Abstract
This article is the second of a two-part series providing a scoping review and summary of the Joint British Diabetes Societies for Inpatient Care (JBDS-IP) guidelines on the use of diabetes technology in people with diabetes admitted to hospital. The first part reviewed the use of continuous glucose monitoring (CGM) in hospital. In this article, we focus on the use of continuous subcutaneous insulin infusion (CSII; insulin pumps) and hybrid closed-loop systems in hospital. JBDS-IP advocates enabling people who can self-manage and are willing and capable of using CSII to continue doing so as they would do out of hospital. CSII should be discontinued if the individual is critically ill or hemodynamically unstable. For individuals on hybrid closed-loop systems, the system should be discontinued from auto-mode, and may be used individually (as CGM only or CSII only, if criteria are met). Continuing in closed-loop mode may only be done so under specialist guidance from the Diabetes Team, where the diabetes teams are comfortable and knowledgeable about the specific devices used. Health care organizations need to have clear local policies and guidance to support individuals using these wearable technologies, and ensure the relevant workforce is capable and skilled enough to ensure their safe use within the hospital setting.
Keywords
Introduction
Effects of concurrent illness, medication changes, altered meal timings, and intake in hospital all contribute to sub-optimal glycemia in hospital. Hyperglycemia and hypoglycemia in hospital are associated with increased risk of infection, length of stay, admission to the intensive care unit, and mortality.1,2
Insulin pumps, also called continuous subcutaneous insulin infusion (CSII) systems are small portable devices that deliver a continuous infusion of rapid-acting insulin subcutaneously. They have the advantage of delivering pre-programmed variable rates throughout the day to mimic physiological “basal” insulin delivery. There is also a “bolus” feature that is used to give additional doses of insulin based on preset insulin-to-carbohydrate ratios and insulin sensitivity factors. These boluses are usually initiated by the individual to provide insulin to cover food intake at meal-times and correct hyperglycemia.
CSII in the outpatient setting has been shown to reduce hemoglobin A1c (HbA1c), particularly in those with baseline sub-optimal glycemia, and reduce hypoglycemia, especially in those with frequent, severe hypoglycemia. 3 Best outcomes in CSII use are particularly observed in those with continued elevated HbA1c and/or disabling hypoglycemia on multiple daily insulin injections motivated to use insulin pumps. 4 Uptake of insulin pumps is between 20% and 50% of people with type 1 diabetes across Western Europe and the United States. It is therefore reasonable to assume that some individuals will wish to continue using CSII on admission to hospital.
This article provides an overview of the upcoming Joint British Diabetes Societies for Inpatient Care (JBDS-IP) guidelines on the use of CSII in people with diabetes admitted to hospital in the United Kingdom.
In-Hospital Use of Insulin Pumps
Safe continuation of use of CSII in hospital in those who can self-manage has been advocated by several professional societies, including JBDS-IP, 5 the Diabetes Technology Network, 6 American Diabetes Association, 7 American Association of Clinical Endocrinologists, 8 and the Endocrine Society. 9
While there are limited randomized studies, observational studies in adults with type 1 diabetes (T1D) showed that there was no difference in mean glucose between those continuing CSII and those not,10,11 with one study indicating significantly fewer episodes of severe hyperglycemia (> 16.7 mmol/L; > 300 mg/dL) and hypoglycemia (< 2.2 mmol/L; < 40 mg/dL) in pump users. 11 Besides reducing the risk of insulin administration errors, people often request to continue CSII use, providing autonomy to self-manage glycemia. 12 People also report high satisfaction (86%) when allowed to do so. 12
On acute admission to hospital, a considered decision should be made on an individual basis as to whether the person can continue to safely use their pump (Figure 1). The key criteria for continuation of CSII include: (1) The individual is medically stable, willing, and capable of self-management, and (2) the treating clinician’s familiarity with the CSII, appropriate hospital policies/guidance on CSII use and inpatient diabetes management teams to support. Shortly after admission, inpatient diabetes teams and/or the endocrinology service should be involved in helping with insulin adjustment and pump settings as well as in coordinating care after hospital discharge. The lack of a 24/7 diabetes team and infrastructure to support in majority of the UK hospitals (manuscript submitted for publication) remains a challenge to effective implementation within usual inpatient care.
