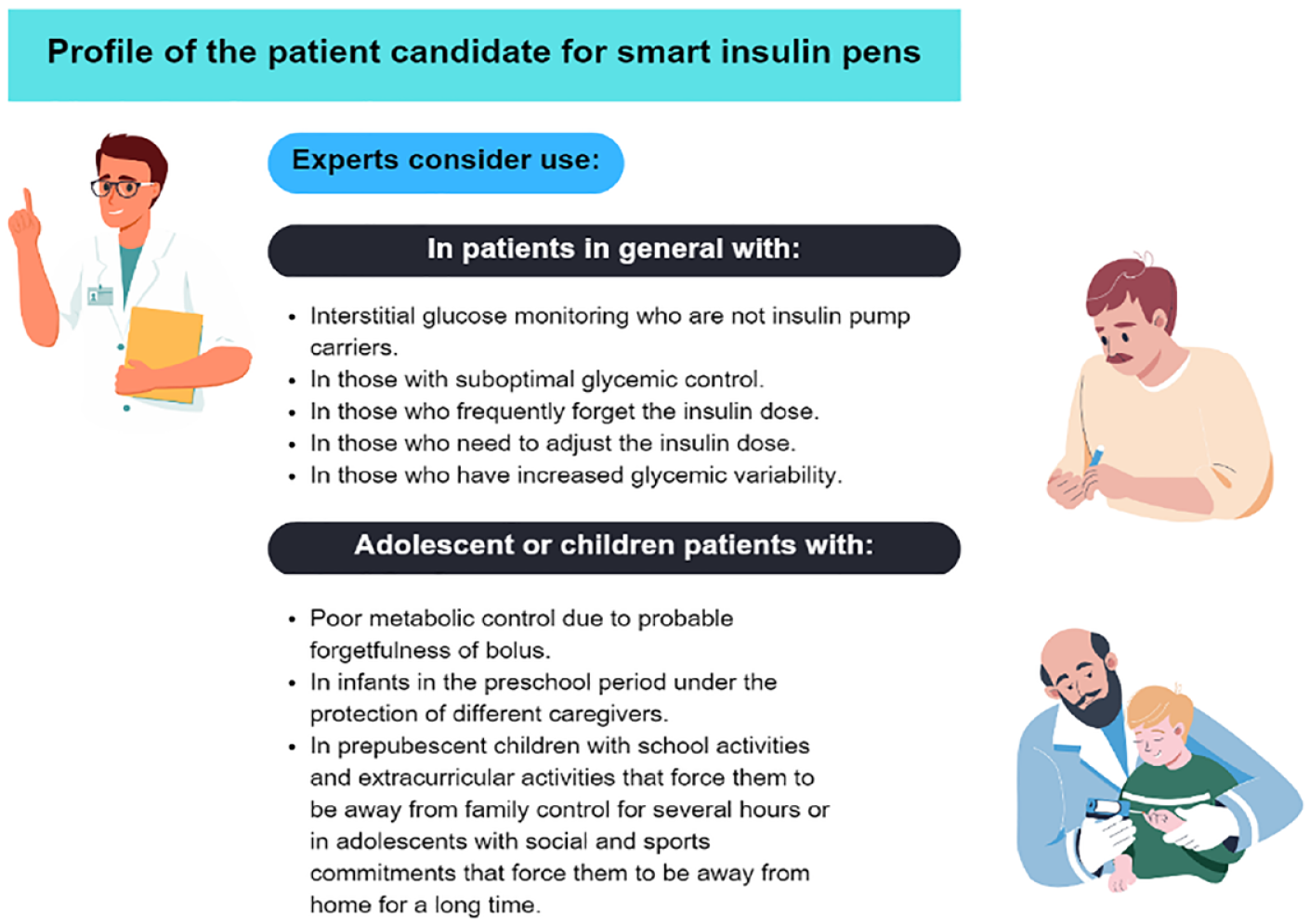
Simple Answer Questions Assessing the Clinical Reality of Control of DM1.
Highlighted in green is the degree of agreement between the experts.
Abbreviation: DM, diabetes mellitus; IRR, incidence rate ratio.
