Abstract
Recent in vitro experiments with patch pumps (PP) Omnipod (OP), Omnipod DASH (OP-D), A6 TouchCare (A6), and Accu-Chek Solo (ACS) have observed periodic fluctuations in the delivered amount of insulin during basal rate and consecutive bolus delivery in some PP, calling for a more systematic characterization of these periodic delivery patterns. Here, it was found that during basal rate delivery of 1 U/h, some devices of OP, OP-D, and A6 showed deviations of up to ±30% from target delivery that consistently repeated every 5 hours, whereas ACS showed no clear periodicity with considerably lower deviations. Similar results were found during consecutive bolus delivery of 1 U, where deviations repeated consistently every five boluses in some devices of OP, OP-D, and A6. However, there was a large variability in the periodic delivery patterns between individual devices of the same PP model. Examining their pumping techniques indicated a connection between the insulin delivery mechanism and observed delivery patterns of the PP. However, the clinical impact of such patterns is unclear.
Introduction
Insulin pumps without visible infusion sets, also referred to as patch pumps (PP), have become increasingly popular among patients with insulin-dependent diabetes due to their discreetness and ease-of-use. 1 In recent in vitro experiments with different PP, their accuracy of insulin delivery during basal rate and bolus infusion was evaluated.2-5 In particular, the following PP were examined: Omnipod (OP) (Insulet Corp, Billerica, MA, USA), Omnipod DASH (OP-D) (Insulet Corp), A6 TouchCare System (A6) (Medtrum Technologies, Inc, Shanghai, China) and Accu-Chek Solo micropump system (ACS) (Roche Diabetes Care GmbH, Mannheim, Germany).
The time courses of insulin delivery at a basal rate of 1 U/h showed, at least in some PP, periodic fluctuations (Figure 1). This has been previously noted; however, further evaluation of this periodic delivery pattern has not been carried out.3,4 A similar periodicity was observed during insulin delivery of 25 successive boluses of 1 U (Figure 2). Furthermore, periodic insulin delivery patterns have already been found with syringe pumps in the 1980s. 6
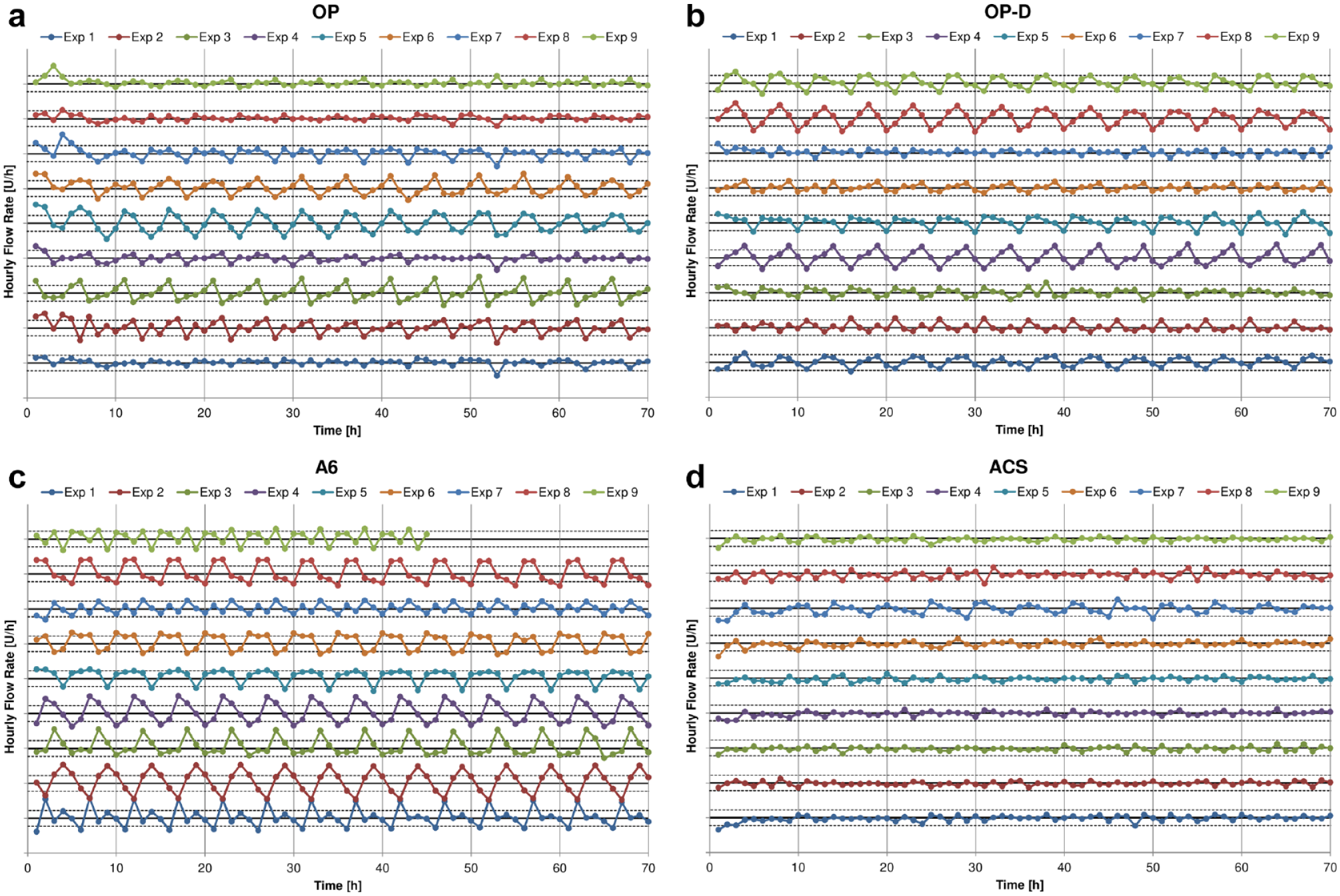
Time course of insulin delivery in non-overlapping 1-hour windows during basal rate delivery with OP (a), OP-D (b), A6 (c), and ACS (d), respectively. The solid black lines indicate the target delivery volume of 1 U/h and the dashed lines above and below this line show the ±15% error limits. In ACS and A6, experiments 1 to 3, 4 to 6, and 7 to 9 were conducted with the same reusable pump base. Experiment 9 with A6 was stopped after 45 hours due to an error notice by the patch pump. Abbreviations: OP, Omnipod; OP-D, Omnipod DASH; ACS, Accu-Chek Solo.
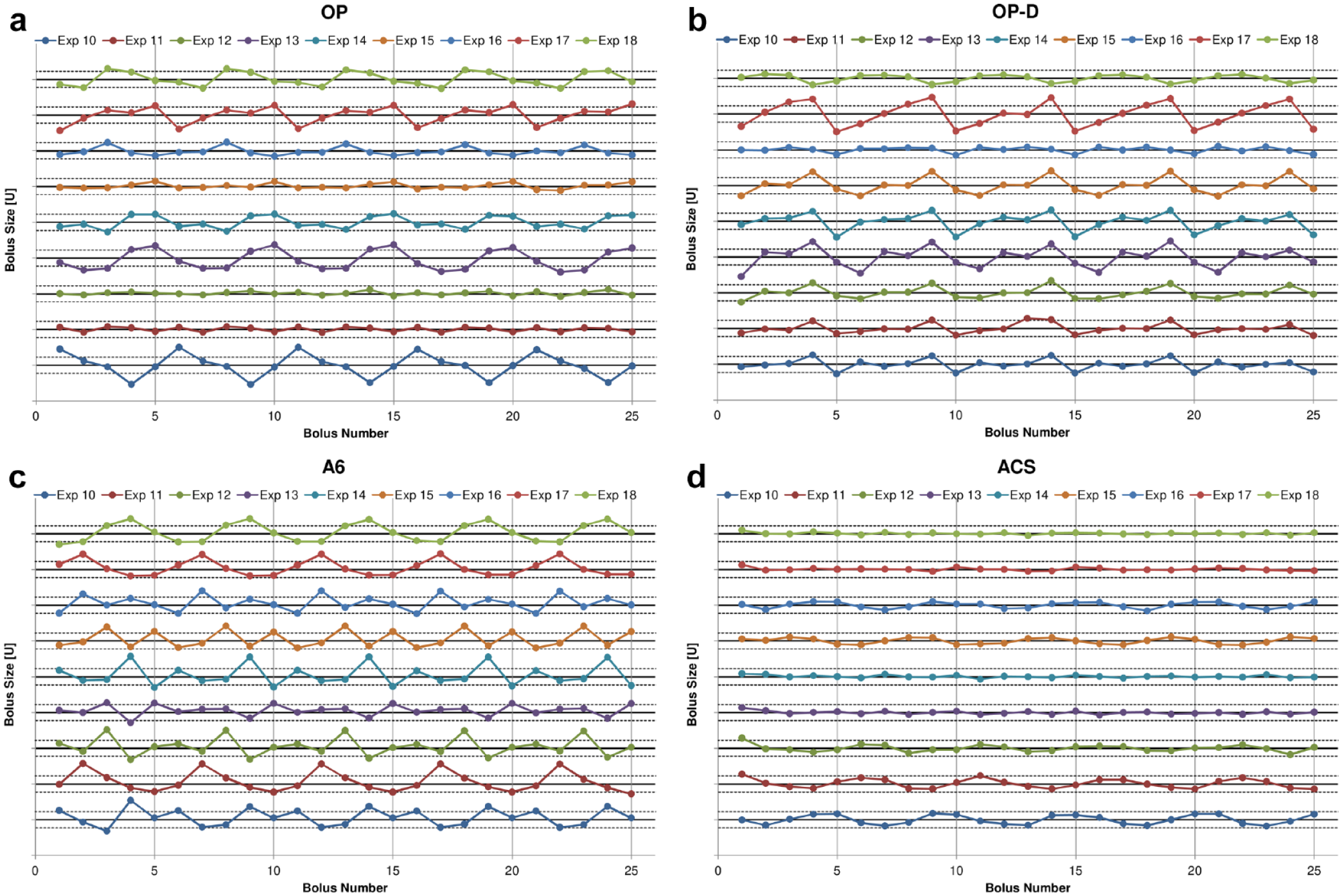
