Abstract
Focus on take-back of waste products is currently enjoying increased importance, as attention on environmental sustainability and circular economy grows. Single-Use or Disposable Medical Devices (SUDs), which in homecare settings often end up in landfills or incineration, are currently subject to attention, regarding the potential to slow the flow of waste and seek new value creation possibilities. Via a descriptive single-case study of the “ReturpenTM” initiative—a collaborative take-back initiative launched in three municipalities in Denmark—characteristics are elicited, of the planning, launch, and implementation, of the first 6-month pilot of the ReturpenTM initiative. ReturpenTM is a collaborative partnership of 15 public and private organizations and is adopting an end-to-end approach for its development and execution, including numerous professional workstreams. The pilot of the ReturpenTM achieved participation of 66 of the existing 73 pharmacies in 3 municipalities (90% participation rate), and an overall return rate of 13% for the used insulin pens, despite the limitations caused by the covid-19 pandemic. The return rates ranged from 10% to 15% in the 3 municipalities, and overall, the second quarter recovery (15%) was higher than the first quarter (11%). ReturpenTM demonstrates how a workstream-based approach can provide a practical framework for the development and implementation of SUD take-back in a homecare setting. The case describes how the pharmaceutical industry is taking proactive measures to contribute to a more circular economy for disposable medical devices, including the infrastructure and ecosystem necessary to ensure a closed-loop system for medical devices.
Introduction
Take-back for waste products is assuming increasing significance, because of the growing waste problem and recognition of the unsustainable nature of the current linear (take-make-dispose) economy.1,2 The healthcare industry is no exception; during the past 30 years, the industry has become increasingly contributory to Single-Use or Disposable Medical Devices (SUDs). 3 Disposable insulin pens (injection devices) are one such SUD, used by diabetes patients. Insulin pens can be reusable (durable) or prefilled (disposable). 4 With proper care, reusable pens can be used for several years, 5 though will still need to be considered for take-back at the end of its use. Used insulin pens are often improperly disposed of, ending up in general household waste.6,7 Despite their many benefits, pens are mechanically more complex than insulin syringes and not designed for disassembly.6,8 Although most insulin pens are proprietary devices, the principal material fractions are plastic, glass (from the cartridge), metal (e.g. spring), and rubber. 9 Furthermore, the complexity and risks in handling used pen devices may additionally come from the needles (potentially contaminated with blood and tissue) and unused left-over active pharmaceutical ingredient (API).
The overarching question this paper seeks to address is how to design and implement a take-back system for SUDs? The paper draws on insights of the first 6-months of ReturpenTM—a collaborative take-back initiative for disposable insulin pens, launched on World Diabetes Day (2020) in three municipalities in Denmark. A descriptive single-case study of ReturpenTM initiative was carried out allowing for a retrospective study of the activities and considerations throughout the planning, launch, and implementation of the first 6-months of the pilot, up to May 2021. During this period, data collection from various sources, including direct observations (e.g. via periodic project team meetings), examination of available documents, and related project reports was performed. To maintain the rigor of the research study, validation, and verification of the study description was performed by seeking feedback on the facts covered from the project team. The subsequent sections of the paper cover insights, regarding (a) why the company initiated the ReturpenTM program, (b) approaches for planning and implementing take-back, focused in this case on diabetes patients in a homecare setting, and (c) the pilot’s results, in terms of the used pen collections in the pilot geographies. Furthermore, a take-back framework is proposed, based on learnings from the case study.
