Abstract
Background:
Stay-at-home orders associated with the SARS-CoV-2 (COVID-19) pandemic were particularly important for older adults with type 2 diabetes, at risk for severe COVID-19 complications. In response, research shifted to remote telehealth methodology. Study participant interests, equipment needs, and ability to adapt methods to the remote/telehealth environment were unknown. Study purposes to assess (1) resource needs (internet/devices accessibility), (2) future telehealth interests, and (3) ability to adapt common research and clinical measures of glycemic control, physical function, activity measures, and quality of life outcomes to a telehealth setting.
Method:
Twenty-one participants with type 2 diabetes and peripheral neuropathy were recruited from a longitudinal study (11 female; age: 66.3 ± 8.3 years; DM: 15.1 ± 8.7 years). Technology needs and future telehealth interests were assessed. A glycemic measure (HbA1c), a five-times chair rise, a one-week activity monitor, and surveys (self-efficacy, depression, and balance) were collected. All aspects of the study were completed remotely over email and video/phone call.
Results:
Twelve participants used computers; nine used phones for study completion. Participants had the following resource needs: connectivity (n = 3), devices (n = 6), and technical support (n = 12). Twenty people expressed interest in participating in future telehealth studies related to balance, exercise, and diabetes management. Methodological considerations were primarily the need for assistance for participants to complete the home HbA1c test, five-time chair rise, wearable activity monitoring, and surveys.
Conclusions:
Older adults with type 2 diabetes and peripheral neuropathy would need technological and personal assistance (connection, device, guidance) to complete a long-term telehealth intervention. Despite technology needs, participants were interested in telehealth interventions.
Clinical Trial:
Parent study, “Metatarsal Phalangeal Joint Deformity Progression—R01 (NCT02616263) is registered at https://clinicaltrials.gov/.
Introduction
Individuals at high risk of severe COVID-19 symptoms and associated complications, including older individuals with type 2 diabetes, were recommended to adhere closely to stay-at-home guidelines and research shifted to the telehealth environment. Access to telehealth medical care changed quickly during this time. Between March and April 2020, NYU Langone Health observed their telehealth visits increase 683%, with the highest usage of telehealth being patients (age: 20-44). 1 With this rapid increase of telehealth visits for non-urgent care, the visits showed feasibility and satisfaction as an alternative to in-person visits.1,2 Meta-analyses showed that individuals with type 2 diabetes using telemedicine reported improved glycemic control compared with regular care.3,4 However, when using telehealth, minority and older patients had mixed results. 5 Technology literacy and accessibility were two barriers to successful monitoring of their health. 5 It is unclear how findings and methods from medical care translate to the research environment for our study participants, who are older adults with type 2 diabetes and peripheral neuropathy.
The purposes of this study were to assess our study participants: (1) technology resource needs (internet/devices accessibility), (2) future telehealth interests, and (3) ability to adapt common research and clinical measures of glycemic control, physical function and activity measures, and quality of life outcomes to a telehealth setting.
Methods
Participants were recruited from a longitudinal, randomized controlled trial (NCT02616263). We measured participants’ technology capability, interest in future telehealth interventions, glucose control, activity levels, and quality of life surveys near the end of the most strict phase of COVID-19 pandemic restrictions in our locality (St. Louis, Missouri, November 2020-March 2021). All aspects of the study were completed remotely over email and a video/phone call. The participants gave written consent to participate in the parent study (IRB# 201511090). To participate in the substudy, participants received a consent letter in the mail and then provided verbal consent during a phone call. The study (IRB# 202007197) was approved by the University’s Institutional Review Board and adhered to the guidelines of the World Medical Association’s Declaration of Helsinki.
Twenty-one of the 30 contacted individuals from the parent study agreed to participate. Participants were contacted and offered to participate in the substudy by their order of entry into the parent study. An effort was made to recruit an equal number of females/males and a representative sample of race based on our city statistics. Participant characteristics are found in Table 1. Participant inclusion criteria for the parent study included type 2 diabetes mellitus and peripheral neuropathy (full details are reported in previous publications).6,7
Participant Characteristics.