Consecutive bolus delivery of 1 U for nine experiments of OP (a), OP-D (b), A6 (c), and ACS (d), respectively. Please note that the x-axis gives the bolus number and not the time. The solid black lines indicate the target delivery volume of 1 U and the dashed lines above and below this line show the ±15% error limits. In ACS and A6, experiments 10 to 12, 13 to 15, and 16 to 18 were conducted with the same reusable pump base. Abbreviations: OP, Omnipod; OP-D, Omnipod DASH; ACS, Accu-Chek Solo.
This technology report provides a systematic characterization of such periodic delivery patterns and discusses possible causes in relation to the pump technology used in the examined PP.
Experimental Procedures and Characterization of Periodic Delivery Patterns
Each PP model was examined in 18 separate experiments using a microgravimetric measurement system based on the standard IEC 60601-2-24 on the performance of infusion pumps, that was adapted for tubeless insulin pumps. 2 Experiments 1 to 9 tested the basal rate delivery of 1 U/h for 70 hours, and experiments 10 to 18 each examined 25 successive bolus deliveries of 1 U. For OP and OP-D, each experiment was conducted with a new disposable pod device. A6 and ACS consist of a reusable PP base and one or more single-use parts including the insulin reservoir and cannula. Experiments were therefore repeated with new single-use parts, but the same three PP bases were used in all experiments (Figure 3).
