Abstract
Objective:
We investigated the association between the complexity of diabetic foot ulcers (DFUs) and frailty.
Research Design and Methods:
Individuals (n = 38) with Grade 2 Wagner DFU were classified into 3 groups based on the Society for Vascular Surgery risk-stratification for major limb amputation as Stage 1 at very low risk (n = 19), Stage 2 at low risk (n = 9), and Stage 3 to 4 at moderate-to-high risk (n = 10) of major limb amputation. Frailty status was objectively assessed using a validated digital frailty meter (FM). The FM works by quantifying weakness, slowness, rigidity, and exhaustion over a 20-second repetitive elbow flexion-extension exercise using a wrist-worn sensor. FM generates a frailty index (FI) ranging from 0 to 1; higher values indicate progressively greater severity of frailty. Skin perfusion pressure (SPP), albumin, and tissue oxygenation level (SatO2) were also measured. One-way analysis of variance (ANOVA) was used to identify group effect for wound complexity. Pearson’s correlation coefficient was used to assess the associations with frailty and clinical endpoints.
Results:
Frailty index was higher in Stage 3 and 4 as compared to Stage 1 (d = 1.4, P < .01) and Stage 2 (d = 1.2, P < .01). Among assessed frailty phenotypes, exhaustion was correlated with SPP (r = −0.63, P < .01) and albumin (r = −0.5, P < .01).
Conclusion:
Digital biomarkers of frailty may predict complexity of DFU and thus triage individuals who can be treated more simply in their primary clinic versus higher risk patients who require prompt referral to multidisciplinary, more complex care.
Introduction
Individuals suffering from nonhealing diabetic foot ulcers (DFUs) are mostly in the aging population with multimorbidity and frailty.1,2 Frailty is a geriatric syndrome and represents a state of multisystem impairments leading to decreased physiological reserve and resistance to stressors, leaving patients more vulnerable to poor health outcomes.3-5 It is not unusual for frail older adults to have varying degrees of “organ failures,” such as cardiac or renal. Few studies suggest that frailty may also lead to “skin failure,” one of the largest organs, increasing the likelihood of impaired healing.4,6,7 Thus, a better understanding of the interactive nature of frailty and nonhealing DFUs may assist in designing an individualized therapeutic plan for the aging population to reduce the consequences of nonhealing like lower extremity amputation (LEA).
In an ideal scenario, a multidisciplinary team comprising podiatry and vascular surgery is recommended to manage DFUs and prevent unnecessary LEA.8,9 However, this is not always feasible for community clinics (i.e., rural and city suburbs) providing care to these targeted populations. Furthermore, the multidisciplinary wound center, which is a “last resort” referral model, has its limitations as these facilities are easily overloaded due to over-taxed staff. 1 This may explain the higher risk of LEA among Hispanic, African American, and low-income populations as compared to others.10,11 To address the gap, a practical and quick triaging system to make timely and smart referall to a multidisplinary care, when needed, is despretly needed.