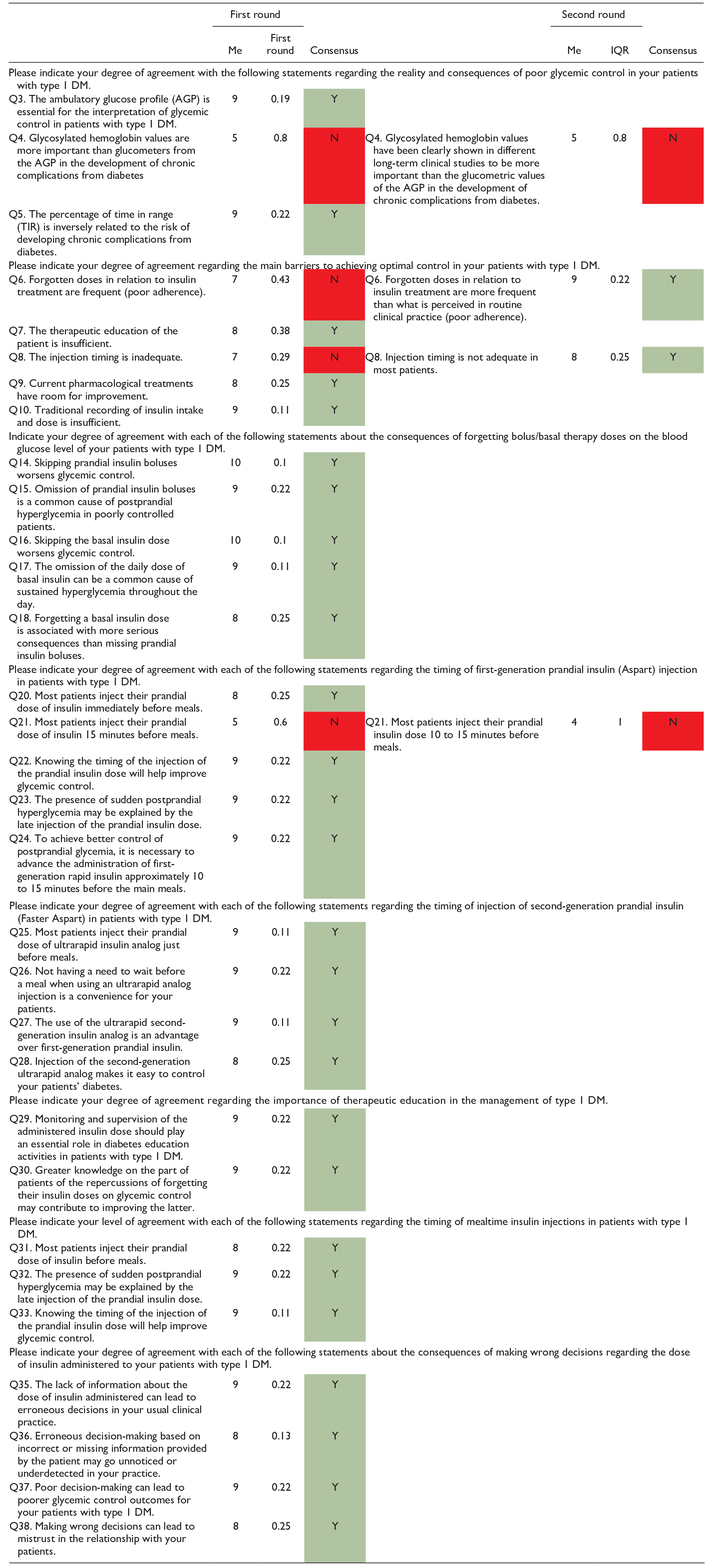
Likert Scale Questions on the Assessment of the Clinical Reality of Control of DM1.
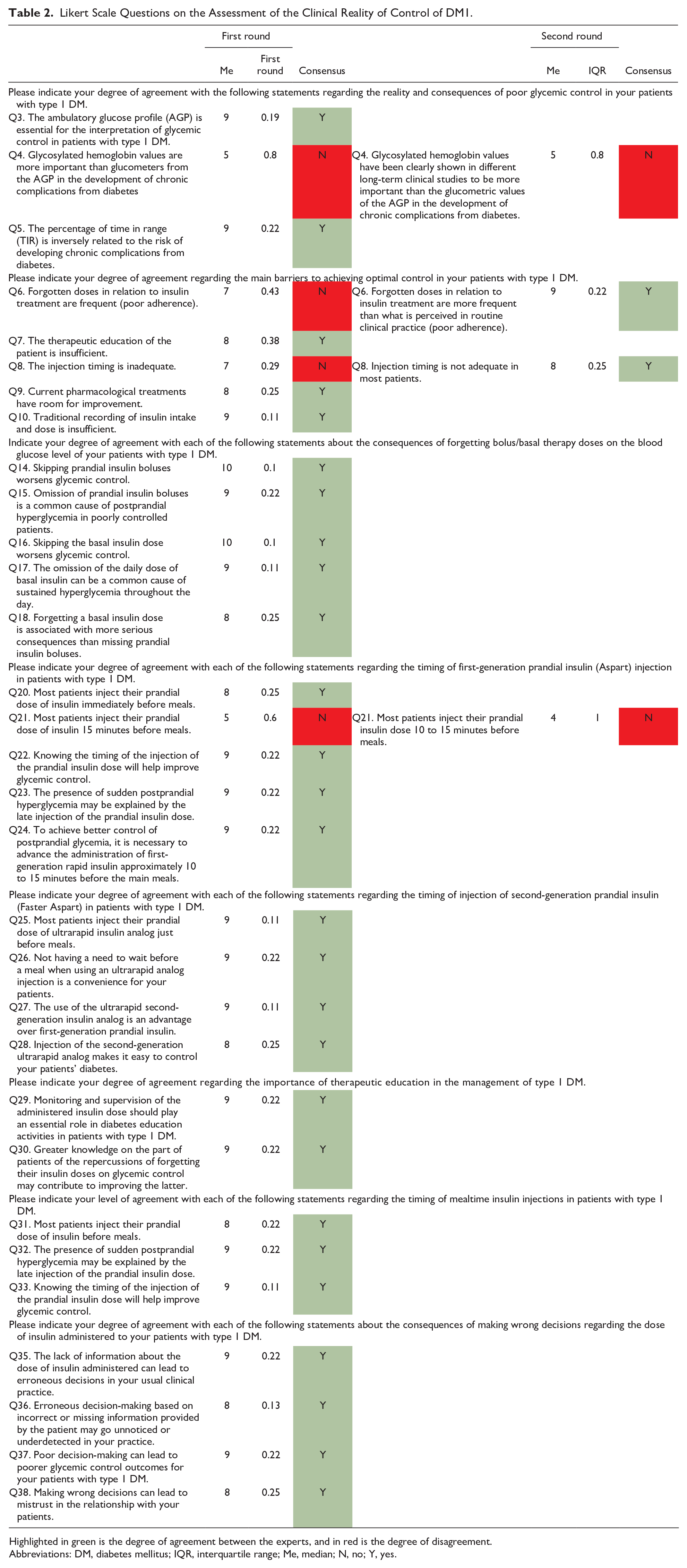
Highlighted in green is the degree of agreement between the experts, and in red is the degree of disagreement.