A Zoom (Zoom Video Communications Inc, San Jose, California) video or phone call was scheduled with each participant based on their preference. We queried the participants about (1) technology resources they would need to successfully participate in the telehealth intervention (need for internet connection, a device—iPad/tablet, and/or technical support), (2) level of interest (definitely-, probably- or might be interested; probably not- or definitely not interested), and (3) types of interventions they would find most beneficial (open-ended question) and multiple choices were allowed.
Hemoglobin A1c (HbA1c) home test kits (PTS Diagnostics, Whitestown, Indiana) were mailed to the participants and were collected during the call. Directions (paper and video) for HbA1c collection were provided by the manufacturer. For participants using Zoom, the video was shown prior to collecting their blood. Each kit had multiple tests in case errors resulted and the test needed to be repeated. Participants also reported their HbA1c results from the last three months, measured by their clinician.
During the call, the participants performed a five-times sit-to-stand chair rise test while the researcher recorded the time. The participants used a nonwheeled chair placed against a wall to maintain safety. Time began as soon as the participant started to stand and ended when the participant was seated from the fifth chair rise. Individuals performed two trials with approximately 30 to 60 seconds of rest between trials. During the rest time, the researcher inquired if they were able to do another trial and if they had any dizziness, shortness of breath, lightheadedness, or nausea. For the participants using Zoom, they set the phone/tablet in a location where the researcher could view the task and time it appropriately. For the participants using a phone (no video access), they were asked to begin the task when the researcher said, “Go,” and they were to count out loud each time they sat down. The researcher stopped timing after the fifth chair rise was completed.
Participants received an Actigraph wGT3X-BT activity monitor (ActiGraph, LLC, Pensacola, Florida) in the mail prior to the video/phone call. The activity monitor was worn on the nondominant wrist for seven days and then returned. It has been shown that wearing the activity monitor on the wrist improves wearing compliance 8 yet may overestimate the number of steps.9,10 The monitor was downloaded using the ActiLife licensed software where the data were integrated into 60-second epochs prior to being analyzed. Each day was separated into sedentary, light, moderate and vigorous activity levels (minutes/day and percentage of time spent in each level of activity). The first and last days were not used in the final analysis, because each day did not constitute a full 24 hours. The activity during the middle days was averaged and is reported for descriptive purposes and to show feasibility/participant compliance. Percent of time spent in each level and average number daily steps was recorded.
Participants were emailed a Research Electronic Data Capture (REDCap: created by Vanderbilt University, a secure web platform for building and managing online databases and surveys) link to the surveys for the study. For the participants without email access, a researcher asked the survey questions and recorded their responses into the REDCap database. They completed the following Patient-Reported Outcomes Measurement Information System (PROMIS) surveys to assess quality of life domains: (1) PROMIS Bank v1.0: Self-Efficacy for Managing Chronic Disease, Emotions, Social Interactions, and Daily Activities; (2) PROMIS Short Form v1.0: Depression 4a, Pain Interference 4a, Fatigue 4a, Anxiety 4a. Self-Efficacy Managing Chronic Disease: Emotions, Social Interactions, and Daily Activities surveys have a clinical population with chronic disease for reference.11,12 The short form series v1.0 (Depression 4a, Pain Interference 4a, Fatigue 4a, Anxiety 4a) uses a general population as its reference.13,14 The PROMIS surveys have been found to be valid and reliable in a variety of patient populations, including older adults.15-17 These surveys have been designed with a mean T score of 50 for each measure and a standard deviation of 10. If the T score is higher than 50, then more of the concept is being measured. 18
The Activities-specific Balance Confidence (ABC) is a measure of confidence in balance.19,20 The scoring system is an 11-point scale and rating that consists of whole numbers (0-100) for each item. 19 ABC scores lower than 50 indicate a low level of physical functioning; scores between 50 and 80 indicate a moderate level of function, and scores above 80 indicate physically active older adults. 20
The University of Alabama at Birmingham (UAB) Life-Space Assessment was used to determine participants’ mobility within their homes and communities. 21 Scores range from 0 to -120, where zero represents a person who is bedbound and 120 represents one who travels daily out of town without assistance. 21
Statistical Analysis: Lin’s concordance correlation coefficient (CCC) 22 was used to compare the home test kit to the self-report. A 95% confidence interval (CI) was calculated to quantify the agreement between the home test kit result and the self-report. The ranges used for the CCC can be interpreted as almost perfect (>0.99), substantial (0.95-0.99), moderate (0.90-0.95), and poor (<0.90). 23
Results
Twelve (57%) participants used Zoom to complete the study: 11 used their personal device, and one participant borrowed a laptop in order to complete the study. The remaining nine participants (43%) used their phones to complete the study: four participants were unable to download the Zoom software and five participants had no connectivity or disinterest in using a computer.