Digital health technologies and wearable devices related to telemedicine are poised to become an integral part of modern healthcare, mostly due to the surge in remote care necessitated by the COVID-19 pandemic.12-16 However, remote assessment of individuals with complex DFUs using current gold standard methods is complicated by the inability to assess the peripheral vascular status or the inavailability of proper technology. For instance, the Society for Vascular Surgery (SVS) Wound, Ischemia, and Foot Infection (WIfI) risk stratification system for major limb amputation requires in-person clinical visits for assessment by an expert. 17
To facilitate triaging highest-risk patients who require prompt referral to multidisciplinary care, we propose screening frailty to distinguish between patients with DFUs that can be treated more simply in their primary clinic versus those who require prompt referral to multidisciplinary, more complex care. Frailty is often characterized by assessing physical fitness, called physical frailty (PF). Multiple tools have been developed to objectively determine PF.18-20 Fried frailty phenotype has been the gold standard for clinical assessment of PF. 21 This method determines PF based on five phenotypes: exhaustion, inactivity, shrinking, slowness, and weakness. The first three phenotypes are subjectively assessed with surveys, whereas the last two are objectively measured with a grip force, and 4.5 meters walk test. The administration of this test, specifically the walking test, is challenging in patients with DFUs or lower-extremity amputation.4,22 However, the lack of the ability to walk does not necessarily indicate PF, and in addition, incomplete phenotype assessment compromises the predictive power of the tool.23,24
To improve frailty screening among individuals with limited mobility, like individuals with DFUs, we have developed and validated a frailty meter (FM) using a wrist-worn inertial sensor.5,25-27 The FM works by quantifying weakness, slowness, rigidity, and exhaustion over a 20-second repetitive elbow flexion-extension exercise using a wrist-worn sensor. The frailty meter generates a frailty index (FI) ranging from 0 to 1; higher values indicate progressively higher severity of PF. The FM has been applied to mobility-impaired individuals and can be remotely administered, unlike conventional frailty assessment tools, which require mobility tests such as gait assessment and in-person visits.5,25,28
Previously, FM was validated against the Fried frailty Phenotypes Criteria, 29 among 117 community-dwelling older adults 25 with a sensitivity of 100% and 87% and specificity greater than 95% for frail and prefrail cases, respectively. The FM has also been used in several clinical studies, including prediction of adverse events postvascular surgery, early-stage Alzheimer’s screening, and prediction of adverse events among long-term obstructive pulmonary disease patients.4,30-33 We hypothesize that frailty determined using FM is associated with the complexity of DFUs stratified based on the WIfI stage. In addition, we hypothesize that frailty-related phenotypes will be correlated with some of the key risk factors associated with nonhealing wounds, such as malnutrition, poor skin perfusion, and poor tissue oxygenation level.
Research Design and Methods
Participants
This is a cross-sectional cohort study. The cohort of this study was generated from a subanalysis of two previously approved protocols for people with nonhealing DFU (NCT03476876, NCT03821675, approved by the local Institutional Review Board [IRB] at the Baylor College of Medicine (Houston, TX)). Inclusion criteria involved age of 18 years and older, clinically diagnosed with type 2 diabetes, and the presence of one or more noninfected Grade 2 Wagner Ulcer classification or long-term (more than 4 weeks without healing) wounds. Individuals with a severe active infection; gangrene or osteomyelitis, inability to fully comply with follow-up protocol, pregnancy, nursing or actively lactating, active Charcot foot, dementia or impaired cognitive function, and excessive lymphedema were excluded from the study. All participants provided written informed consent before initiating any assessment or data collection.
Measurement Procedure
All assessments were collected in the outpatient vascular clinic at Baylor St Luke’s Medical Center (Houston, TX, USA). Participants were asked to perform a 20-second repetitive elbow flexion and extension exercise while performing a memory task (counting backward from a two digits number, e.g., 80). Using a wrist-worn sensor (Biosensics LLC, MA, USA) attached to the dominant arm, elbow flexion-extension speed was measured, and then using a validated machine learning model,5,25-27 Frailty index (range between 0 to 1, higher value indicate more frailty severity) and key frailty phenotypes like slowness, weakness, and exhaustion were estimated.