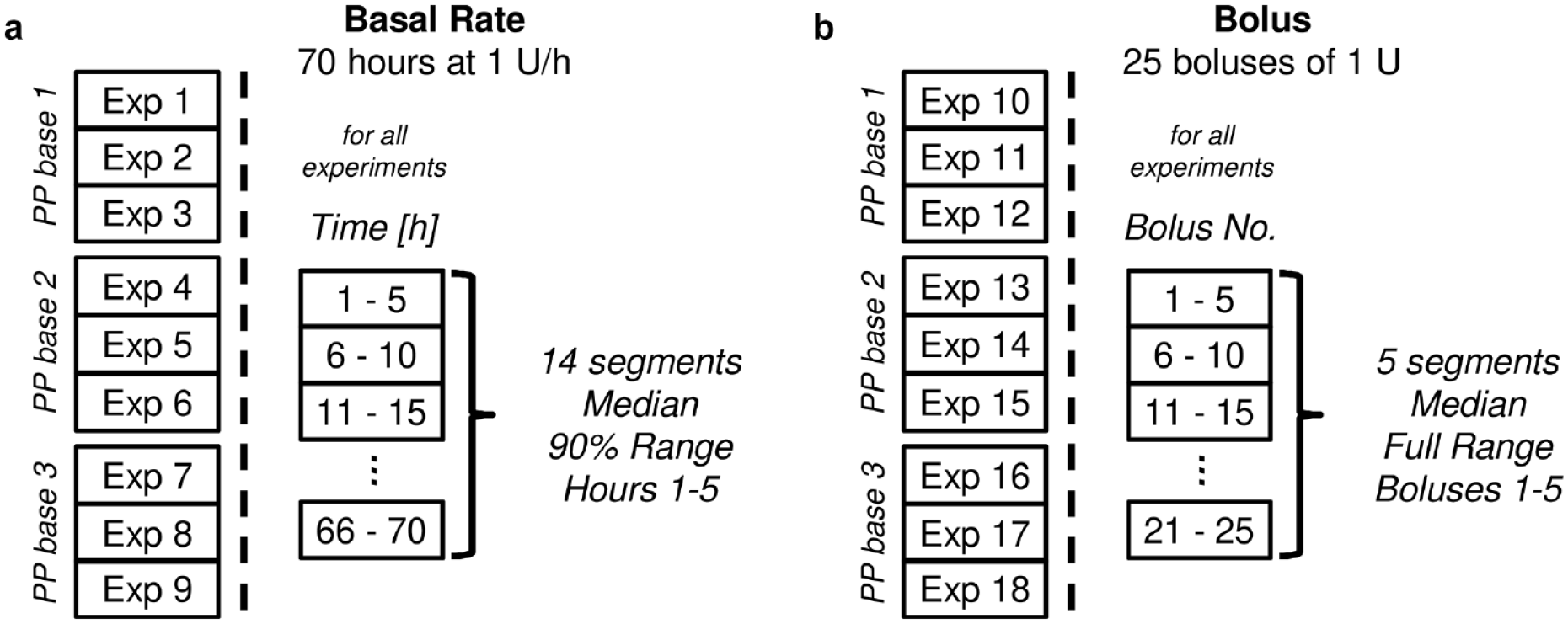
Schematic depiction of the experimental procedures and evaluation of periodic delivery patterns with basal rate (a) and bolus (b) experiments conducted on each patch pump (PP) model. Each experiment was conducted with new single-use parts. The specification of PP bases only applies to experiments with A6 and ACS. Abbreviation: ACS, Accu-Chek Solo.
To identify the fundamental oscillation frequencies in the delivery profiles of the different PP, a frequency domain analysis was carried out for each experiment. Applied to the time course of basal rate delivery in Figure 1, this analysis showed that almost all experiments with OP, OP-D, and A6 showed a fundamental oscillation period of 5 hours and a higher order oscillation with a period of 2.5 hours. The ACS did not show a similarly clear pattern, however, in experiments 5 to 7, the ACS appeared to display a minor periodic variability with a slightly larger fundamental period of approximately 5.3 hours. (Detailed analysis results are provided in the Supplementary Material).
To quantify the amplitude of the periodic basal rate delivery, a characteristic pattern that indicates the typical cyclic delivery behavior from each experiment was extracted. Based on the identified fundamental period of 5 hour, median and 90% range (minimizing the influence of outliers) of the insulin delivery in hours 1 to 5 of the 14 consecutive 5 hour segments (70 hours total duration) were calculated (Figure 3a). The characteristic delivery behavior was then expressed as relative deviation of the target delivery of 1 U/h. An example for its calculation is given in Figure 4.