Telehealth interest survey results are found in Figure 1. Briefly, 20 of 21 participants expressed interest in participating in a future telehealth intervention. Balance, exercise, and diabetes management were the interventions of most interest.
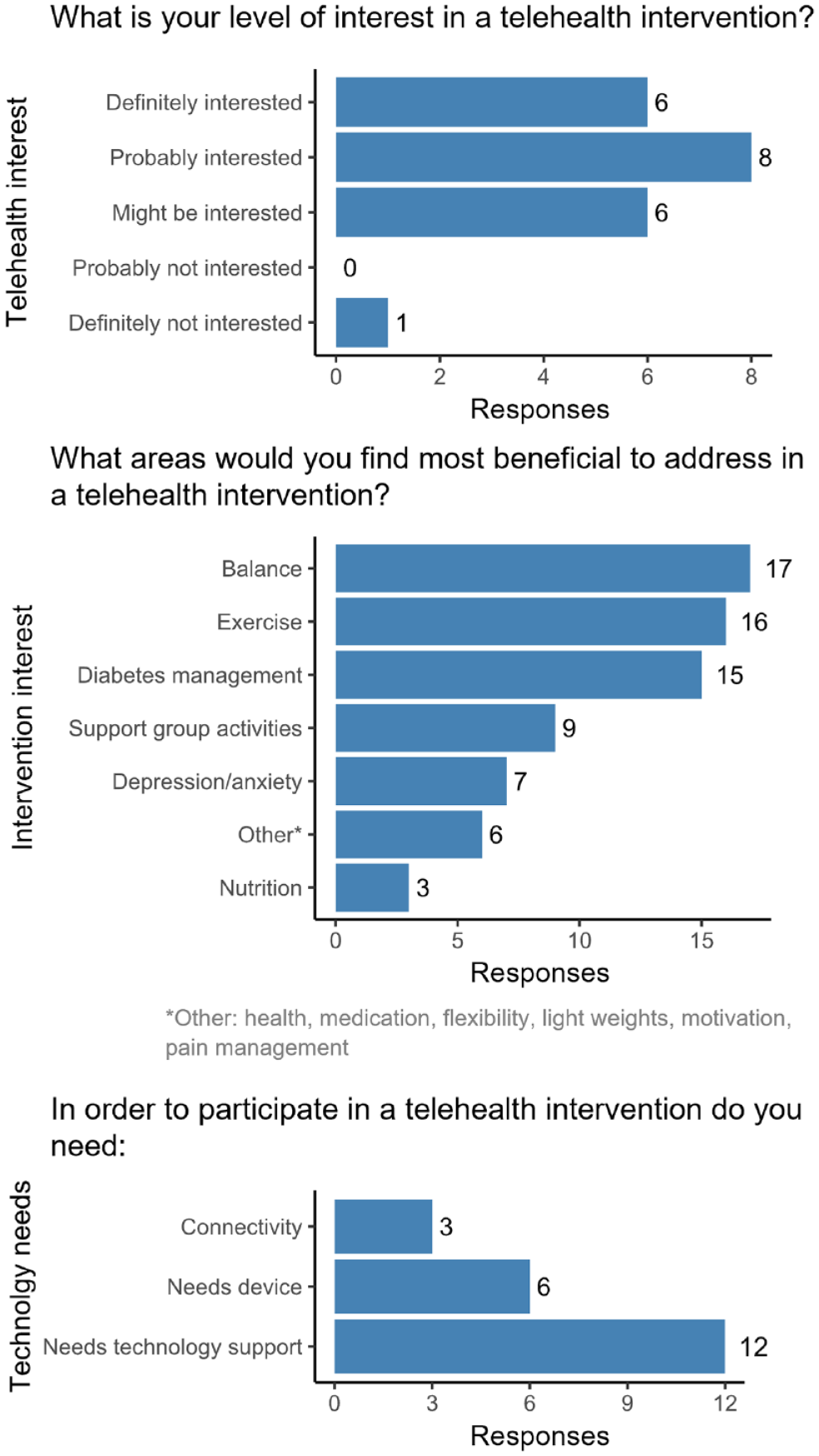
Telehealth survey results.