Research coordinators (N.M., R.B. and A.Z.) reviewed electronic medical records for patient medical history, demographic data, and other relevant clinical or laboratory assessments. Albumin levels (g/dL) were collected to determine protein malnutrition status. Participants were considered at risk of malnutrition if albumin levels were less than 3.5 g/dL. Several patient-reported outcomes were collected, including overall well-being using the Patient-Reported Outcomes Measurement Information System (PROMIS), 34 sleep quality assessed using the Pittsburgh Sleep Quality Index (PSQI), 35 pain assessed using a visual analog scale (VAS), 36 cognition using the Montreal Cognitive Assessment (MoCA), 37 concern for falls using the Falls Self-Efficacy Scale (FES-I), 38 and the depression level using the Center for Epidemiologic Studies Depression scale (CES-D). 39
To quantify the complexity of wounds, we objectively measured wound area, skin perfusion, and tissue oxygenation saturation, known clinical markers for nonhealing wounds. The wound area was assessed with a noninvasive 3D camera (Silhouette®, Aranz Medical, Christchurch, NZ), which provides wound length, width, and depth. Skin perfusion pressure (SPP) was assessed using the SensiLase system (Väsamed, Minnesota, USA). A sensor and a blood pressure cuff were placed around the distal gastrocnemius ipsilateral to the evaluated wound, and the cuff was inflated to determine the SPP. Additional parameters for wound perfusion included tissue oxygen saturation (SatO2) and total tissue hemoglobin (TTHb). These values were determined using a validated, noninvasive near-infrared (NIR) spectroscopy camera (Snapshot NIR, KENT Imaging Inc., Calgary, AB, Can). The wound perimeter was traced using the NIR camera to determine the SatO2 and TTHb levels, providing a possible indication for tissue perfusion.40-42
A board-certified podiatrist or vascular surgeon administered WIfI staging to determine the risk of LEA and wound complexity according to the guidelines developed by the SVS. 17 The examiner was blinded to the frailty status of the individual. Wounds were graded on a scale of 0 to 3 based on its measurements and tissue loss rate. Ischemia was graded on a scale of 0 to 3 based on the patient’s Ankle Brachial Index (ABI), toe pressure (TP), or transcutaneous oxymetry TcPO2 value. Foot infection was graded on a scale of 0 to 3 and determined based on laboratory results and clinical manifestations of infection. 17 A final WIfI score was calculated based on the clinical stages for risk of major limb amputation according to the SVS guidelines as follows: clinical stage 1 = very low risk, clinical stage 2 = low risk, clinical stage 3 = moderate risk; and clinical-stage 4 = high risk. Considering the sample size in this study, stages 3 and 4 were merged as a single group as they represent severe wounds.
Statistical Analysis
All continuous data were presented as mean ± standard error. All categorical data are expressed as counts and percentages. The chi-square test was performed to identify between-group differences for categorical data (eg, sex and previous revascularization). One-way analysis of variance (ANOVA) was performed to assess group effect (ie, stratified based on WIfI: Stage 1, Stage 2, and Stage 3-4 combined). Normality and homogeneity of variances were assessed using the Shapiro–Wilk and Levene’s tests (P > .05). When a significant result was detected, the least significant difference (LSD) post hoc test was conducted to compare each pair of groups separately when the equality of variances was assumed. We used Tamhane’s T2 post hoc test when the equality of variances was not assumed. We used the nonparametric Kruskal–Wallis H test for non-normality distributed data to examine the difference between the groups. We used Mann–Whitney tests as the post hoc test for significant results identified using the Kruskal–Wallis H test. The effect size was expressed using Cohen’s d effect size and represented as d in the Results section. Values ranging from 0.20 to 0.49 indicate small effects, and values between 0.50 and 0.79 indicate medium effects. Values ranging from 0.80 to 1.29 indicate large effects, and values above 1.30 indicate very large effects. 43 Pearson’s correlation coefficient was used to assess the associations with frailty and clinical endpoints. Cut-offs of 0.01 to 0.19: negligible, 0.20 to 0.29: weak, 0.30 to 0.39: moderate, 0.40 to 0.69: strong, and 0.70 to 1.00: very strong were selected for correlations. 44 We assumed a P value less than .05 as significant to reject the null hypothesis. All statistical analyses were performed using SPSS statistical software version 28 (IBM) and MATLAB version R2021b (MathWorks).
