Abstract
With the prevalence of diabetes higher than ever, governments and people with diabetes are facing significant treatment and indirect costs associated with managing their condition. An ultra-low-cost insulin pump is a possible solution to improving health disparities. This article presents test results for an insulin-pump built from low-cost components (bill of materials < $US100). All testing was completed in accordance with IEC60601-2-24, and results were benchmarked against a commercial pump. Results showed the ultra-low-cost pump has comparable accuracy to the commercially available insulin pump with testing displaying an overall accuracy of 0.089% and −0.392%, respectively. These results show that an ultra-low-cost pump can accurately deliver insulin in limited bench testing. Testing in other environments and scenarios is required to fully meet IEC60601-2-24 standards.
Introduction
Insulin-pump therapy, or continuous subcutaneous insulin infusion is considered the best available treatment method for insulin-dependent diabetes. 1 Continuous subcutaneous insulin infusion is associated with improved clinical outcomes in both type 1 diabetes (T1D)2 -4 and insulin-dependent type 2 diabetes (T2D).5,6
High initial purchase costs and ongoing costs for consumables ($NZ8800-$NZ10,000, $NZ2340, respectively) 7 mean this technology is underutilized. Those without health insurance may not be able to afford an insulin pump. In countries with subsidized healthcare, high costs can lead to a rationing of care and can push costs onto the patient. 8
In New Zealand, the greatest diabetes prevalence is seen in Māori, Pacific, and Indo-Asian ethnic groups. 9 With Pacific and Māori peoples reported to have the lowest median weekly income, diabetes disproportionally affects those least able to afford care.8,10 -12 This trend is also seen worldwide, with rapidly increasing rates of diabetes occurring in developing countries.13 -16
If the cost of insulin pumps were significantly lower, their use could be extended to patients who could not previously afford them, resulting in potential future savings from reduced complications.
For an ultra-low-cost insulin pump to be viable, it must offer acceptable accuracy for clinical use. The International Electrotechnical Commission (IEC) testing standard does not specify minimum accuracy, specifying only how data should be collected. 17 Therefore, acceptable accuracy is by comparison to commercially available models and accuracy parameters provided by manufacturers and the U.S. Food and Drugs Administration (FDA) regulatory documents. For the commercial pump used in this study, the manufacturer states an accuracy of ±20% for boluses less than 0.1 U and ±5% for all other boluses and basal rates, 18 while the Summary of Safety and Effectiveness Datasheet states all hourly intervals should be within ±5%, with a cumulative error no greater than ±10%. 19
This article uses standards-based testing to document the performance of an ultra-low-cost insulin-pump prototype against a commercially available device, using published data 20 as an independent comparison to validate the overall test setup and results.
Methods
An ultra-low-cost, open-source 21 insulin pump was designed using inexpensive components. The pump mirrors designs of commercially available devices, using a stepper motor and plunger to compress an insulin cartridge, and a battery to store energy. The ultra-low-cost pump allows basic user input and feedback and could also be controlled from a Bluetooth-capable device. The pump uses standard consumables (cartridge and infusion set), and the 3D printed case design is modifiable to accommodate cartridges from different manufacturers. The total bill of materials is less than $US100 at this writing.
A commercially available insulin pump (In-warranty Medtronic 640G, Medtronic, Northridge, CA, USA) was used to benchmark against. Figure 1 shows the ultra-low-cost pump and an exploded view showing individual components. Table 1 lists the main pump components.
