Abstract

There is an ongoing need for manufacturers of blood glucose monitoring (BGM) systems to ensure that the accuracy of their in-market products continues to exhibit performance consistent with that submitted to the regulator at the time of product clearance. High-profile post-market surveillance (PMS) studies focusing on the clinical accuracy of market-leading BGM products in the United States and Europe have indicated that some products may fall consistently short of claimed performance, raising concerns for patient safety.1,2 Consequently, regulators such as the Food and Drug Administration (FDA) and their European counterparts are demanding that manufacturers play a more proactive role in implementing appropriate PMS programs. While PMS may take many forms, here we provide an update to our previously reported PMS data regarding the clinical performance of the OneTouch Verio test strip platform, manufactured by LifeScan Inc (Malvern, PA, USA). 3
The methodology has been previously reported. 3 Briefly, the clinical accuracy of randomly sampled test strip batches was assessed using fingertip capillary blood samples drawn from a target of 100 subjects with diabetes and tested by a clinician, as per instructions for use. Results were compared with YSI STAT PLUS. Calculation of percent bias and test strip lot mean absolute relative difference (MARD), expressed as absolute bias and mean absolute difference (MAD) at low glucose (<100 mg/dL), is as previously detailed. 3 Definitions of clinical accuracy were as per FDA guidance and International Organization for Standardization (ISO) 15197:2013, which stipulate that ≥95% of results should have biases within ±15% (or ±15 mg/dL at glucose <100 mg/dL in the case of ISO 15197:2013) of comparator.4,5 Surveillance error grid (SEG) and consensus error grid (CEG; type 1 diabetes) analyses were performed.
A total of 14 283 paired meter-comparator readings were recorded across the latest surveillance reporting period (2019-2021), giving a total of 88 507 paired readings since product launch (Table 1). A lower number of tests were performed in 2020/2021 due to clinic access issues caused by the COVID-19 pandemic. Clinical accuracy, as defined by FDA guidance and ISO, remained consistent over the latest reporting period compared with full product history (2009-2021), with 97.7% versus 97.4% of readings within ±15% of comparator, respectively, while 97.8% to 97.9% of readings were within ±15%/15 mg/dL of comparator, irrespective of the reporting period. Similarly, no significant shift in performance was seen regarding distribution of data pairs within the CEG risk zones between the reporting periods. To date, a total of 88 289 values met SEG analysis criteria, 6 of which 86 867 (98.39%) were within SEG risk category “None,” 1369 (1.55%) and 49 (0.06%) within the “Slight, Lower” and “Slight, Higher” categories, respectively, three values within the “Moderate Lower” category, and one value within the “Moderate, Higher” category. Overall, product MARD (calculated as MAD at <100 mg/dL glucose) was 4.90%.
BGM Device Clinical Accuracy Across Surveillance Reporting Periods: 2009 (Product Launch)-2018, Latest Reporting Period 2019-2021, and Overall (Product Launch-2021).
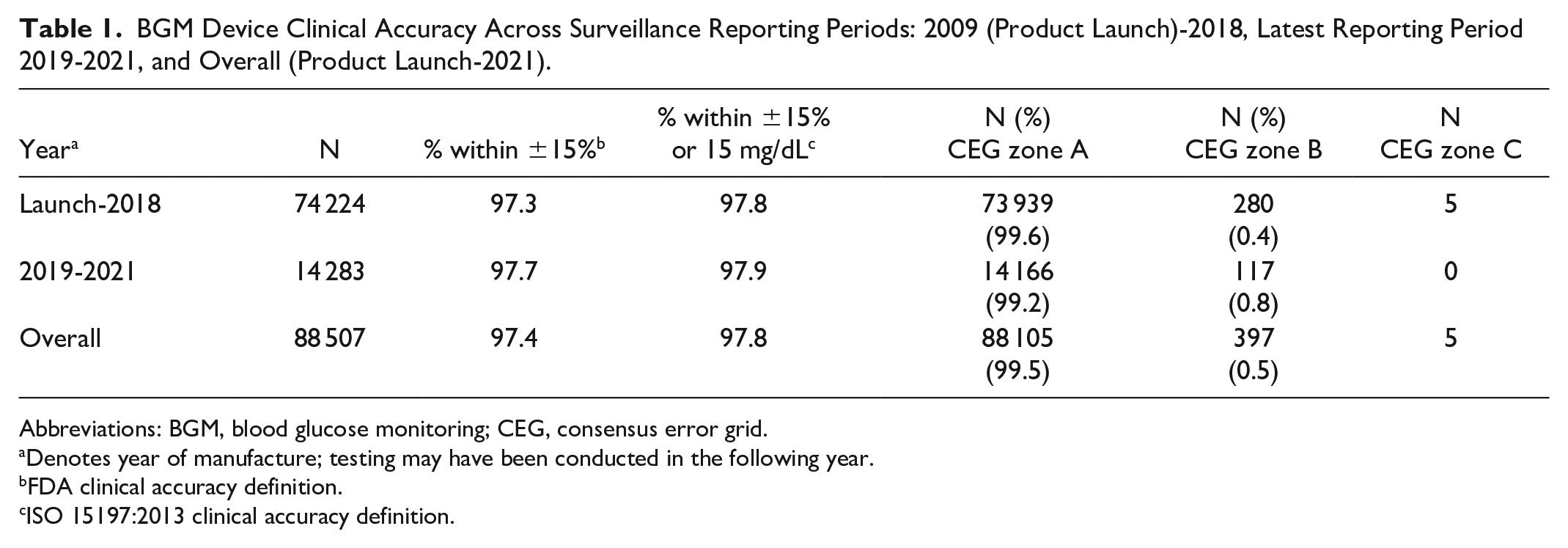
Abbreviations: BGM, blood glucose monitoring; CEG, consensus error grid.
Denotes year of manufacture; testing may have been conducted in the following year.
FDA clinical accuracy definition.
ISO 15197:2013 clinical accuracy definition.
In conclusion, based on several widely recognized parameters of clinical accuracy, the OneTouch Verio test strip continues to demonstrate a high level of performance consistent with data supplied to the regulator at the time of product registration. The manufacturer will continue to assess the post-market clinical accuracy of its test strip platform products as one of its initiatives to maintain patient safety.
Footnotes
Abbreviations
BGM, blood glucose monitoring; CEG, consensus error grid; FDA, Food and Drug Administration; ISO, International Organization for Standardization; MAD, mean absolute difference; MARD, mean absolute relative difference; PMS, post-market surveillance; SEG, surveillance error grid.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SS, SP, HC, and MG are all employees of LifeScan Scotland Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by LifeScan Scotland Ltd.