Results
Table 1 summarizes the differences between the group for demographic, patient-reported outcomes, clinical information, and wearable-based frailty outcomes. Based on WIfI, 19 individuals (age = 61.3 ± 2.8, body mass index [BMI] = 31.1 ± 1.6, female = 26%) were classified as stage 1, 9 individuals (age = 60.8 ± 4.0, BMI = 31.1 ± 2.1, female = 33%) were classified as stage 2, and 10 individuals (age = 61.6 ± 4.0, BMI = 30.1 ± 2.5, female = 40%) were classified as stage 3 to 4. There was no group difference for demographics (age, BMI, and sex, P > .05). The PROMIS scores were significantly higher among individuals belonging to the stage 1 compared to stage 2 (d = 1.4, P < .01) and stage 3 and 4 (d = 1.4, P < .01). The MoCA scores were significantly lower among individuals belonging to stage 3 and 4 compared to stage 1 (d = 0.92, P = .03). Furthermore, reported depression symptoms were significantly higher among individuals belonging to stage 2 (d = 1.2, P = .02) and a similar, but not significant, trend was observed for stage 3 and 4 (d = 0.90, P = .06) compared to stage 1.
Demographic, Patient-Reported Outcomes, and Clinical Information (Mean ± S.E.).
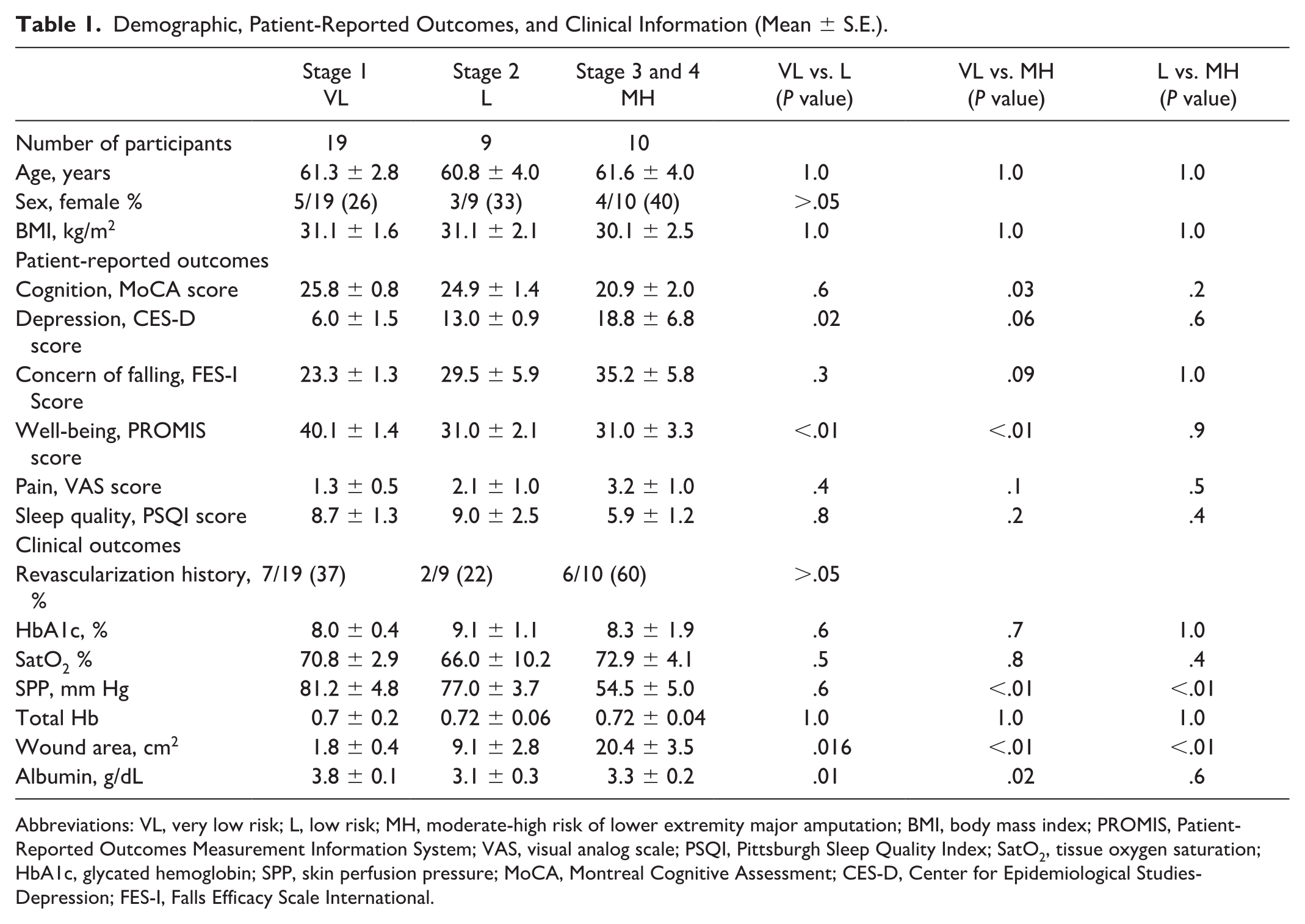
Abbreviations: VL, very low risk; L, low risk; MH, moderate-high risk of lower extremity major amputation; BMI, body mass index; PROMIS, Patient-Reported Outcomes Measurement Information System; VAS, visual analog scale; PSQI, Pittsburgh Sleep Quality Index; SatO2, tissue oxygen saturation; HbA1c, glycated hemoglobin; SPP, skin perfusion pressure; MoCA, Montreal Cognitive Assessment; CES-D, Center for Epidemiological Studies-Depression; FES-I, Falls Efficacy Scale International.