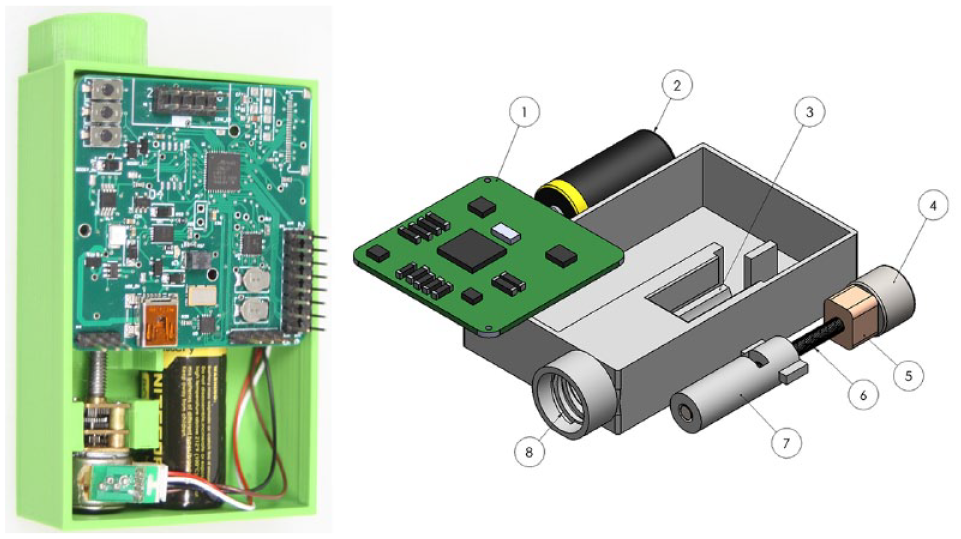
Ultra-low-cost insulin-pump prototype photograph (left) and exploded view Solidworks render (right), where numbered components are defined in Table 1.
Components Shown in Figure 1 (Right) Include.

The pump was tested using methodology given in IEC60601-2-24 (IEC60601-2-24), Sections 50.104 (basal testing), and 50.106 (bolus testing). 17 The methodology in the unit standard is used when testing commercial insulin-pumps so was chosen to test the ultra-low-cost insulin pump. A diagram showing the testing setup is given in Figure 2.
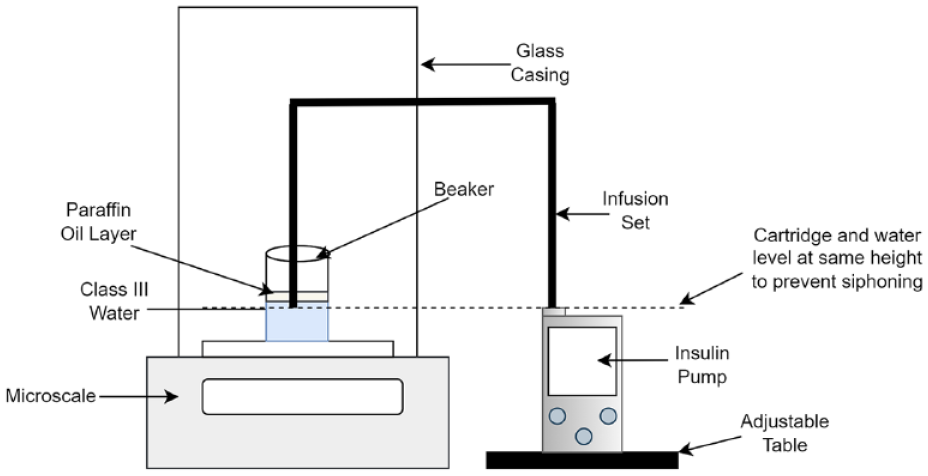
Setup for insulin-pump testing, as per IEC60601-2-24. 17
A microscale (Mettler Toledo XP105) giving 5dp of accuracy was used. The use of a full standard infusion-set and cannula without site hub removed ensures results are comparable to use in-vivo. The class III water was degassed before use, ensuring consistency between tests. A layer of paraffin oil of <3 mm thickness was floated on the water to eliminate evaporation and is shown to produce more accurate results.20,22,23 IEC60601-2-24 requires the pump to be run at a “typical rate.” Therefore, 1 U/h was chosen as a suitable basal rate to evaluate accuracy. In addition, it has been used in other studies.20,24
To ensure repeatable data collection, a settling of time of 5 minutes was used, and the scale was reset between boluses. A 180 U cartridge was used, and 5 U was selected as the maximum bolus size to ensure a cartridge change would not be required midway through testing. The 5U bolus results also exist in other studies. 25 Box and whisker plots are used to present bolus data, for easy, nonparametric interpretation without assuming a distribution shape of the results.