Abbreviations: DM, diabetes mellitus; IQR, interquartile range; Me, median; N, no; Y, yes.
The experts recommend using the AGP report and the different TIR metrics to assess poor glycemic control. The experts agreed that 31% of their patients have less than 70% TIR values and 65% are diagnosed by their HbA1c values.
Regarding forgetfulness in administering insulin treatment, the experts consider that less than 10% of patients remember to administer their basal dose of insulin. However, forgotten doses are more frequent than perceived in the usual clinical setting. In the case of basal insulin, forgetfulness worsens glycemic control and can be the usual cause of sustained hyperglycemia throughout the day. Regarding the administration of prescribed insulin guidelines, both basal and bolus, the forgetfulness of patients is between 10% and 30%. Skipping prandial insulin boluses worsens glycemic control and is a common cause of postprandial hyperglycemia in poorly controlled patients. In any case, forgetting a dose of basal insulin is associated with more severe consequences than forgetting a dose of prandial insulin.
Regarding the moment of injection of first-generation prandial insulin (aspart, lispro, and glulisine), experts agree that to achieve better postprandial glycemic control, it is necessary to advance the administration by approximately 10 to 15 minutes in the main meals.
However, most patients do not administer their rapid insulin regimen correctly, which could justify the presence of sudden postprandial hyperglycemia. The experts consider injection timing to be inadequate in most patients and that proper timing would improve glycemic control.
Regarding the moment of injection of second-generation prandial insulin (faster aspart), the experts agree that using an ultrafast analog is an advantage over first-generation prandial insulin. Because its use does not require a waiting period before eating, its use facilitates the control of diabetes.
The main barriers to achieving optimal glycemic control are poor adherence to treatment, inadequate injection timing in most patients, the margin for improvement of current pharmacological treatments, insufficient traditional recording of intake and dose of insulin, and insufficient therapeutic education. For this last barrier, the experts consider that therapeutic education would improve glycemic control as they must be aware of the monitoring and supervision of the administered insulin dose and the repercussions of forgetting their insulin doses.
Regarding the information provided by the patients, the experts have identified that when patients do not know whether they have injected the last dose of insulin (basal or prandial), they usually base themselves on the evolution of interstitial glucose to decide the dose of the next injection. However, the experts consider that patients need more information about their medication habits, such as the number of insulin doses administered. Consequently, it can lead the clinician to assume that the patients are carrying out the treatment correctly and make erroneous decisions in clinical practice, worsening the results on glycemic control and generating mistrust in the doctor–patient relationship.
Finally, they did not reach a consensus in considering glycosylated hemoglobin values more important than AGP glucose measurements to assess the development of chronic complications or in assuming that most of their patients injected the prandial insulin dose 10 to 15 minutes before meals.
Advantages of Using Technology to Improve DM Control
The experts reached a consensus of 82.50% agreement in the first round and 94.74% in the second round. The results are shown in Tables 3 to 5. The experts answered the assessments using their experience in managing patients with DM.
Simple Answers to Questions About the Advantages of Using Technology to Improve DM Control.
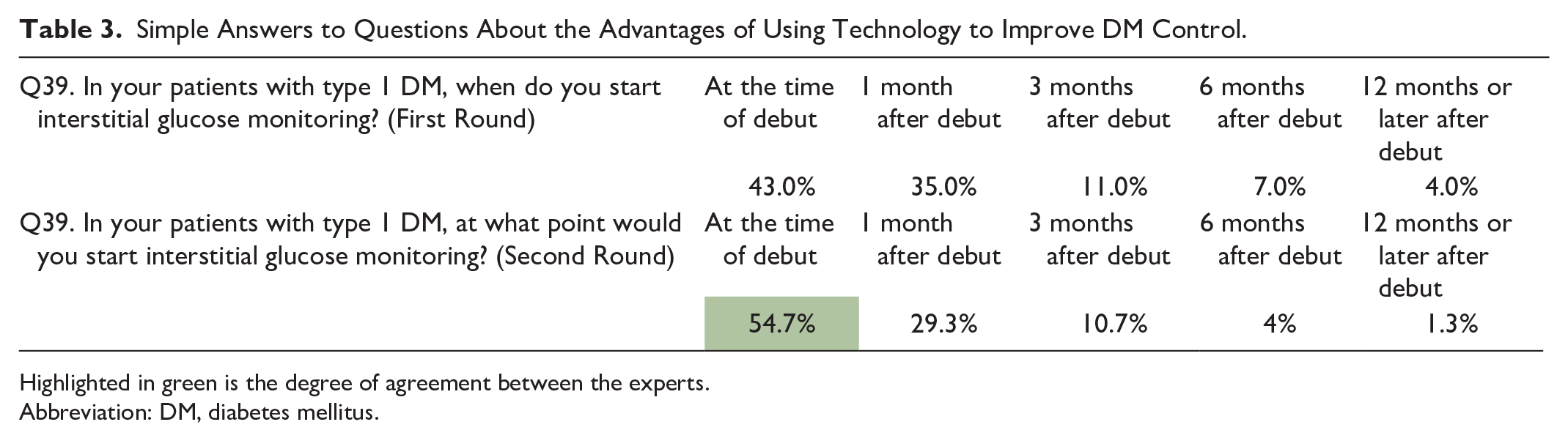
Highlighted in green is the degree of agreement between the experts.
Abbreviation: DM, diabetes mellitus.
Questions in Order From the First Wave About the Advantages of Using Technology to Improve DM Control.