Results showed that all the data satisfied the normality assumption, except exhaustion. We observed significant difference for FI (P < .01) and post hoc analysis showed significantly higher FI in DFUs at stage 3 and 4 compared to DFUs at stage 1 (d = 1.4, P < .01) and stage 2 (d = 1.2, P < .01) stage 2 (Figure 1). Furthermore, results suggest that the exhaustion index is increasing with an increase in complexity of DFU (P = .023). The pairwise comparisons showed significantly lower exhaustion in DFUs at stage 1 compared to stage 3 and 4 (d = 0.85, P = .016). Similarly, the exhaustion index was lower in stage 1 compared to stage 2 with medium effect size, but the trend did not achieve a statistical significance level in our sample (d = 0.72, P = .076; Figure 1).
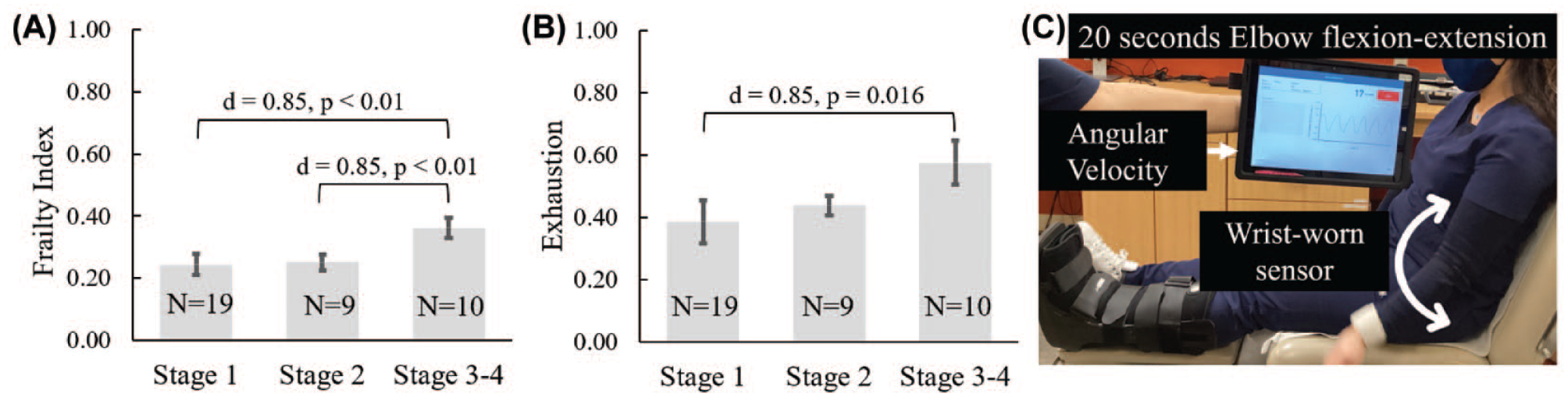
(A) Represents frailty was prevalent among individuals with diabetic foot ulcers (DFUs) at stage 3-4 compared to individuals with DFUs at lower or moderate risk of lower extremity amputation (LEA). (B) The level of exhaustion was higher among individuals with DFUs at stage 3-4 compared to individuals with DFUs at stage 1 and similar trend among individuals with DFUs at stage 2. Furthermore, (C) demonstrate the frailty meter (FM) protocol."d" denotes Cohen's effect size. "N" represents the number of subject in each group.