Results
Results for basal testing are presented as trumpet curves in Figures 3 and 4. The commercial pump displays an overall accuracy of −0.392%, and a maximum variability of 44%. The ultra-low-cost insulin pump shows an overall accuracy of 0.089% and a variability of 24%.
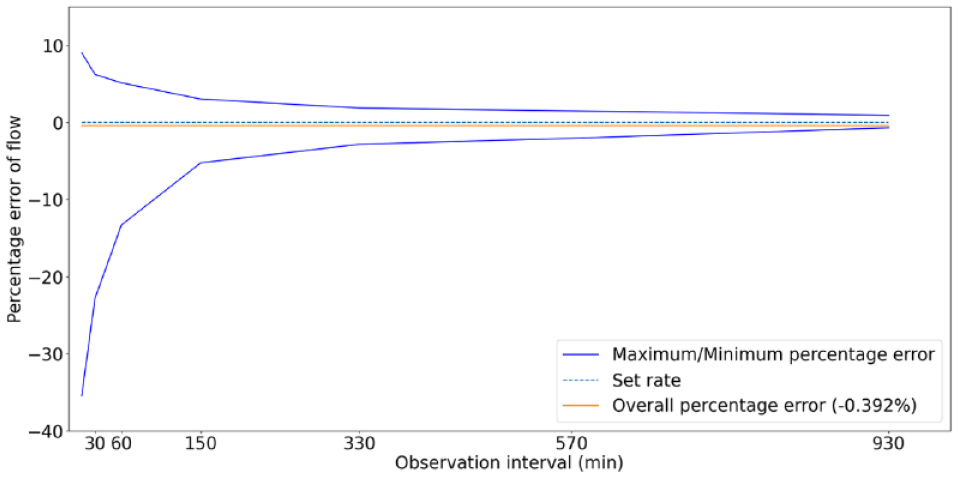
Trumpet curve presented as per IEC60601-2-24 for the commercial pump.
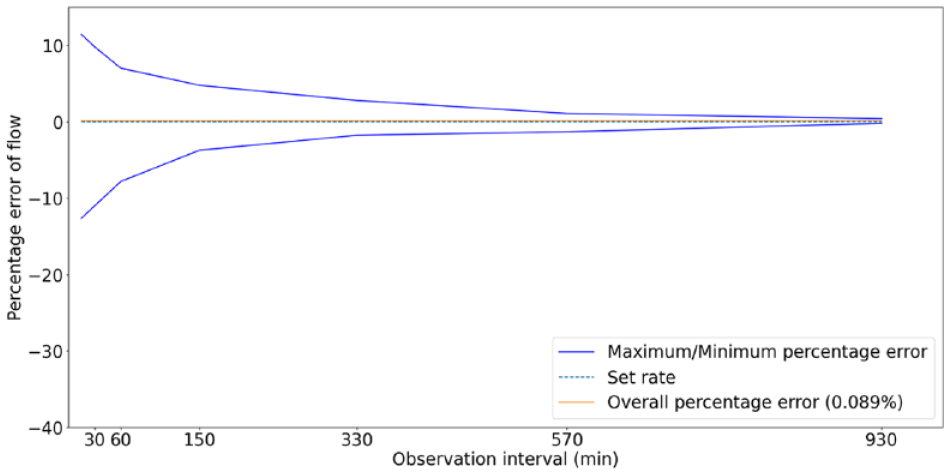
Trumpet curve presented as per IEC60601-2-24 for the Ultra-low-cost insulin-pump.