All participants were able to don and doff the physical activity monitor and return the monitor to the researcher by mail. The physical activity results are found in Table 2 and Figure 2 and indicate a large variation in daily steps (Figure 2B) and in sedentary, light, and moderate activity; no vigorous activity was recorded (Figure 2C and 2D).
Physical Activity Results.
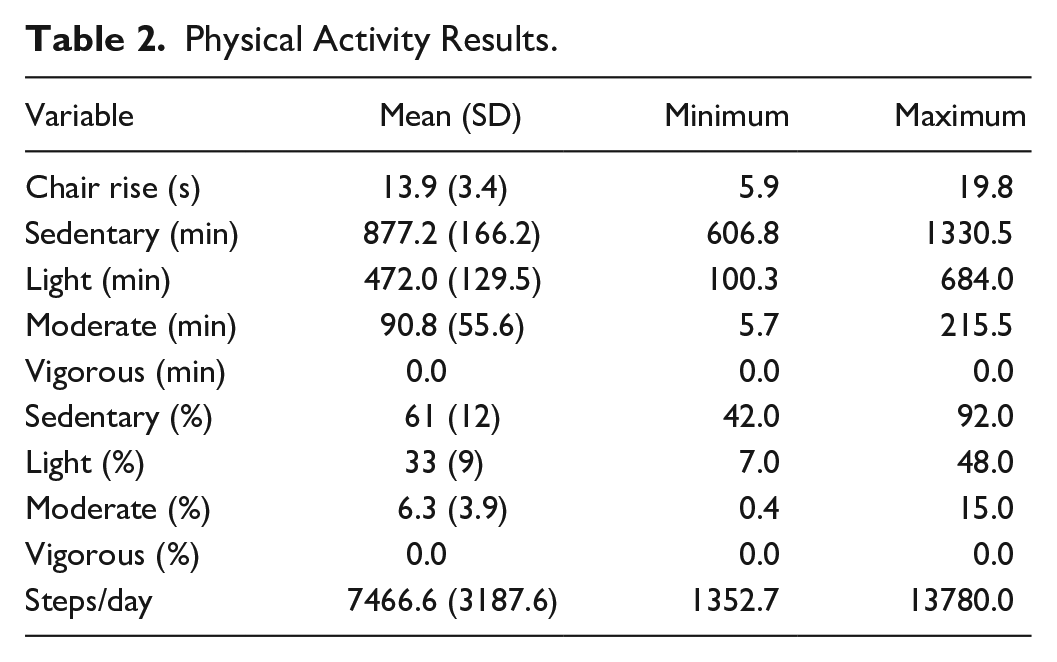
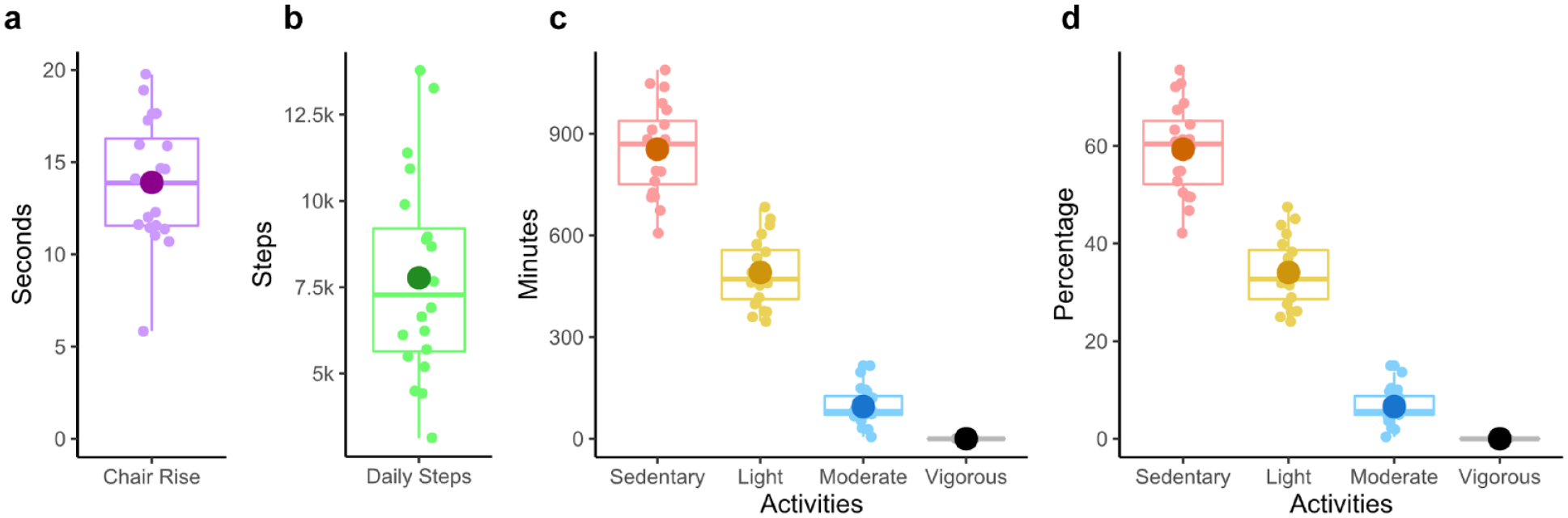
Physical activity results.
We were able to collect chair rise task data on 20 of 21 individuals, with times ranging from 5.9 to 19.8 seconds (Figure 2A). One person only had a wheeled chair and did not complete the chair rise task.
The HbA1c and survey results are found in Table 3. All participants required step-by-step verbal instructions from the researcher during blood collection and processing to perform the methods correctly. The HbA1c results from the home kit and the clinical self-reports showed inconsistencies. Therefore, we retrospectively examined agreement between the home test kit and participant self-report of their most recent laboratory HbA1c test result using CCC. 22 The CCC was 0.74, 95% CI = [0.41, 0.89], suggesting a poor relationship between the home kit and the self-reported laboratory value of HbA1c.
Glycemic Control and Survey Results.