As expected, DFUs at stage 3 and 4 had larger wound area compared to DFUs at stage 1 (d = 3.1, P < .01) and stage 2 (d = 1.3, P < .01). Moreover, we found significant higher group differences for Albumin level (P = .02), and SPP (P < .01) in DFUs at stage 3 and 4. Pairwise comparison showed DFUs at stage 3 and 4 had lower blood perfusion compared to DFUs at stage 1 (d = 1.9, p < .01) and stage 2 (d = 1.6, p = .02). In addition, risk of malnutrition among individuals with DFUs at stage 1 was significantly lower compared to stage 2 (d = 1.1, P = .01). But despite the large effect size, the trend was not significant in our sample, when DFUs at stage 1 and DFUs at stage 3 and 4 were compared (d = 0.96, P = .06). Furthermore, we found the level of exhaustion was associated with the blood perfusion (r = −0.63, P < .01), risk of malnutrition (r = −0.5, P < .01), and trend toward association with tissue oxygenation level (r = −0.33, P = .051) across the WIfI stages (Figure 2).
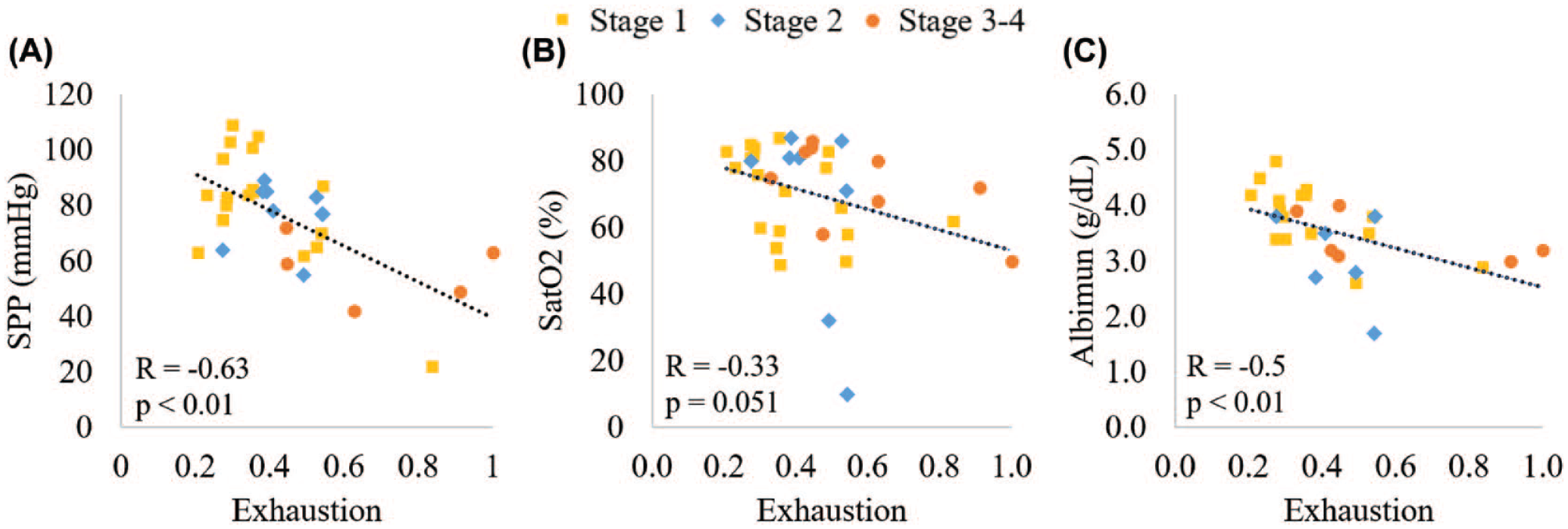
Representations of association between the level of exhaustion with the (A) skin blood perfusion (SPP, mmHg), (B) tissue oxygenation level (SatO2, %), and (C) risk of malnutrition (albumin, g/dL) across the wound, ischemia, and foot infection (WIfI) clinical stages. Abbreviations: SatO2, tissue saturation oxygenation level; SPP, skin perfusion pressure.
Discussion
This study aimed to evaluate the digital frailty screening of people with DFUs across the WIfI spectrum. Our goal was to determine whether a digital assessment of frailty and its phenotypes can be used as digital biomarkers of DFU complexity. Our results showed that the digital FI was higher in individuals with DFU with a higher risk of LEA (ie, WIfI stages 3 and 4); this supports our first hypothesis. Among assessed frailty phenotypes, the exhaustion index was the most sensitive digital biomarker to determine wound severity. We speculate that exhaustion or fatigue reflects malnutrition and poor perfusion, two major risk factors with nonhealing wounds. This is supported by prior studies suggesting that inadequate nutrition is invoked as one of the mechanisms underlying fatigue. 