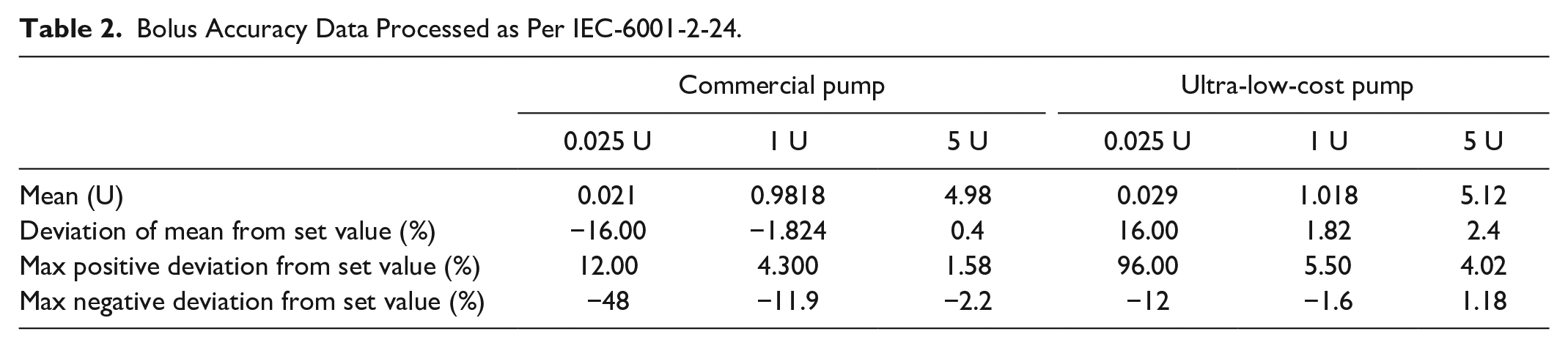
Box and whisker plots for all bolus sizes are given in Figures 5 to 7, and data processed per IEC60601-2-24 is presented in Table 2. Outliers have been classified as any data points laying outside the 5th to 95th percent range. On any plot, if less than 4 outliers appear, datum lie on top of each other. A dashed line is given at 0% error.
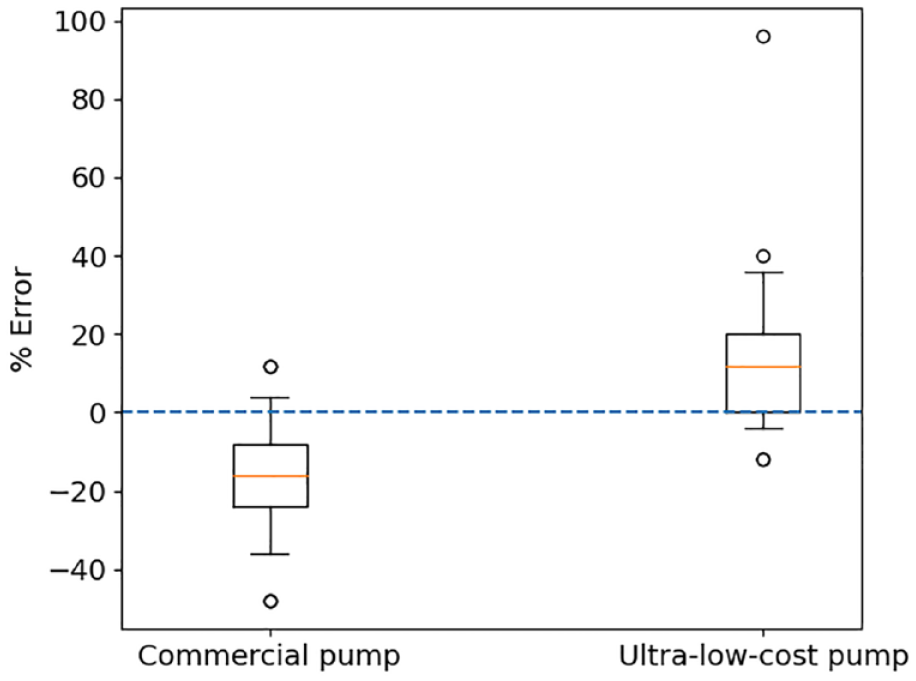
Percentage deviation from a requested dose of 0.025 U for the commercial pump and ultra-low-cost insulin pump over 25 individual boluses.