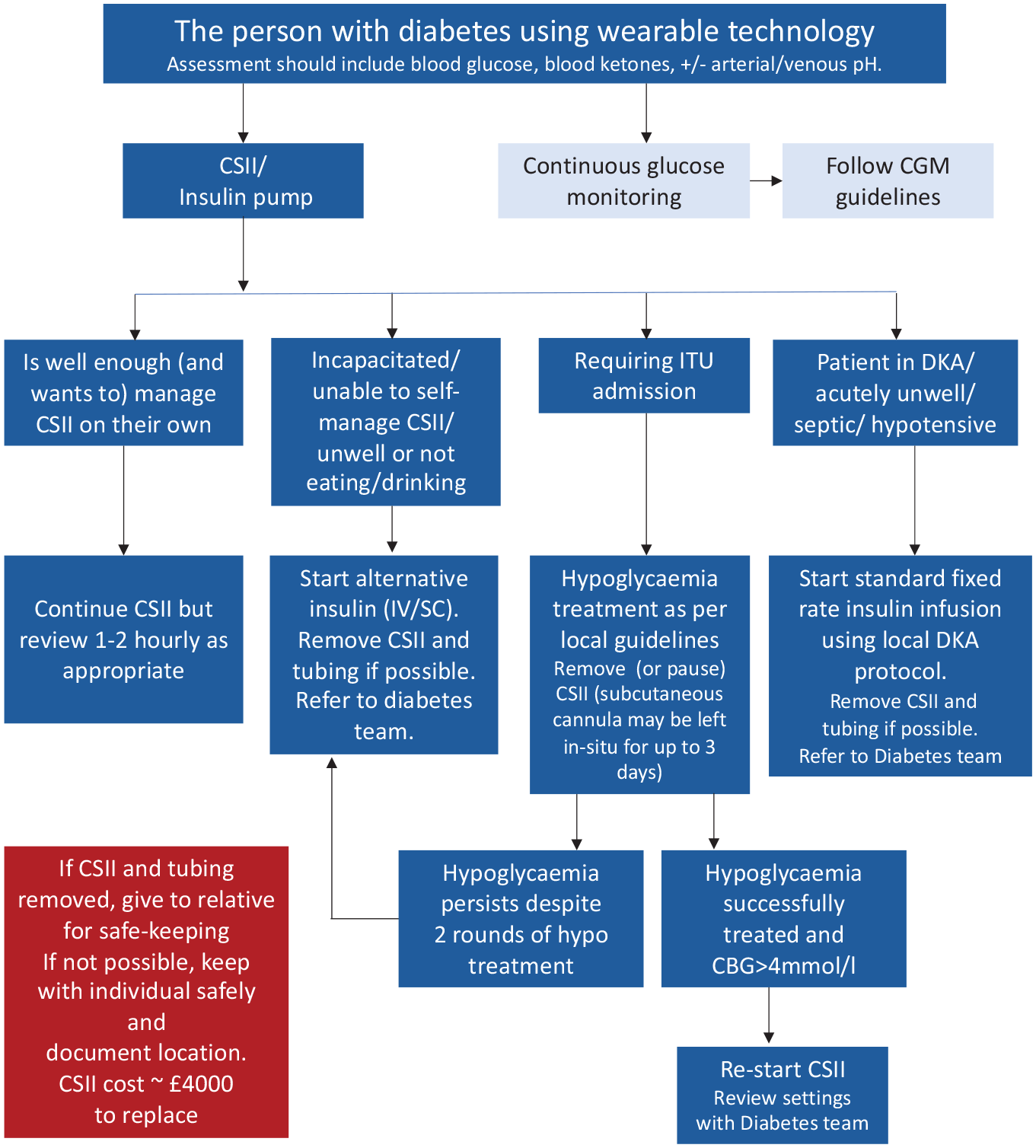
CSII management on admission to hospital. Figure adapted with permission from the Diabetes Technology Network guidelines. 6 Abbreviations: CBG, capillary blood glucose; CGM, continuous glucose monitoring; CSII, continuous subcutaneous inulin infusion; DKA, diabetic ketoacidosis; ITU, intensive care unit; IV, intravenous; SC, subcutaneous.