45 In addition, other studies suggested that fatigue is associated with anemia and poor perfusions.46,47 Prior studies also support that both inadequate nutrition and impaired perfusion are predictors of nonhealing wounds.46,48 Further exploration supports the reciprocal association between fatigue, inadequate nutrition, and poor skin perfusion, as a moderate to large correlation was observed between exhaustion index, albumin level, SPP, SatO2, and WIfI clinical stages.
While these clinical outcomes provide valuable information about DFU and vascular conditions, they are not assessed regularly due to limited time. A quick frailty assessment using a wearable device can provide a practical digital biomarker, which may serve as a surrogate for these clinical outcomes. Moreover, a higher level of exhaustion may indirectly indicate a higher risk of complications, 49 including malnutrition and lower-extremity skin perfusion and tissue oxygenation levels. Therefore, patients identified with exhaustion phenotype may take a different health trajectory (i.e., longer healing duration, higher chances for recurrent wound) than individuals with similar conditions without exhaustion.
Recently, a systematic review emphasized adopting the effective and rapid triaging of the patients as this was one of the key factors for the multidisciplinary team’s success in reducing the risk of major amputations. 8 While 31 out of 33 studies (96%) showed a decrease in major amputation due to a multidisciplinary team, the tools used for triaging were basic and not standardized. 8 The specialists involved in the multidisciplinary teams are a limited resource. 1 Therefore, it is crucial to involve the primary-care providers in taking care of less-complicated DFUs and be empowered to timely triage high-risk patients so that the multidisciplinary teams can focus on patients with severe ulcerations. The proposed FM can be instrumental in triaging high-risk patients to the multidisciplinary DFU team in the real-world setting and reducing LEA risk. This, in return, may advance health equity, reduce scheduling backlogs, reduce emergency department (ED) visits, and, most importantly, reduce the likelihood of lower limb amputation among the underserved population.