The Driving Forces Behind ReturpenTM Program
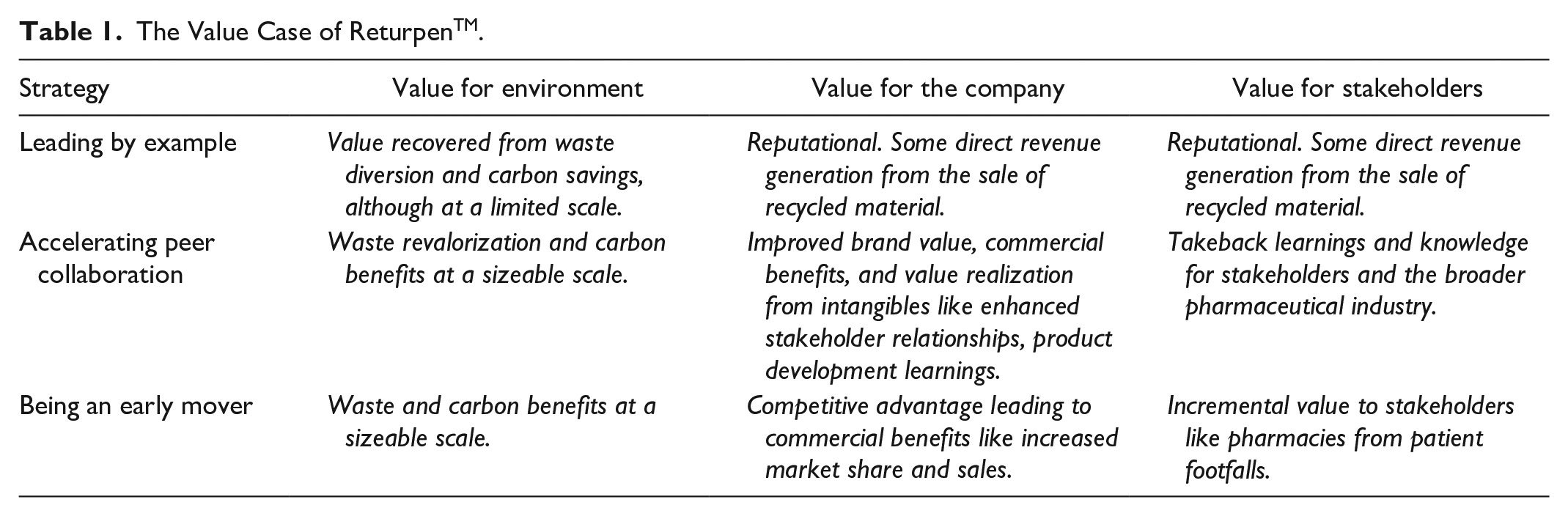
The company behind ReturpenTM is a market leader in the pharma industry with a history of innovation. It wants to pioneer an industry solution for transforming used insulin pens, focusing on avoiding plastic waste sent to landfills or incineration. Plastic is a significant material in the medical devices produced by the company, which realizes that the current linear take-make-dispose model does not maximize the value of the waste materials. Although currently challenging, an ultimate goal is to be able to reuse the post-consumer recycled materials in the company’s own production. The company recognizes the potential reputational value from being an early mover and being recognized as a “green leader.” Table 1 elaborates the potential value case envisaged for itself, for the environment and for various stakeholders.
The Value Case of ReturpenTM.
Approaches for Planning and Implementing Take-Back
ReturpenTM aimed to take an end-to-end approach (users to the recycling usage market), which required different competencies. Therefore nine workstreams were established for ReturpenTM, as described in the following subsections.
Project Scoping
ReturpenTM started as a pilot in three municipalities in Denmark (Aarhus, Copenhagen, and Kolding) to understand the complexities, develop a proof-of-concept and gain critical learnings for scale-up. The team explored various collection models, for example, pharmacy drop-off, mail-back, and the possibility of adapting existing models, similar to the deposit-refund schemes for bottles and cans or battery collection routines found in Denmark and other countries. To assess these models, the team identified certain design principles, through external consultations. These included: scalability; patient-centric behavioral considerations; low cost and simplicity; leveraging existing infrastructure; logistical setup and collaboration; and design for lowering environmental footprint, trending toward zero environmental impact. Based on these principles a pharmacy drop-off over the counter models was chosen as the front end solution for the Danish pilot. With this, the insulin pen users receive a pen collection bag as part of their purchase of insulin pens at the pharmacy. Consequently, next time they visit the pharmacy store, they would drop off the collected items, while purchasing new insulin pens. The collected pens are subsequently aggregated in drop-off containers located in the back-office of the pharmacy, ready for pick-up by the distributors/ wholesalers.
Regulatory Framework Conditions
This workstream examined current disposal practices for used pens. It mapped the regulations and permits to operate the take-back program and the external stakeholders in regulatory bodies, institutions, and policymakers (stakeholders listed in Table 2).
External Stakeholders From Regulatory Perspective.

The peculiarities of the different parts of a pen device (i.e. potentially contaminated needles, unused API, and the end-of-use pen device) may have implications on how used pens are classified and how they can be collected, stored, transported, and treated. Other environmental or pharma-specific legislation may be already in place or emerging that may influence the take-back model design, for example, Extended Producer Responsibility (EPR) or regulation around direct communication between pharma companies and patients.
Discussions with authorities confirmed that the used pens would be handled as “clinical risk” waste, to mitigate any real or perceived contamination risks arising from the needles. This classification meant working with logistics partners for compliance on transportation and storage and with recycling partners for sterilization, before further treatment or processing. The workstream focused on engaging with local municipalities and national authorities, including the Danish Environmental Protection Agency and the Ministry of Environment, to obtain approval.