It is important to note that errors in insulin pump management can potentially result in severe hypoglycemia or hyperglycemia that may not be caught by typical hospital safe-guards, such as pharmacy review or scheduled point-of-care testing. Thus, continued use of CSII in the inpatient setting needs to be carefully considered. Individuals continuing with CSII in hospital should follow local hospital self-management policies. 5 People continuing CSII or hybrid closed-loop systems in the acute care setting should be provided with a detailed information sheet and hospitals may wish to consider introducing a hospital CSII policy, in addition to self-management policies already in place. An example used in the United States has been published by Galindo et al. 13
CSII Management in the “Well” Person
Unless incapacitated, most people admitted to hospital using CSII, who are physically and mentally able to continue to use their pumps, are safe to remain on CSII if admitted to hospital. It is important to note that a significant proportion of medical staff will be unfamiliar with this technology. Specialist diabetes staff are required to be available to give advice and guidance if required.
If the individual is unable or unwilling to manage the pump, unable to safely demonstrate pump settings, and/or no specialist advice is immediately available, the pump should be removed and a conventional intravenous (IV) insulin infusion or a subcutaneous basal-bolus insulin regimen started. Table 1 provides a list of other contraindications for CSII use in hospital. CSII devices are expensive and we recommend any devices removed, are labeled, stored in a safe place, and documented.
Contraindications to Insulin Pump Therapy in Hospital.

Abbreviation: CSII, continuous subcutaneous insulin infusion.
It may not be possible for hospitals to have all the necessary CSII supplies, as each device model will have different components that a hospital would not typically have on formulary, therefore, the individual or family needs to be able to provide the necessary pump supplies, such as infusion sets and insulin reservoirs. If supplies are not available, then CSII therapy will need to be discontinued while in hospital.
On admission, rapid-acting insulin used in the pump reservoir should be prescribed for use in an insulin pump (either on parenteral section of the drug chart or on electronic record). For electronic CSII order sets, hospitals should alert the medical team on how to deal with CSII discontinuation.
Individuals should be made aware of differing glucose targets in hospital, as it likely differs from outpatient goals.
CSII in the Critically Ill, Unconscious, or Incapacitated Individual
CSII continuation is not recommended in people who are critically ill or hemodynamically unstable, as well as in those who are not able to demonstrate appropriate use of their insulin pump. In a retrospective analysis, 24% of people admitted to hospital using CSII in the outpatient setting were unable to correctly demonstrate use of critical pump skills during hospitalization. 10 This underlines the importance of involving the inpatient diabetes team in assessing whether it is safe for the individual to self-manage particularly given the unusual environment of inpatient care and for those acutely unwell the stress on illness.
CSII During Hyperglycemic Emergencies
In people with diabetic ketoacidosis, the altered tissue perfusion may affect insulin absorption, making CSII unreliable, thus, CSII should be temporarily discontinued and switched to an IV insulin infusion as per local protocol. The person may be transitioned back to CSII after resolution of DKA, when they are clinically stable and when the acid-base disorder is corrected.
It is essential that all people on CSII admitted with DKA should have their equipment checked and in particular their infusion sets changed as well as their pump skills reviewed by the diabetes teams as failure of the former and/or issues with the latter may have precipitated the DKA episode. CSII should not be restarted until reasons for the hyperglycemic emergency have been determined and it is clear the individual knows what they are doing.
It is critical that hospital staff are familiar with departmental guidelines pertaining to CSII use. People using CSII do NOT usually take any long-acting insulin, so if there is any interruption to insulin delivery (eg, if the cannula is blocked or dislodged), hyperglycemia and then ketoacidosis can develop very quickly (see the “Discontinuing/Restarting CSII” section).
CSII and Radiology Investigations
The pump should be suspended and removed prior to magnetic resonance imaging (MRI) scanning, and not be taken into the scanning room. Pump manufacturers also advise removing the pump prior to computed tomography (CT) scan, however, some guidelines suggest that the pump may be covered by a lead apron. 15 For positron emission tomography (PET) scans, the pump needs to be off for at least 1 hour prior to the study (no bolus insulin < 4 hours prior). 15
For plain X-rays, there is no need to remove the pump, unless its position obscures the area of interest. The pump should be reconnected immediately after any radiological investigation.
Pumps can be safely suspended/removed for up to an hour at a time without needing alternative insulin. A correction bolus may be needed on reconnecting the pump.
CSII Management for Surgical Procedures
Minor surgical procedures (no more than one missed meal) under general anesthetic or with/without sedation
For many people undergoing minor surgical procedures (no more than one meal missed, ie, expected to eat and/or drink within 2-3 hours), they can continue using their CSII device during the procedure. The prerequisites for safe perioperative use of CSII are outlined in Table 2. However, some people may feel unable to self-manage following the procedure and should discuss this with their diabetes team in advance.