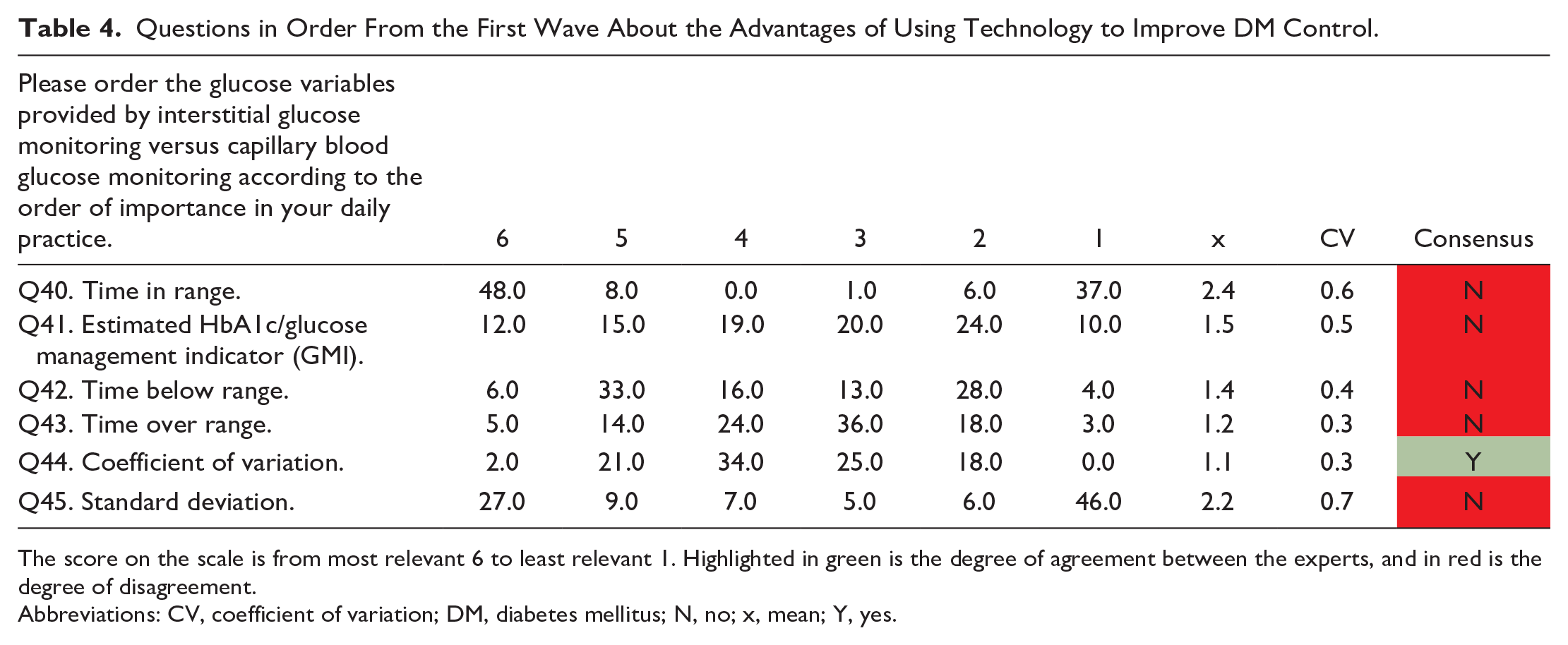
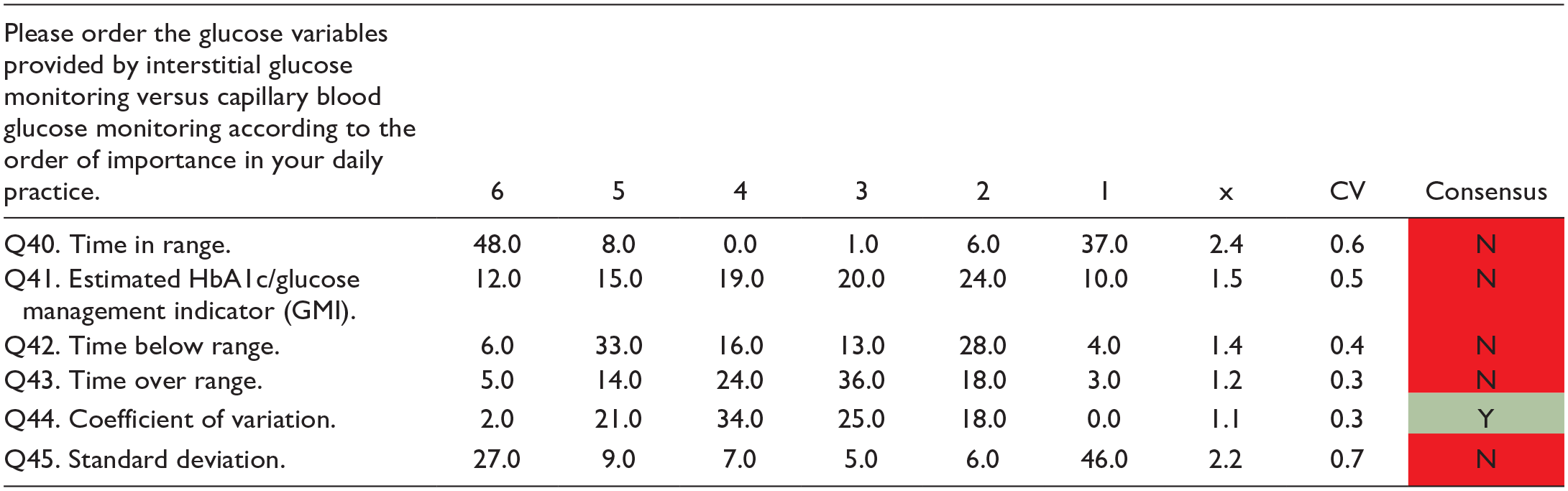
The score on the scale is from most relevant 6 to least relevant 1. Highlighted in green is the degree of agreement between the experts, and in red is the degree of disagreement.