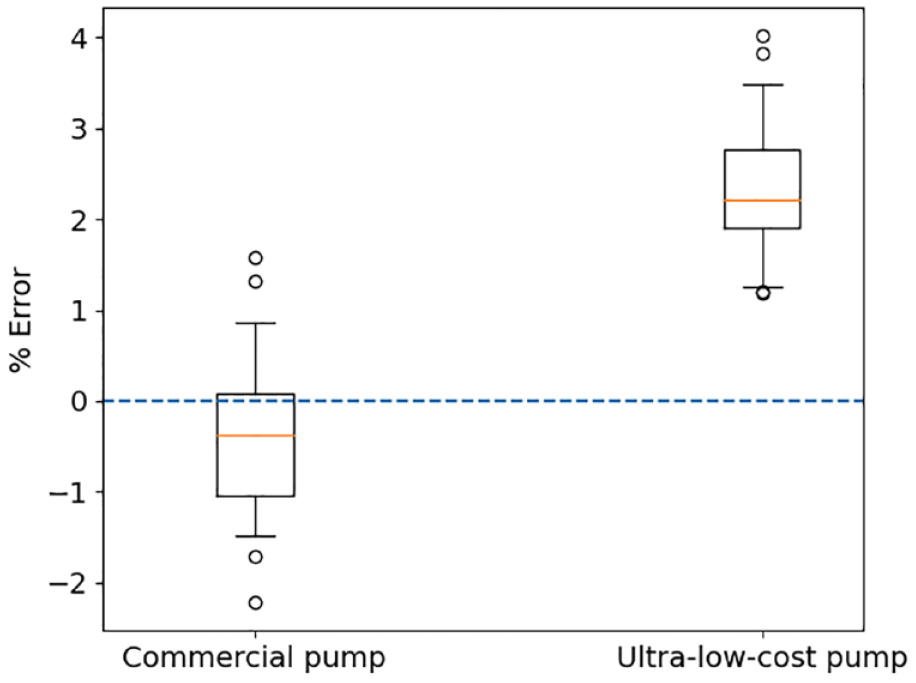
Percentage deviation from a requested dose of 1 U for the commercial pump and ultra-low-cost insulin pump over 25 individual boluses.
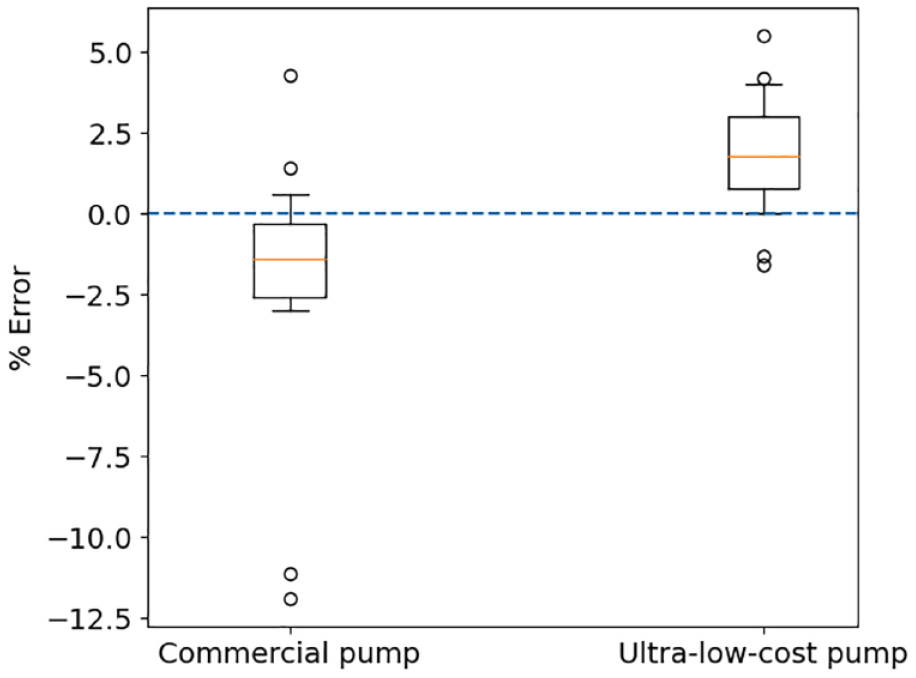
Percentage deviation from a requested dose of 5 U for the commercial pump and ultra-low-cost insulin pump over 25 individual boluses.