Requisites for Safe Perioperative Use of CSII (All Conditions Must Be Met).

Abbreviation: CBG, capillary blood glucose; CSII, continuous subcutaneous insulin infusion.
Fasting (especially for short durations) is not usually a problem when on CSII and standard basal rates may be continued. From the day of admission for surgery, aim for glycemia between 6 and 10 mmol/L (108-180 mg/dL).
- If blood glucose is within target range of 6 to 10 mmol/L (108-180 mg/dL) or within acceptable range of 6 to 12 mmol/L (108-180 mg/dL), standard basal rates may be continued.
- If the person with diabetes usually wakes up in the mornings with a capillary blood glucose < 6 mmol/L (< 108 mg/dL), recommend reducing the infusion basal rate to 80% of their normal rate at bedtime.
- If the person with diabetes usually has capillary blood glucose < 6 mmol/L (< 108 mg/dL) during the day, recommend reducing basal insulin to 80% of normal on awakening on the day of surgery. 16
In terms of diathermy, manufacturers advise removal of CSII devices prior to diathermy treatment due to the potential for electrical conduction and there is limited data on safety and impact on CSII performance. However, CSII is increasingly used during cesarean section with both unipolar and bipolar diathermies. 17 Guidance may therefore be individualized for each center. People should be advised that only a Teflon cannula be used, as infusion sets with steel needles are contraindicated due to the hypothetical risk. CSII should be situated away from the operative site and the diathermy pad(s).
The surgical team/anesthetists should work in close collaboration with the diabetes inpatient team, with close monitoring and a clear plan and knowledge of using CSII. Further guidance on the safe perioperative use of the CSII can be found in the multidisciplinary guidance produced by the UK Centre for Perioperative Care, 16 as well as a checklist for hospital teams to use in the perioperative period (Online Appendix).
Major surgical procedures (> 1 missed meal)
For elective and emergency admissions for major surgical procedure (> 1 missed meal) CSII should be stopped, the pump removed, and stored in a safe place (this should be documented or alternatively handed over to family/friend for safe-keeping). Once the CSII is removed, a variable rate intravenous insulin infusion (VRIII) should be started immediately. CSII can be restarted once the individual has recovered and able to manage pump (see the “Discontinuing/Restarting CSII” section).
Peripartum Pump Management
The use of CSII in pregnancy is increasing, with observational data in 161 pregnant women with T1D demonstrating that women who continued to self-manage their diabetes using CSII achieved lower glucose levels during delivery than those who were switched to IV insulin. 18 In most cases (including elective Cesarean delivery), glycemia is best achieved by continuing CSII. 17
If the mother-to-be (or her partner) does not feel confident managing the pump during labor, or if blood glucose levels are not appropriately controlled, then VRIII should be started instead. VRIII should also be considered if two consecutive blood glucose levels are above the target range (7.0 or 8.0 mmol/L [126 or 144 mg/dL]). 17 Following delivery, insulin requirements typically drop rapidly, and women are advised to revert to pre-pregnancy basal infusion rates to minimize their risk of hypoglycemia. 17
Obstetricians and anesthetists should work in close collaboration with the diabetes inpatient team, with close monitoring and a clear plan and knowledge of using these systems. Further guidance on the safe of CSII in the peripartum period is found in the JBDS-IP guidelines on “Managing hyperglycaemia during antenatal steroid administration, labour and birth in pregnant women with diabetes.” 17
CSII Use During Cardiac Arrest Situations
During a cardiac arrest situation, CSII devices should ideally be removed for external direct-current (DC) cardioversion. This is due to potential steel cannula sets, which may act as a conductor, and therefore, there is a theoretical risk of energy being dispersed at the CSII site. We recognize that in a cardiac arrest situation it may not be known whether the individual has an insulin pump in-situ and resuscitation should not be delayed, but where possible the device should be removed.
Discontinuing/Restarting CSII
If the decision is made to discontinue the CSII on admission, individuals should be transitioned to a subcutaneous insulin injection regimen consisting of basal and bolus insulin, or continuous IV insulin infusion. Similarly, if an individual’s medical condition changes during the hospital admission, and if there may be any concerns or a contraindication arise, CSII should be discontinued. For example, administered medications or analgesia may impair consciousness or cause confusion, and if any concerns, the CSII should be discontinued. Other examples include development of renal injury or hepatic failure resulting in uremia or encephalopathy, which could impair the ability of the person to operate the pump. Furthermore, decreased renal function can result in reduced insulin clearance of insulin potentially leading to hypoglycemia. 15
If transferring from CSII to subcutaneous insulin
The 24-hour basal dose of insulin delivered by CSII should be replaced by long-acting basal insulin (glargine or detemir). Insulin delivery via the pump should ideally be continued for at least 2 hours after the first dose of long-acting (basal) insulin is given or ensure some rapid acting is given at the same time as the long-acting (Table 3). Administration of basal insulin is especially important in people with T1D who need insulin on board at all times to prevent DKA. Rapid-acting insulin boluses should be given for mealtime coverage.