Abbreviations: CV, coefficient of variation; DM, diabetes mellitus; N, no; x, mean; Y, yes.
Likert Scale Questions About the Advantages of Using Technology to Improve DM Control.
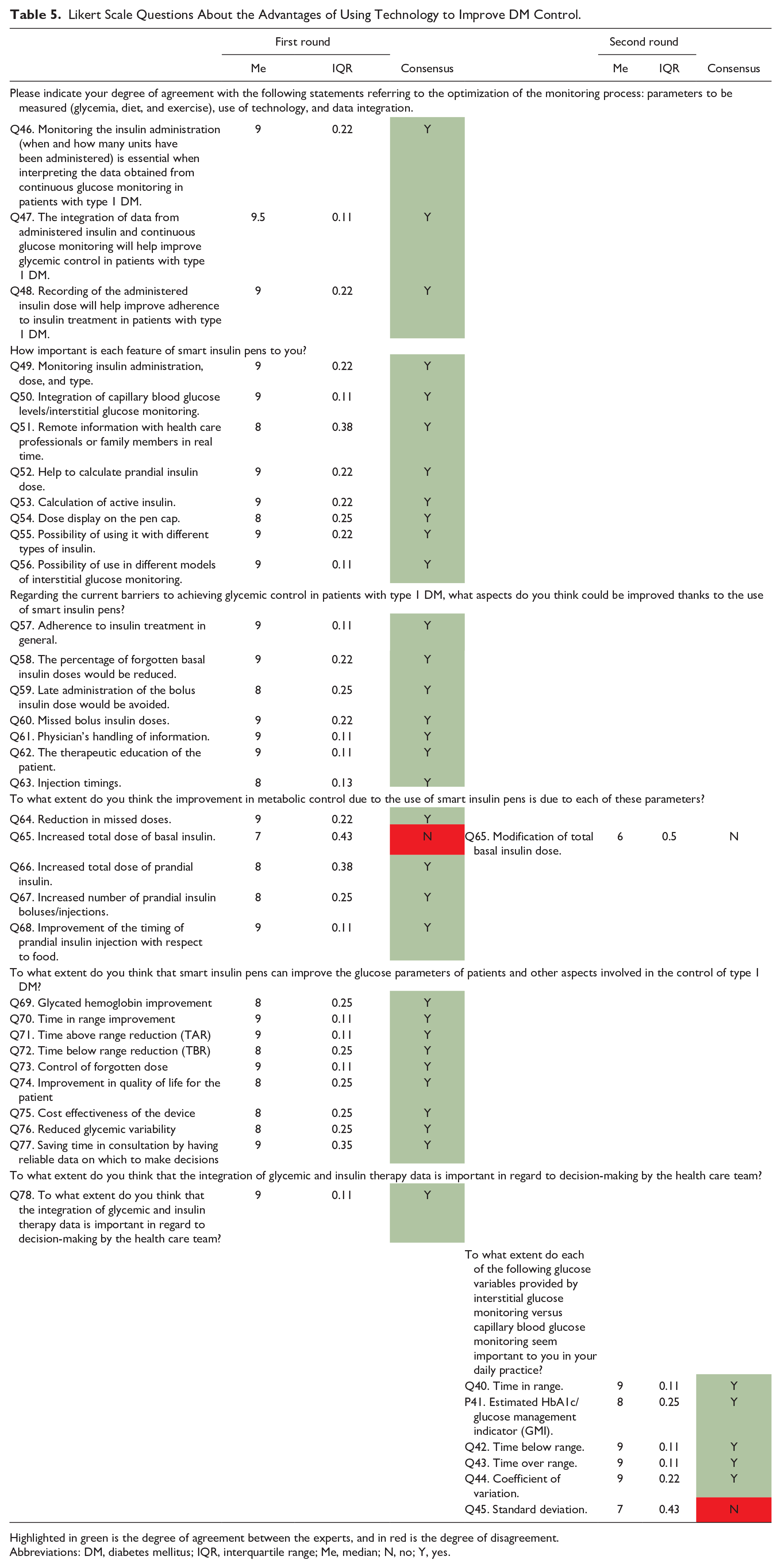
Highlighted in green is the degree of agreement between the experts, and in red is the degree of disagreement.
Abbreviations: DM, diabetes mellitus; IQR, interquartile range; Me, median; N, no; Y, yes.