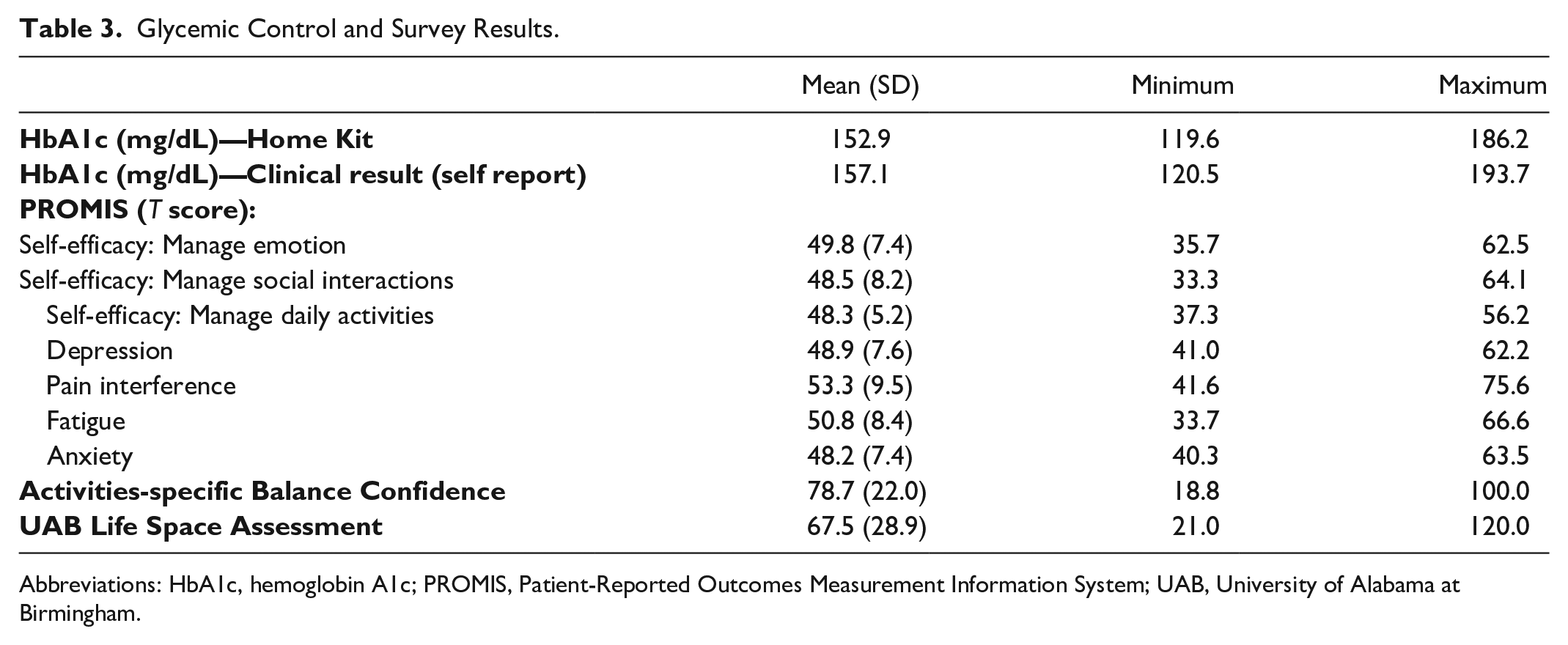
Abbreviations: HbA1c, hemoglobin A1c; PROMIS, Patient-Reported Outcomes Measurement Information System; UAB, University of Alabama at Birmingham.
Table 3 summarized all 21 participants’ survey responses. The responses were completed via email or phone. No individuals reported any difficulties completing the surveys and no individuals reported difficulty managing their emotions, social interactions, and daily activities.
Discussion
The technology acceptance model (TAM) 24 and TAM2 25 showed that users will accept technology if they perceive it to be easy and useful to their lives.24,25 These models are reflected well in our substudy as all participants use smart phones daily and more than half (57%) were able to successfully participate in a Zoom-based telehealth session on a computer/tablet. All participants indicated a need for some type of technology assistance, ranging from connectivity to a device to the need for technology education. To successfully complete a future telehealth study, we would need to ensure connectivity for all participants and improve technology literacy5,26 with user-friendly applications. 27
The results from the survey inquiring about their choices for telehealth interventions indicated the top three choices were balance, exercise, and diabetes management. These are excellent choices given the deficits measured in physical activity and diabetes management. Although our cohort did not report deficits in balance as measured by the ABC questionnaire, the request for balance intervention suggests they either recognize the importance of balance28,29 or have identified balance deficits in themselves that were not captured by the survey. For a telehealth intervention, exercise and diabetes management are important for improving strength and maintaining glycemic control.30-32 During COVID-19, telehealth visits have been shown to help keep glycemic measures in better ranges, 33 as well as improve the use of continuous monitoring devices. 34
All participants were able to complete the HbA1c at-home test kit, though it did require precise instruction from the researcher. These home kits have been found to be accurate and reliable by the manufacturer. Yet, the HbA1c values using Lin’s CCC from the at-home kit with participant self-report of their most recent laboratory value indicated poor agreement. 23 Our study design did not allow us to determine whether the home tests were accurate as we did not have a concurrent lab measurement of HbA1c. The self-reported lab value could have occurred at any point within the previous three months and was reliant on participant recollection of their most recent value. In addition, test kits were mailed to participants, and we cannot be sure that temperature/storage requirements outlined by the manufacturer were met. Future remote research should consider the above concerns during study design and consider the importance of checking accuracy prior to reliance on the results.