Starting Basal Insulin From CSII Therapy.

Abbreviations: CSII, continuous subcutaneous insulin infusion; T1D, type 1 diabetes; VRIII, variable rate intravenous insulin infusion.
Choice of basal insulin for the hospital admission should ideally include Lantus or Levemir. Ultra–long-acting insulins (Degludec or Glargine U-300) may be more challenging when switching back to CSII given their longer duration of action.
For restarting CSII, the person with diabetes is ideally best placed to restart this as they will have received training and will be experienced in this process. Individuals would need to meet the same criteria as for continuation of their insulin pump in hospital (Table 1).
If transferring from IV insulin infusion to CSII
Ask the individual to insert new cannula, restart CSII after performing a fixed prime (there is no need to wait until a meal) and wait 60 minutes before discontinuing IV insulin.
If transferring from subcutaneous insulin to CSII
Individual inserts new cannula, perform a fixed prime and restarts CSII. CSII settings may need to be reprogrammed (Table 4 provides guidance on recalculating CSII settings). The individual may need to temporarily reduce background insulin infusion rate (eg, drop to a 70% temporary basal rate for 24 hours) while long-acting subcutaneous insulin is still active, with increased glucose monitoring may be required. No further subcutaneous insulin doses should be required once CSII restarted. Capillary blood glucose should be checked 1 to 2 hours after CSII restarted.
Restarting CSII Therapy From Basal Insulin.
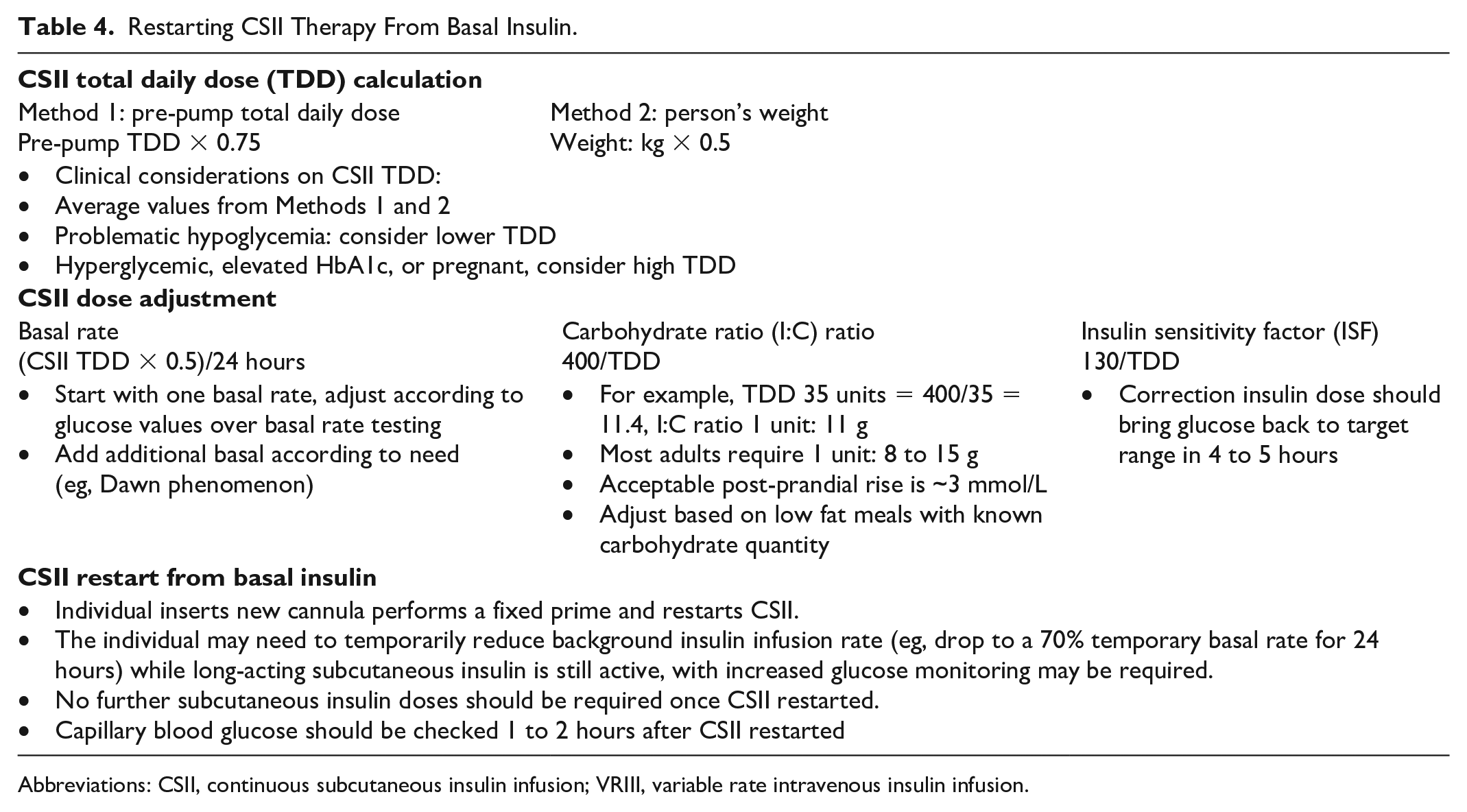
Abbreviations: CSII, continuous subcutaneous insulin infusion; VRIII, variable rate intravenous insulin infusion.
In-Hospital Use of Hybrid Closed-Loop Systems