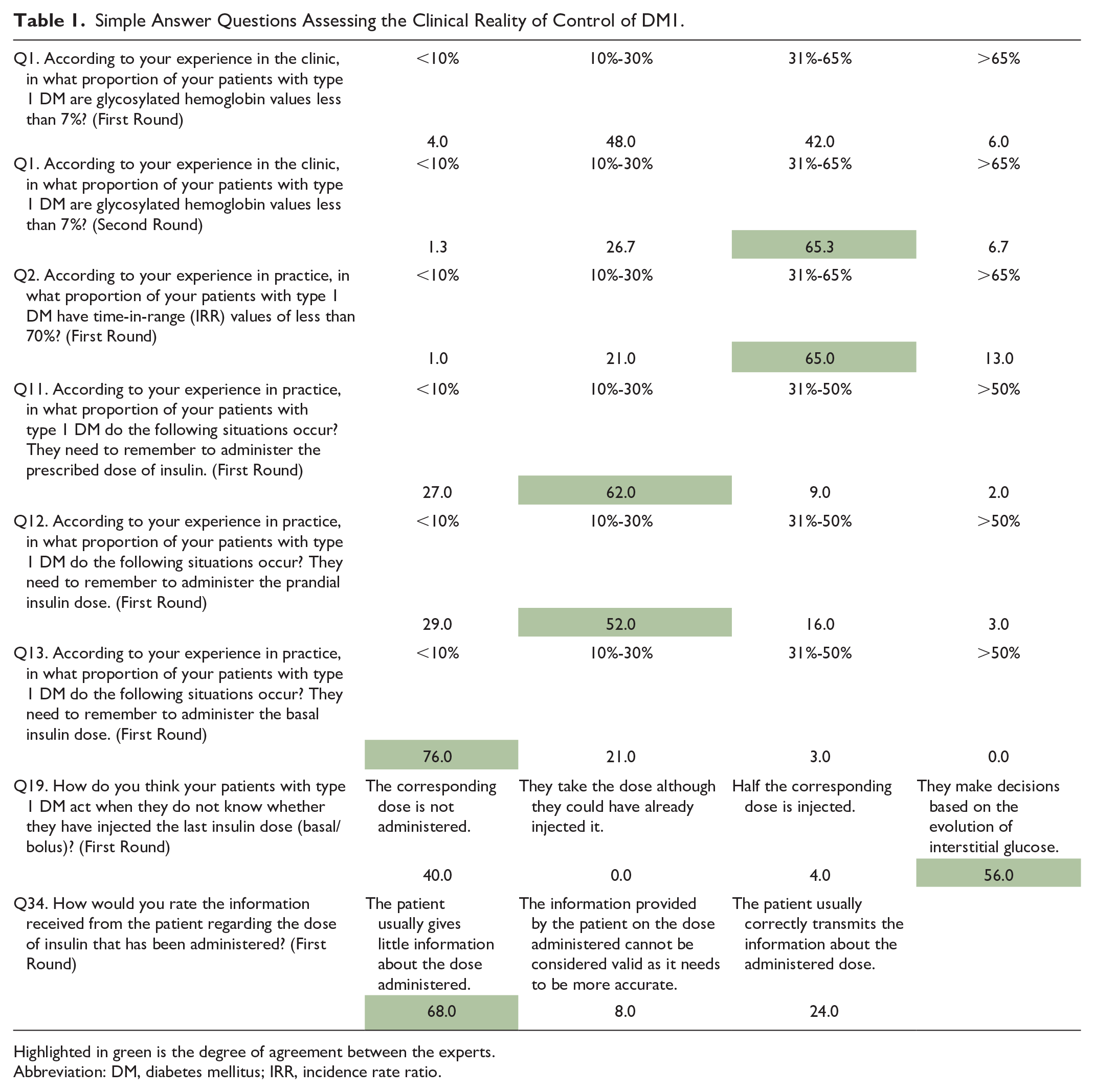
Regarding the development of smart insulin pens, the experts agreed that they should have the following characteristics: good cost-effectiveness; the ability to monitor insulin administration, including the dose and type; the ability to integrate capillary blood glucose levels and monitor interstitial glucose; the ability to remotely inform health care professionals or family members in real time; and the ability to help calculate the dose of prandial insulin and/or active insulin. The experts recommend that smart insulin pens have a dose display and may be used with different insulin types and with different interstitial glucose monitoring models.
The experts consider that using smart insulin pens would clinically favor patients as the percentage of missed basal and prandial insulin doses would be reduced, the injection timing of prandial insulin would improve, and the total prandial insulin dose would increase (although they disagree that their use would increase the total dose of basal insulin). All these benefits would allow for obtaining better glucose values in HbA1c and TIR (reducing the time above and below the range).
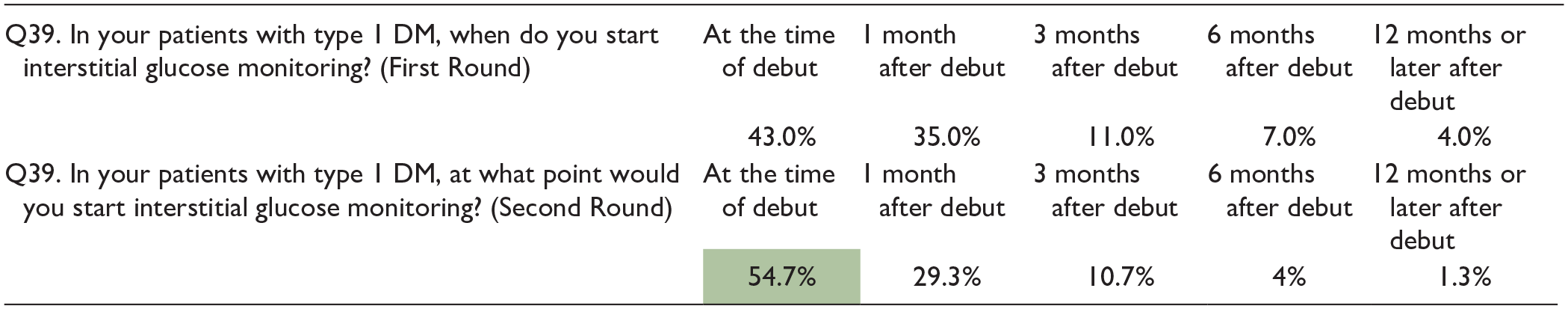
In addition, to assess glycemic control, the experts recommend starting interstitial glucose monitoring at the time of disease onset, considering the following variables to be important for clinical practice (compared with capillary glucose monitoring): TIR (time below or above range), the estimated HbA1c, and the coefficient of variation.
The experts integrated the data obtained from continuous glucose monitoring on the insulin administered (when and how many units) to see whether it would help improve glycemic control and adherence to insulin treatment. Thanks to the synchronization of the data obtained from the insulin pen, there would be greater adherence to treatment, therapeutic education in the patient would be promoted, glycemic variability would be reduced, and more time would be saved in the consultation by having reliable data on which to make decisions.