Patient and User-Experience Design
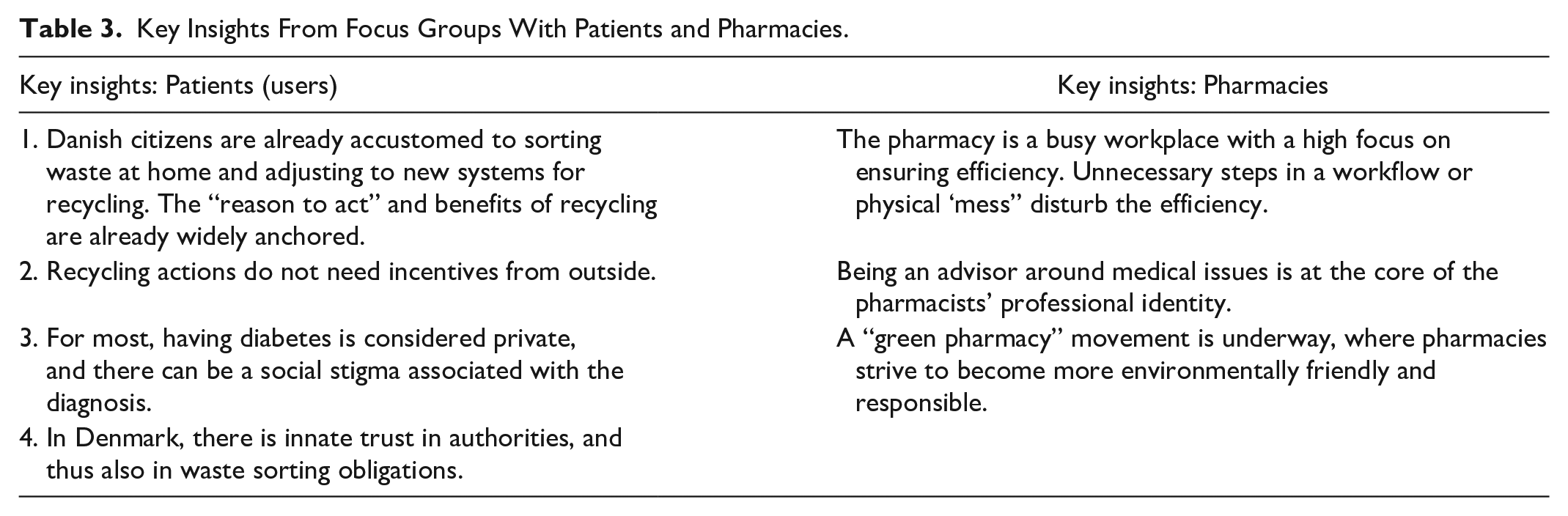
Designing a (insulin pen) user-centric take-back solution is a priority for the workstream. Through qualitative research and knowledge built over many years, the workstream identified the target group for ReturpenTM. They are people with Type 2 diabetes, typically 45+ year-olds, under daily treatment through injections of insulin, in regular contact with pharmacy and Health Care Professionals, visiting pharmacies at least every 3 months. A key stakeholder that the users would interface with, is the pharmacy staff, who would inform the users about the ReturpenTM pilot and receive the used pens, once collected by these users. The workstream conducted focus group studies with these stakeholders (a few insights elaborated in Table 3), which revealed that the target group is already accustomed to the idea of sorting other wastes at home and the benefits of recycling are already anchored, therefore not requiring incentivization.
Key Insights From Focus Groups With Patients and Pharmacies.
Communication and Marketing
This workstream put in place a communication and engagement plan, which included identifying where users want to learn about take-back, which tools and media to use, and where (within pharmacies) would users like to hand in their pen devices. The working group involved key stakeholders in capturing the nuances and sensitivities in communicating on diabetes. It came up with the concept—“too good to throw away.” The core story explained how other products could be made from used pens and urged users to hand them in at the pharmacy for recycling. Additionally, it came up with a visual identity (Image 1) that was simple yet bold, explaining the life cycle of an injector pen.

Visual identity for ReturpenTM.
Back-End (Recycling and End-Markets)
The significant goals under the back-end workstream were to identify recycling solutions in terms of recycling technology and infrastructure, in line with the broader take-back strategy, and find end markets for the recycled materials. Before initiating the take-back program, the company had experience with developing a solution for treating the production site waste. The in-house developed solution could crush the product and sort the material fractions, based on different techniques. For example, it uses density-based separation for the plastic- polypropylene fraction. Putting the rest of the fractions onto a drum magnet can help sort the metal fractions. Furthermore, eddy current magnets sort out the springs. Experience with such machine helped to provide a direction for a possible technical solution for both the production site waste and used pens.
A significant addition is sterilization of the used pens to avoid contamination risks. Having harvested and processed learnings from this, the team scouted for a recycling partner and initiated discussions, in parallel, with design firms to find innovative solutions to exploit and use the recovered plastics and glass fractions. Currently, production scrap has successfully been re-purposed in making demonstration products such as office chairs and lamps, thus demonstrating an end-to-end value chain potential for SUDs and hence also the potential for a take-back program (see Image 2).