There are limited data and guidance on real-world use and safety of hybrid closed-loop systems in the hospital setting. In research settings, small randomized trials demonstrate good efficacy data with improvement in percentage time in range, and lower average glucose levels without increased hypoglycemia.19,20 There are also some case reports suggesting safe use in hospital.21,22 Despite this evidence, the effect of hybrid closed-loop systems on clinical outcomes, the best application of these devices, and cost-effectiveness are yet to be determined. 15 It is also worth noting that many of the early studies were conducted in people with type 2 diabetes (Table 5), and the hence the safety profile for risk of hypoglycemia/ketonemia may be different. Furthermore, non-diabetes specialists will be unfamiliar with these systems.
Summary of Key Studies Assessing Hybrid Closed-Loop Use in Hospital.
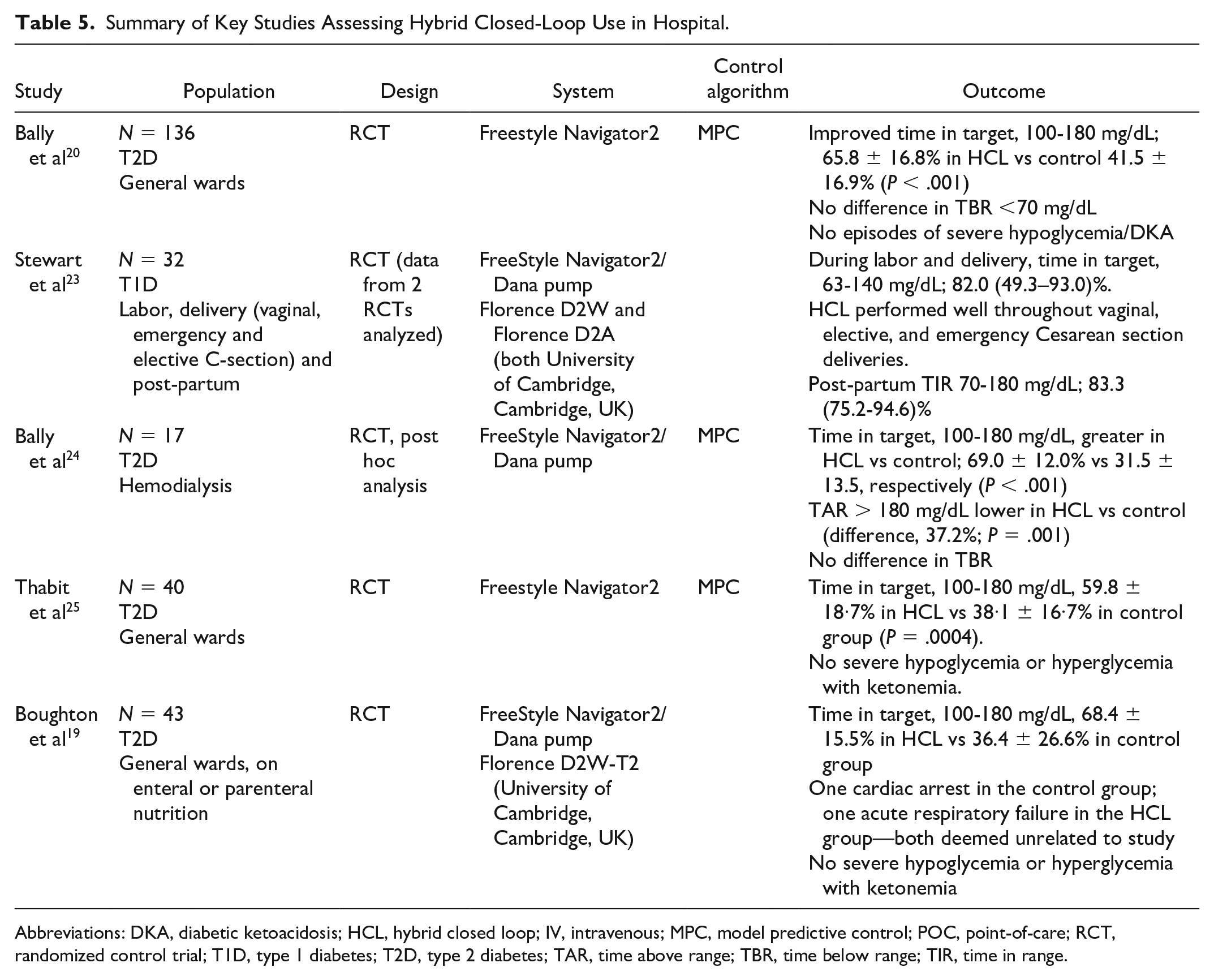
Abbreviations: DKA, diabetic ketoacidosis; HCL, hybrid closed loop; IV, intravenous; MPC, model predictive control; POC, point-of-care; RCT, randomized control trial; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range.
In someone who is well, and may be in hospital for a short elective procedure or investigation, it may be appropriate to let the hybrid closed-loop continue to control their glucose. In someone who is unwell, insulin requirements can change rapidly from day to day, therefore, JBDS-IP recommend closed-loop algorithms are “disengaged” and systems are switched to “manual” control in the hospital setting. This allows the individual with support from their diabetes team to adjust insulin pump settings, including glucose target range, insulin sensitivity factor, and basal rates. 15
Further challenging scenarios for hybrid closed-loop systems in hospital, include medications, such as glucocorticoids, which may cause severe insulin resistance and uncontrolled hyperglycemia, presenting a challenge for some hybrid closed-loop algorithms. 13 Other challenging scenarios include nausea and vomiting in people on hybrid closed-loop systems, and during periods of parental or enteral nutrition through nasogastric/percutaneous endoscopic gastrostomy (PEG) feed. 13 In these scenarios, we recommend closed-loop algorithms to be “disengaged.”