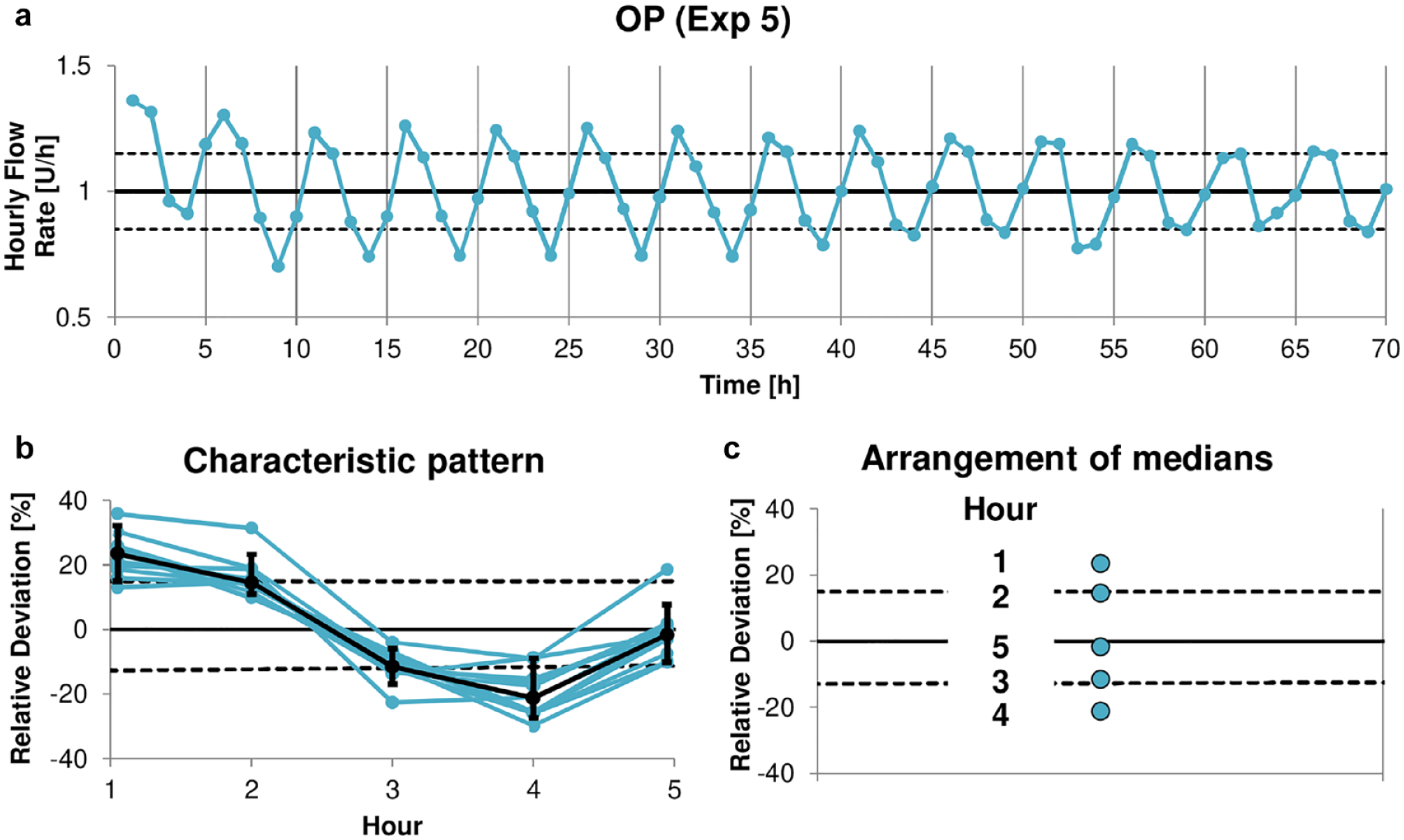
Example calculation of the characteristic insulin delivery pattern from basal rate experiment 5 of OP: (a) The time course of basal rate delivery was divided into 14 consecutive 5-hour segments. The horizontal lines indicate target delivery and ±15% deviation. (b), The 14 segments were stacked (blue) and the characteristic pattern was calculated as median and 90% range (black dots and antennae) in hours 1 to 5 over all segments, (c) The medians in hours 1 to 5 are arranged vertically. Abbreviation: OP, Omnipod.
Applying the frequency domain analysis to the bolus delivery profiles in Figure 2 revealed that almost all experiments with OP, OP-D, and A6 showed a fundamental delivery period that repeated approximately every 5 boluses. For the ACS, a similarly clear pattern was not apparent; however, in some experiments the ACS also displayed a minor periodicity with a slightly larger period that repeated approximately every 5.3 boluses. To characterize the amplitude of this periodicity, a similar procedure as for the basal rate data was used. The characteristic pattern of bolus delivery was formed by five consecutive boluses and calculated from five consecutive segments of five boluses (Figure 3b). Due to the small number of segments (5), the full range of data was used to characterize the variability within each experiment.