Bolus Accuracy Data Processed as Per IEC-6001-2-24.
Discussion
The ultra-low-cost insulin pump demonstrated a high level of accuracy in insulin delivery, with an error of <1% over the 930-minute testing schedule. Results were similar to those observed with a commercial pump, except the test pump showed less variability. Results obtained for the commercial pump are validated by using the same methodology and providing similar overall results to those in previous literature, 20 reinforcing the testing methodology used was robust. Specific results may differ due to differences in laboratory conditions between the two reports.
It was observed that the delivery of the very small 0.025 U boluses had the most variability, in both the test and commercial insulin pump. While there are no available data on the accuracy of commercial pumps at this resolution, Ziegler et al 24 showed an over-delivery of 9.1% on average, with only 70.7% of doses delivered within ±15% with the same commercial pump at 0.1 U increments. Percentage errors are known to increase with smaller dose sizes23 -25, and therefore, these observations are not unexpected.
The low-cost insulin pump has shown a tendency to over deliver at all bolus sizes (Figures 5-7). Any clinical impact of measured dose inaccuracy is currently unknown. However, an in-silico study examining the insulin required to change blood glucose in children found an increased dose of 0.1 U affected blood glucose by 12.6 mg/dl in only 5% of the population, 26 suggesting this dose inaccuracy may not be significant in much of the population, but for smaller children there may be added risk.
In terms of pump cost, and equity of access to care, the effect of a low-cost insulin pump varies depending on where it is used. In countries such as New Zealand, insulin costs are low, with a vial costing between NZ$25 and NZ$45. 7 Therefore, the insulin pump represents a more significant proportion of the total cost of diabetes care. In countries where insulin is more expensive, the pump may represent a smaller percentage of total cost to those with diabetes, and thus have less effect on equity of access to care.
The main limitation of this investigation is only a single ultra-low-cost pump and commercial pump were tested. There is likely to be variability between pumps from any production line, and quality control would require interval sampling. Also, insulin-pumps usually have an in-warranty lifespan of 4 years, and hence testing over such a timeframe would also be important to highlight accuracy changes with extended use. Whilst the commercial pump used in this study was not brand new, the device was within warranty and would be expected to perform within the manufacturer’s stated accuracy parameters. Full comparison to manufacturer provided accuracy results is not possible because the given trumpet curve is not presented as required by the unit standard and no description of test methodology is given. 18 Therefore, the authors refer to the commercial pump user guide as a reference to manufacturer-stated accuracy only.
The microscale used for testing had a readability of 0.01 mg, and a repeatability of 0.007 mg, meeting the IEC standard requirements. The maximum possible error when is therefore ±5.6% for a 0.025 U dose. 27 Using a microscale with a better repeatability and readability would allow more accurate results to be obtained for insulin dosing in this range, potentially with less variation. The accuracy of the microscale would be unlikely to affect results significantly for 1 U and 5 U boluses, with a maximum error of 0.14% and 0.028%, respectively.
Finally, the accuracy of the ultra-low-cost pump was calibrated using the same scale used for testing, which could explain why the overall accuracy of this pump is closer to the set value. The design of the ultra-low-cost pump is also more limited in terms of features than the commercial pump, with a feedback control system and other safety systems not yet implemented. It is possible that this extra hardware may affect pump accuracy.
Conclusions
The ultra-low-cost insulin-pump design demonstrated positive accuracy results. Further research will test the pump in a clinical setting to ascertain if laboratory-based results can be extended to use in-vivo. Pump hardware will also be modified to meet all required aspects of IEC60601-2-24 and include Bluetooth capability, to allow the pump to be controlled via a mobile device.
Footnotes
Abbreviations
CSII, continuous subcutaneous insulin infusion; IEC60601-2-24, International Electrotechnical Commission 60601-2-24; T1D, type 1 diabetes; T2D, type 2 diabetes; TDD, total daily dose.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NZ National Science Challenge 7, Science for Technology and Innovation (2019-S3-CRS), and the University of Canterbury Doctoral Scholarship program.