Individuals would need to meet the self-management criteria as for normal continuation of their insulin pump in hospital. It is important to recognize that individuals may need adjustment in their basal rates when switched to standard CSII mode or basal insulin, and a review should be made of glycemia, pre-admission basal rates, and number of automated insulin suspensions. It is worth noting that some hybrid closed-loop systems, such as Omnipod 5, may not provide specific details on basal patterns, but reports the total amount of insulin given in the day. In such situations, conversion to multiple daily injections can be done through estimation from total daily insulin or weight calculations (Table 4).
Hybrid Closed Loop in Surgery
There are few case reports of using hybrid closed-loop systems in surgery,21,22,26 but overall data on the safety or maximum safe duration of hybrid closed-loop control during anesthesia are limited. 13
Elective surgeries provide the opportunity for pre-admission preparation, and these systems should only be used with the anesthetist working in close collaboration with the diabetes inpatient teams, with close monitoring and a clear plan and knowledge of using these systems. Specific adjustments to a hybrid closed-loop system perioperatively are system dependent and therefore any adjustments should be made under specific guidance by a diabetologist (eg, adjusting to a higher target or switching to activity mode to achieve a higher target). Urgent surgeries do not allow for such planning, and therefore, hybrid closed-loop systems should not be used in this setting.
In the immediate perioperative period, for either elective or urgent surgical procedures, the inpatient diabetes team should be notified.