Challenges with technology usage aside, our study participants were able to complete the physical activity assessments. All but one participant could safely perform the timed chair rise task in their home. We chose the five-times chair rise task to take a conservative approach to testing participants without study personnel physically present and to see if this approach was feasible for telehealth interventions. Four participants (19%) took up to three times longer than the fastest participant (5.85 seconds) to complete the task. Future research should compare performance with a laboratory-based collection to assess if performance is impaired in these individuals because of strength deficits or concerns about safety when research personnel are not physically present.
All participants completed surveys either online or by having study personnel read the surveys to them on the phone/Zoom session. Our participants reported minimal levels of depression, pain interference, fatigue and anxiety, similar to those measured by Adams et al 35 in community dwelling older adults pre-COVID-19 pandemic (anxiety: Adams = 49.1 ± 7.2, Bohnert = 48.2 ± 7.4, depression: Adams = 48.3 ± 8.2, Bohnert = 48.9 ± 7.6, and fatigue: Adams = 50.9 ± 8.5, Bohnert = 50.8 ± 8.4). Our study participants had a high level of confidence with activities requiring balance (ABC score: 78.7 ± 22.0). Our findings were similar to those in previous studies of people with diabetes, with neuropathy (78.02 ± 17.01) 36 and without neuropathy (72.08 ± 7.07). 37 The similarity of the survey scores from in-person data collections35-37 gives us confidence in survey results we collected during the telehealth study and suggests our study participants are managing their emotional health well and have balance confidence in their current environment. The UAB Life Space Assessment has separate categories assessing activities occurring in their home and outside their town. One category of responses indicated that the participants were not routinely moving outside their community, suggesting adherence to the guidelines of the stay-at-home orders of the pandemic.
Our study may be limited by selection bias as the participants were already participating in an exercise intervention study; their survey answers could be biased toward an exercise intervention. Participants were from a similar geographic area (St. Louis, Missouri), so our findings may not be generalizable to other regions where technology access may be more or less of a challenge. About 30% of individuals approached to participate in this study declined, which may indicate a disinterest in participating in telehealth-based interventions not captured by the results of the study. Finally, a strength of the study was the variability in the physical tasks (chair rise, daily steps) and survey results. All participants received the same instructions. Therefore, we believe the variability indicates that our study sample had different physical capabilities. These differences showed the ability of a variety of levels of frailty to safely complete the remote research methods sampled in this study.
Conclusions
We found that this group of older adults with type 2 diabetes and peripheral neuropathy would need significant technological assistance (connectivity, device, guidance) to complete a telehealth intervention. Despite technology needs, participants were interested in telehealth interventions, particularly those addressing balance, exercise, and diabetes management. Research methods, including physical assessments, home blood analysis, and surveys, could be completed remotely with assistance from research personnel.
Footnotes
Abbreviations
ABC, activities-specific balance confidence; CCC, concordance correlation coefficient; CI, confidence interval; COVID-19, SARS-CoV-2; HbA1c, hemoglobin A1c; PROMIS, Patient-Reported Outcomes Measurement Information System; REDCap, Research Electronic Data Capture; TAM, technology acceptance model; UAB, University of Alabama at Birmingham.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases [DK107809, F32 DK123916] and the National Center for Medical Rehabilitation Research of the Eunice Kennedy Shriver National Institute of Child Health and Human Development [T32 HD07434].Clinical and Translational Science Award (CTSA) Grant [UL1 TR000448] and Siteman Comprehensive Cancer Center and NCI Cancer Center Support Grant P30 CA091842; the Administration for Community Living, U.S. Department of Health and Human Services 90ARHF0006.