A comparison of the delivery amplitudes across all experiments and PP without temporal information is given in Figure 5. The detailed, individual characteristic delivery patterns over time of all experiments, exemplified in Figure 4b, are displayed in the Supplementary Material.
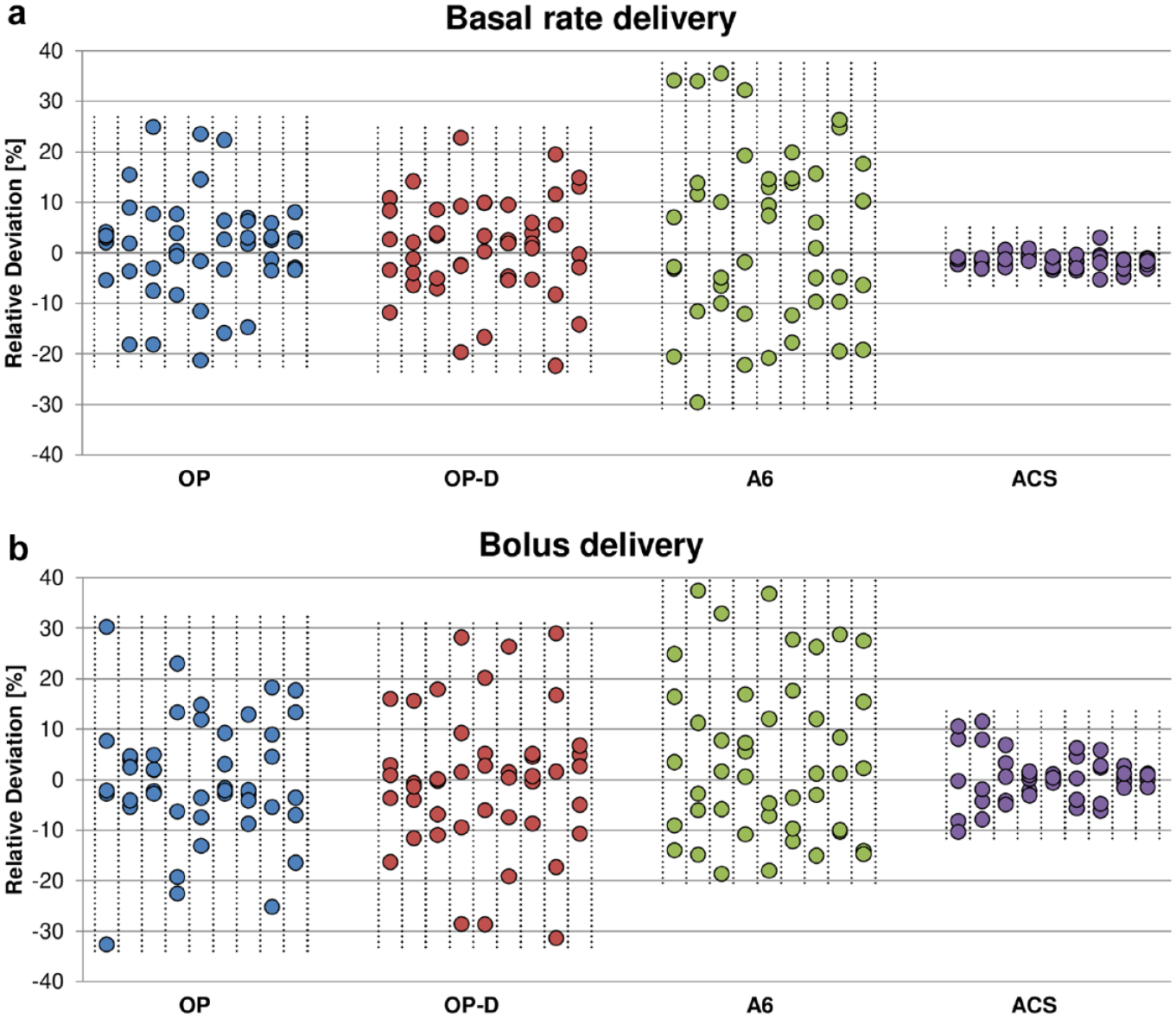
Delivery errors in the characteristic delivery patterns of basal rate (a) and bolus (b) delivery without temporal information. For each experiment (separated with vertical lines), the median delivery error in hours/boluses 1 to 5 are displayed vertically.