Challenges and future directions
Like CGM devices, CSII devices are infrequently encountered in the hospital, therefore, hospital staff are likely to have little familiarity with their operation. Upskilling of hospital staff is required for clinical and allied health care professionals to understand and identify between insulin delivery systems and glucose monitoring systems. Furthermore, hospital diabetes teams are required to support implementation, and at present, there remains great variation at a national level for out-of-hours and weekend specialist input (manuscript submitted for publication).
While the evidence for hybrid closed-loop systems for people in hospital is growing, clinical outcomes of this approach are yet to be determined. 15 Further costs may also arise through need for sufficient hospital pump and CGM supplies, and replacements.
Enhancement of data management platforms will also be critical in making data from different closed-loop systems readily accessible to both users and health care professionals. The integration of Continuous Glucose Monitoring Data into the Electronic Health Record (iCoDE) Project aims to facilitate integration of CGM data and provides standards towards supporting this.27,28 Generating automated clinical insights to support optimal usage of this technology, may potentially reduce workload of health care professionals (accompanying Flanagan et al paper available at https://doi.org/10.1177/1932296819849478). Data acquisition processes will need to be compliant with regulatory privacy rules and cybersecurity policies. 29
Finally, as artificial intelligence becomes more established, the dosing algorithms for insulin delivery in people in hospital will become individualized for closed-loop control of glycemia. 30 Integration of additional inputs other than glucose, such as heart rate, may enable to accurate reflection of changing insulin requirements during illness, further optimizing glycemia in hospital.
Conclusion
Wearable diabetes technology offers tremendous opportunity in optimizing glycemia for people with diabetes in hospital. CSII and CGM systems may be continued in certain individuals, with the appropriate support and protocols from the diabetes team. Further studies are required on safety, efficacy, and cost effectiveness within the hospital setting. There is now an even greater need and potential for inpatient diabetes teams to support implementation of diabetes technology in hospital.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221137335 – Supplemental material for Insulin Pumps and Hybrid Close Loop Systems Within Hospital: A Scoping Review and Practical Guidance From the Joint British Diabetes Societies for Inpatient Care
Supplemental material, sj-docx-1-dst-10.1177_19322968221137335 for Insulin Pumps and Hybrid Close Loop Systems Within Hospital: A Scoping Review and Practical Guidance From the Joint British Diabetes Societies for Inpatient Care by Parizad Avari, Alistair Lumb, Daniel Flanagan, Gerry Rayman, Shivani Misra, Pratik Choudhary and Ketan Dhatariya in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
SM is supported by the National Institute for Health Research (NIHR) Biomedical Research Centre at Imperial College Healthcare NHS Trust.
Abbreviations
CBG, capillary blood glucose; CGM, continuous glucose monitoring; CSII, continuous subcutaneous inulin infusion; DKA, diabetic ketoacidosis; HCL, hybrid closed loop; ITU, intensive care unit; IV, intravenous; JBDS-IP; Joint British Diabetes Societies for Inpatient Care; MPC, model predictive control; MRI, magnetic resonance imaging; PEG, percutaneous endoscopic gastrostomy; POC, point-of-care; RCT, randomized control trial; SC, subcutaneous; TAR, time above range; TBR, time below range; TDD, total daily dose; TIR, time in range; T1D, type 1 diabetes; T2D, type 2 diabetes; VRIII, variable rate intravenous insulin infusion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AL has received payments for speaking and advisory boards from Insulet, Dexcom, Abbott Diabetes Care, Novo Nordisk, Sanofi, and Institutional Research Support from Abbott Diabetes Care, Novo Nordisk. DF is the national lead for the UK diabetes care accreditation program and has received speaker honoraria from AstraZeneca, Novo Nordisk, and Sanofi Diabetes. SM is appointed to the Board of Trustees at the Diabetes Research & Wellness Foundation and is in receipt of funds from Dexcom for an investigator-initiated research study. GR has received personal fees from Abbott Diabetes Care, Sanofi Aventis, and Eli Lilly. PC has received personal fees from Abbott Diabetes Care, Dexcom, Diasend, Eli Lilly, Insulet, Medtronic, Novo Nordisk, Roche, and Sanofi Aventis. KD is the chair of the Joint British Diabetes Societies for Inpatient Care and has received speaker fees, travel, or taken part in advisory boards for AstraZeneca, Sanofi Diabetes, Boehringer Ingelheim, Lilly, and Novo Nordisk. PA has no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