Furthermore, our results are aligned with previous authors 50 suggesting that patients with DFU are likely to experience a lower quality of well-being, with increased cognitive impairment and depression as wound complications increase. Therefore, DFU complications are not only limb-related but psychological features must be taken into consideration by the clinicians involved in the treatment of DFU patients.
Future studies are warranted to test the efficacy of this triaging model of care to improve patient referral workflow and reduce limb amputation among the underserved population. In particular, the longitudinal scope of this study is warranted to assess wound reoccurrence rate, time to heal, and LEA. We expect that patients with higher levels of exhaustion would present higher rates of wound reoccurrence. This means that patients with lower measured baseline exhaustion may remain wound-free for more extended periods. Ultimately strengthening the use of frailty assessment as a digital biomarker for wound prognosis.
Limitations
There were several limitations to our study. The study’s main limitation was our limited sample size of 38 patients and the single-center design. We also did not capture the entire spectrum of WIfI-outcomes, given our study’s exclusion of DFUs with active infection. The trials involved in this cross-sectional study excluded individuals with severe active infection, gangrene, or osteomyelitis due to protocol treatment. However, these features should be included in future studies as this is a crucial group in tertiary care centers that often overload the work of healthcare providers. Ideally, the study cohort should be expanded to include patients with DFUs across multiple centers to increase the sample size. Second, this study was cross-sectional; as such, there was no patient follow-up reported. Lastly, we did not evaluate the correlation of frailty with LEA but used the WIfI score as a marker of risk of LEA.
Conclusions
Digital biomarkers of frailty, measured over a 20-second test, may serve as a practical tool to triage patients with nonhealing DFU. Patients identified with the exhaustion frailty phenotype may be at high risk to presenting higher wound severity. These findings may advance health equity, reduce scheduling backlogs, and risk for LEA among underserved populations. While, in our cohort, those with active infection were excluded, this exclusion criterion would have minimum impact on the usefulness of FI for triaging those who require prompt referral to multidisciplinary and, in particular vascular clinics. Future studies are warranted to test the efficacy of this triaging model of care to improve patient referral workflow and reduce lower limb amputation among the underserved population.
Footnotes
Acknowledgements
The authors wish to thank Ms. Maria Noun who contributed in IRB application and coordination of activities between investigators and Ms. Naima Rodriquez, who contribution in participant recruitment.
Abbreviations
ABI, ankle brachial index; DFU, diabetic foot ulcers; FI, frailty index; FM, frailty meter; LEA, lower extremity amputation; NIR, noninvasive near-infrared; PF, physical frailty; SatO2, tissue saturation oxygenation level; SPP, skin perfusion pressure; SVS, Society for Vascular Surgery; TTHb, total tissue hemoglobin; WIfI, Wound, Ischemia, and Foot Infection.
Author Contributions
R.M. performed the statistical analysis, interpretation of data, drafting the original manuscript, and critical revision of the manuscript for important intellectual content. R.B. and A.Z. were involved in data collection, and critical revision of the manuscript. C.P., M.F., J.R., and B.L. contributed in interpretation of data, critical revision of the manuscript for important intellectual content. B.N. contributed in study concept and design, critical revision of the manuscript for important intellectual content, and securing funding.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.N. is serving as a consultant for BioSensics LLC, which is a manufacture of frailty meter used in this study. However, his consultation is not relevant to the scope of this study. He was also not involved in data analysis from this study. No other potential conflicts of interest relevant to this article were reported. Disclosure for other authors is “none.”
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported, in part, by the National Institutes of Health/National Institutes on Aging (award no. 1R42AG060853-01), the National Cancer Institute (award no. 2R44AG061951-02), and LifeNet Health (VA, USA), and AVAZZIA Inc. (TX, USA). The study sponsors were not involved in the design of the study, the collection, analysis, and interpretation of data, writing the report, or the decision to submit the report for publication. M.F. is a 2020 Fulbright scholar and was supported by an Australian-American Fulbright Future Scholarship.