The analysis of the variability of the delivery amplitude from the basal rate experiments, presented in Figure 5, shows that the insulin delivery in some experiments has varied between approximately ±20% for OP and OP-D, ±30% for A6, and ±5% for ACS within a 5-hour period. Furthermore, the cyclic delivery patterns within each experiment with OP, OP-D, and A6 were strikingly regular, whereas the results show large variability between experiments, with some devices displaying almost constant delivery. Similar results with regard to regularity within, and variability between experiments, were found in the bolus delivery experiments. However, the bolus delivery error was increased with some experiments showing variability between approximately ±30% for OP and OP-D, -20% and +35% for A6, and ±10% for ACS within five consecutive boluses. The asymmetry of the bolus results of A6 indicates the presence of an average positive bias, which has been reported previously. 4
Discussion on Possible Causes of the Variability in Insulin Delivery in Relation to Patch Pump Technology
In principle, the cyclic delivery patterns observed with at least some PP (like the OP, OP-D, and A6) could be caused by the experimental setup: however, as one other PP, the ACS, displayed a far less pronounced variability despite being examined under the same experimental protocol and setup, this is unlikely.
Further information on the possible root cause of the periodicity in insulin delivery can be obtained by examining the insulin delivery mechanism of the OP (Figure 6). Here, each actuation of the pumping mechanism, that is a one-directional pivot by the hook, rotates the ratchet, causing the plunger to be pushed into the reservoir, delivering 0.05 U. 7 The teeth of the two gears on the ratchet are offset to each other, so that one arm of the hook is always engaged with the teeth of one gear, while the other arm sits on top of a tooth. This means that two actuations, that is pivots of the hook (one left, one right), move the gear one full tooth, thereby delivering 0.1 U. 8 Through inspection of one of the OP devices used in the experiments, it could be counted that the gears on the ratchet have 50 teeth each, which means that a full rotation of the ratchet would deliver 5 U. As a consequence, the ratchet would have completed a full rotation every 5 hours and every five boluses during the basal rate delivery experiments with 1 U/h and consecutive bolus delivery experiments with 1 U, respectively. These rotation periods are in agreement with the observed periods of the characteristic delivery patterns in the OP, which suggests a connection between the measured cyclic insulin delivery patterns and its actuation mechanism.
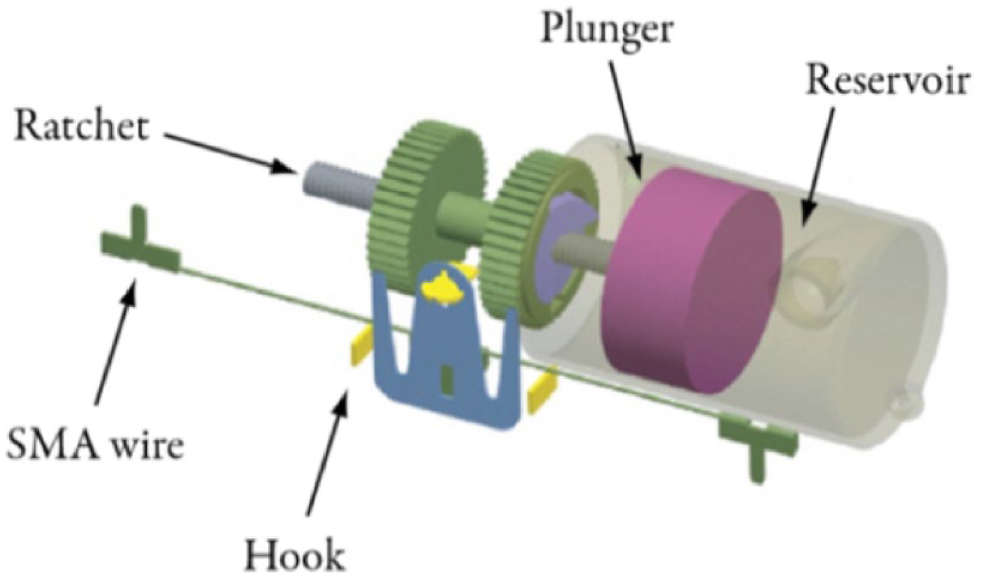
Schematic depiction of the insulin delivery mechanism of the OP (Image was taken from Zisser, 7 which is distributed under the terms of a Creative Commons Attribution Noncommercial 4.0 International License [CC BY-NC]). Abbreviation: OP, Omnipod; SMA, shape-memory alloy.