Finally, the experts disagreed on considering the standard deviation as an important variable for interstitial glucose monitoring versus capillary glucose monitoring in daily practice. The experts also do not consider that modifying the total basal insulin dose improves metabolic control when using smart insulin pens.
Profile of the patient candidate for smart insulin pens
The experts reached a consensus of 100.00% agreement in the first round. The results are shown in Table 6. The experts considered that the use of smart pens might be more beneficial for DM control in patients with interstitial glucose monitoring who are not insulin pump carriers, in those with suboptimal glycemic control, in those who frequently forget the insulin dose, in those who need to adjust the insulin dose, or in those who have increased glycemic variability.
Likert Scale Questions on the Profile of the Patient Candidate for Smart Insulin Pens.
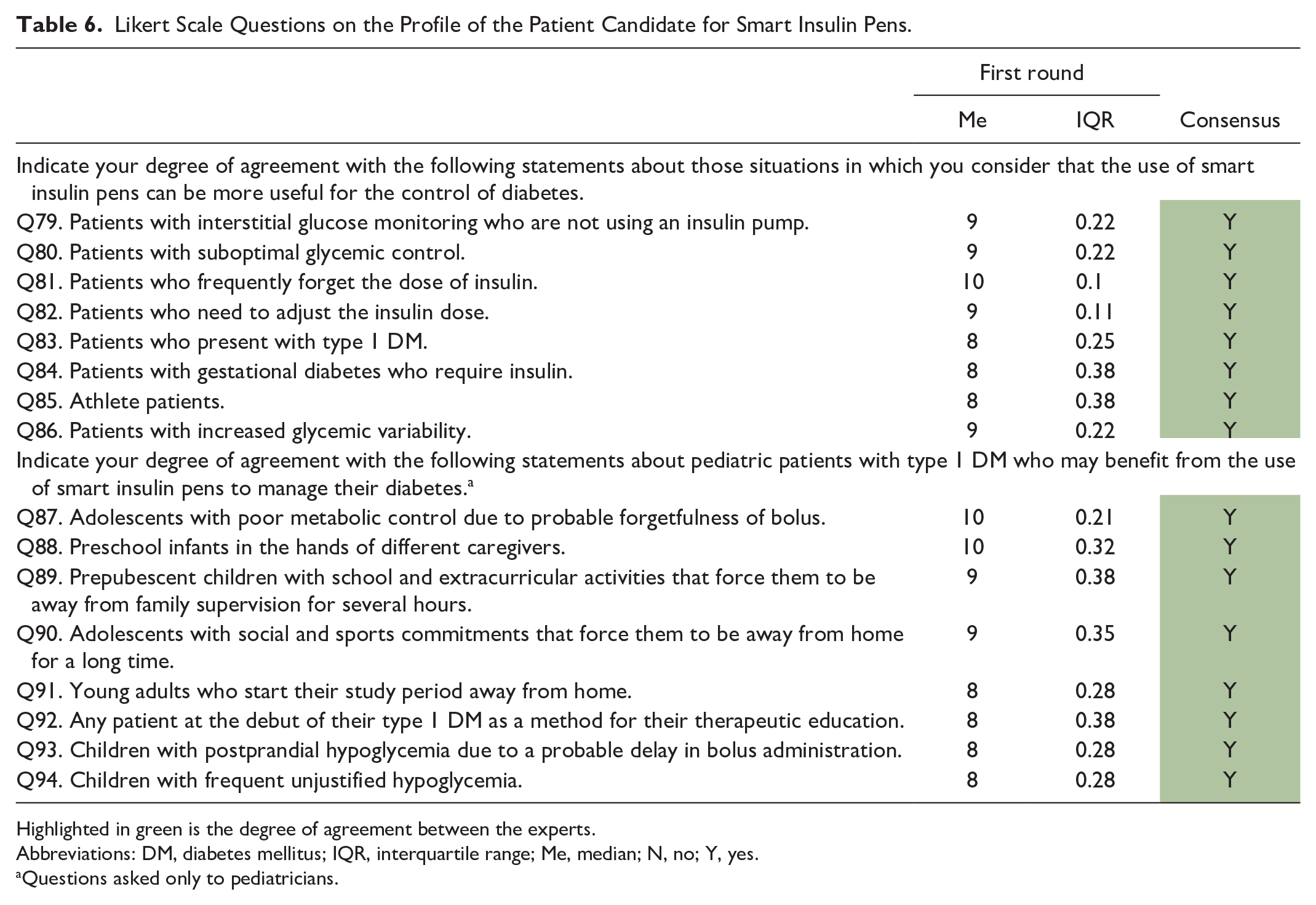
Highlighted in green is the degree of agreement between the experts.
Abbreviations: DM, diabetes mellitus; IQR, interquartile range; Me, median; N, no; Y, yes.
Questions asked only to pediatricians.
Pediatricians recommended the use of smart pens for the control of DM1 up to 18 years of age in adolescents with poor metabolic control due to probable forgetfulness of bolus, in infants in the preschool period under the protection of different caregivers, in prepubescent children with school activities and extracurricular activities that force them to be away from family control for several hours or in adolescents with social and sports commitments that force them to be away from home for a long time.
Discussion
In the scientific literature, there are no consensus studies on the benefits of smart insulin pens; despite this, the degree of agreement reached among the experts was very high.
Among the main barriers that doctors face in achieving reasonable glycemic control in patients with DM1, it is worth highlighting poor adherence to treatment, unreliable recording of insulin intake and dose data, and lack of therapeutic education.
In the systematic review by Davies et al 23 on real-life factors that affect adherence to insulin treatment, barriers perceived by patients were identified: age, female sex, travel, fear of injections, and shame of administering injections in public. The very young age at which DM begins would significantly affect treatment adherence as it is a treatment that will last throughout the patient’s life.