As for OP-D, a recent article gives no indication that its delivery mechanism has been significantly altered in comparison to the OP, 9 which explains the observed similarities in the delivery patterns.
For the A6, the user manual indicates that the reusable pump base only accommodates electronic parts, while the single-use reservoir patch contains all moving parts including the delivery mechanism. 10 This might explain why the experiments conducted with the same pump base show no similarities in the characteristic delivery patterns. Furthermore, the identical periods found in delivery patters of the OP/OP-D and A6 suggest that there are similarities in the pumping mechanisms of the OP/OP-D and A6. However, to knowledge of the authors, no reliable information on the pumping mechanism of the A6 has been published.
The cause of the observed variability in delivery patterns between individual disposable devices of OP, OP-D, and A6 is unclear. However, it could be possible that variability in the manufacturing process might be a contributing factor. In this context, it should be mentioned that within basal rate and bolus experiments, disposable parts from the same lot were used for all devices.
Compared with OP/OP-D, the ACS uses a substantially different insulin delivery mechanism. Moving parts are contained both in the reusable pump base, which accommodates a step motor, as well as in the disposable reservoir containing a rotating threaded bar that advances the plunger. 11 These differences might explain why the periodic variability of insulin infusion is far less prominent with this PP.
Conclusions
This study observed periodic insulin delivery patterns in different PP to a varying extent. To our understanding, these patterns are not caused by the experimental setup or protocol but can be related to the pumping mechanisms of the PP.
The in vitro accuracy of patch pumps, especially OP, has been examined previously and a generally decreased accuracy was found in comparison to durable pumps.3,12-15 However, a similarly detailed assessment of the time course of insulin delivery has not been carried out. A limitation of the present study is that the tested scenarios of constant basal delivery and consecutive bolus delivery are not particularly realistic. Further studies should thus investigate to what extend the periodic delivery patterns are present at other basal rates and how they are affected by intermittent bolus delivery.
The clinical impact of the observed cyclic delivery patterns is unclear. However, an intentional temporal adaptation in basal rate of ±20-30% is certainly within the range of a therapeutic decision of a pump user, which could indicate an effect on glucose levels from the same unintentional delivery pattern. Nevertheless, it should be mentioned that clinical studies investigating the effect of switching from a durable pump to OP/OP-D found very similar or slightly improved glycemic outcomes.16-19 Additional work should thus examine the clinical impact of the observed delivery patterns.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221091843 – Supplemental material for Patch Pumps: Periodic Insulin Delivery Patterns
Supplemental material, sj-docx-1-dst-10.1177_19322968221091843 for Patch Pumps: Periodic Insulin Delivery Patterns by Manuel Eichenlaub, Ralph Ziegler, Lutz Heinemann, Delia Waldenmaier, Ulrike Kamecke, Cornelia Haug and Guido Freckmann in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank Stefan Pleus and Jochen Mende for their support in writing the manuscript and analyzing the data, as well as the independent testing laboratory that carried out the experiments.
Abbreviations
PP, patch pump; OP, Omnipod; OP-D, Omnipod DASH; A6, Medtrum A6 TouchCare; ACS, Accu-Chek Solo.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RZ has received speakers’ honoraria and/or served on advisory boards from/of Abbott, Menarini Diagnostics, Dexcom, Glooko, Lilly, MySugr, Novo Nordisk and Roche Diabetes Care. LH is a consultant for a number of companies that are developing novel diagnostic and therapeutic options for diabetes treatment. He is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany. GF is general manager and medical director of the IfDT (Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies on the evaluation of BG meters, with CGM systems and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IfDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Berlin Chemie, Beurer, BOYDsense, CRF Health, Dexcom, i-SENS, Lilly, Metronom, MySugr, Novo Nordisk, Pharmasens, Roche, Sanofi, Sensile, Terumo, and Ypsomed. UK is an advisor to the IfDT and other diabetes technology companies. ME, DW, and CH are employees of the IfDT.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This evaluation and scientific writing were funded by Roche Diabetes Care GmbH, Germany.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