It is vitally important to manage adherence to treatment as it is one of the factors that most influences glycemic control. 24 Adherence to treatment refers to how well a patient follows a medical regime recommended by a health care provider. 25 Approximately, 9.8% of patients had high adherence. However, it has been observed that between 42.2% and 48.0% of patients have moderate and minimal adherence, respectively. 26
The risk of dose omission occurs in 100% of patients. 27 The study by Norlander et al 24 shows that forgetfulness of prandial insulin is between 2% and 38%. Other studies included percentages within this range (11.4% 28 and 24%) 27 and placed the mean days of forgetfulness per month at 5.7 days. 29 Regarding basal insulin, the percentage of forgetfulness is 36% 27 and the average number of days forgotten per month is 4.3 days. 29 The probability of missing at least one basal dose in 14 days is 22%. 27
Forgetting to administer insulin is one of the main reasons for poor adherence. It has been shown that it contributes negatively to glycemic control and increases the concentration of HbA1c levels. 24 The scientific literature found that the frequency of missed prandial insulin injections per week is 2.1, which would be equivalent to an increase in HbA1c (at least 0.3%-0.4% points). 30 The omission of two doses of basal insulin is associated with a change in the TIR greater than 5% and an increase in the time of hyperglycemia, the mean glycemia, and the glucose management indicator (GMI). 31 In this consensus, the experts agreed that the percentage of forgetfulness is much lower, reporting that at least 10% of their patients forget to administer their basal insulin dose. However, they believe that missed doses are more frequent than they perceive in routine clinical practice and that they may need to be more accurate in assessing the clinical reality.
Regarding education and support in glycemic control of DM, other studies support that they improve the results related to the disease. However, data from the United States show that less than 50% of people with DM receive this service. 32 Patient education is essential; we can find therapies such as behavioral obesity treatment (BOT) or cognitive therapy (CT), which focus on lifestyle changes, including diet and exercise, to help people achieve a healthy weight and improve their overall health.33,34
Training allows patients to improve their knowledge and skills regarding their disease and its treatment. 35 In this way, patients with DM would become more aware of the negative consequences of forgetting their dose and timing of injection administration and the importance of providing health professionals with objective and reliable information.
The management of diabetes in clinical practice has many challenges and digital health care solutions are increasingly consolidated to improve disease control. 36 Some of the technologies incorporated have been continuous glucose monitoring throughout the day, using an interstitial system or smart insulin pens. 37
Numerous authors 38 recommend, similar to the experts in this consensus, replacing the classic DM control parameters (HbA1c and hypoglycemia) with new blood glucose measurements derived from the evaluation of interstitial glucose monitoring39,40 to assess glycemic control. The experts considered that the percentage of patients diagnosed with HbA1c is between 31% and 65%. However, the observational study of patients with DM1 by the Spanish Diabetes Society (SED) revealed that the percentage amounts to 70.3% of patients. 38
Regarding treatments used for diabetes management, multiple daily doses of insulin (MDI) are the most common method for intensive insulin treatment of DM1. 41 They are also increasingly common in the treatment of DM2. Although some of the responses in this Delphi questionnaire may be comparable in DM2, the answers were directed at DM1 and differences may exist in other types of diabetes with MDI.
The limitations of short-term and long-term glycemic control have recently been recognized. 39 It has been shown that the evaluation of HbA1c levels and the TIR percentage may be associated with the microvascular complications of DM.42,43
The use of smart insulin pens makes it possible to record the moment and dose of insulin and integrate data from continuous glucose monitoring. 37 If patients do not speak the local language, this would also be an advantage as the information recorded by the smart insulin pens is language-independent. The experts consider that using smart insulin pens would improve current glycemic control barriers, adherence to treatment, management of information in the doctor–patient relationship, and therapeutic education of the patient. The results of our study coincide with other investigations that show the improvement of glycemic control, 44 its profitability, 16 and the increase in adherence.45,46 These technologies would be beneficial in-home care and nursing as they would improve medication adherence and allow for more optimal monitoring of patient medication administration.
The literature review by Heinemann et al 11 on smart insulin pens shows a need for more high-quality studies in the scientific literature. This consensus study is the only one published in Spain, whose results show the benefits perceived by physicians for the use of smart insulin pens. Well-designed clinical trials and real-life studies are necessary.
Conclusions
In conclusion, the perception of physicians in their usual practice leads them to recommend the use of AGP and TIR for glycemic control. Forgetting to administer insulin, both prandial and basal, is very common, according to what has been reported in different studies, and does not correspond to clinicians’ perceptions. Technological improvements increase treatment adherence and, in this sense, the incorporation of smart insulin pens would help improve glycemic control due to excellent connectivity. In addition, they strengthen the doctor–patient relationship and educate and improve patients’ quality of life.
Footnotes
Acknowledgements
The authors thank Rosa Rojo (medical writer, ScarletMed Scientific Consulting, Madrid, Spain) for drafting the manuscript and Liquid Care SL for the support and management.
Abbreviations
ADA, American Diabetes Association; AGP, ambulatory glucose profile; BOT, behavioral obesity treatment; CT, cognitive therapy; DM, diabetes mellitus; DM1, type 1 diabetes; DM2, type 2 diabetes; FDA, Food and Drug Administration; GMI, glucose management indicator; HbA1c, glycated hemoglobin; IQR, interquartile range; IRR, incidence rate ratio; MDI, multiple daily doses of insulin; SED, Spanish Diabetes Society; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jesús Moreno Fernández reports receiving fees as a consultant and speaker from Novo Nordisk. Juan Girbes reports having received fees for presentations, registration for congresses, and registration for Novo Nordisk courses. Gonzalo Díaz and Francisco Javier Arroyo declares that there are no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Novo Nordisk.