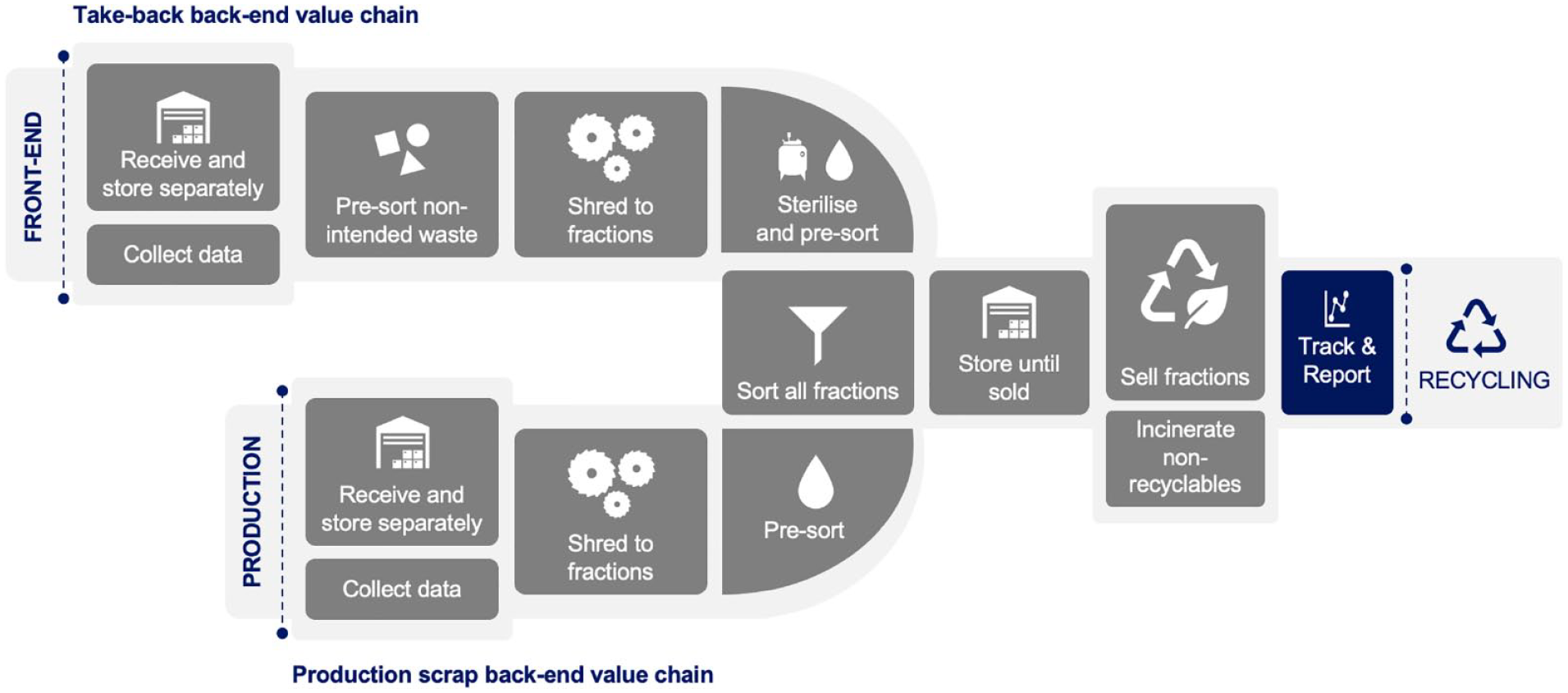
Back-end in ReturpenTM.
Logistics and Operations
The logistics solutions needed to be lean and utilize the existing setup to the greatest extent possible. Partnerships with current wholesalers and logistics partners effectively transported containers with collected pen devices to the warehouse. The workstream conducted risk assessments, ensured compliance with transport and storage regulations like ADR, 10 and trained staff handling logistics.
Partnerships and Collaboration
ReturpenTM is a collaboration of 15 partners across the public and private sectors, including: the Danish state, at the municipal and regional levels; distributors and wholesalers; trade associations; the pharmacy association; and diabetes and patient associations. A shared vision of diverting insulin pens from landfills or incineration was instrumental in bringing together the diverse stakeholders.
Other Workstreams
Besides the above, ReturpenTM had workstreams on program management and constructing the business model, looking into the financial and environmental cases by the use of Life Cycle Assessment (LCA). Underneath this stream the impact tracking and KPI has been consolidated to achieve visibility and subsequently ensure data are transparent and presented to management and stakeholders.
Achievements
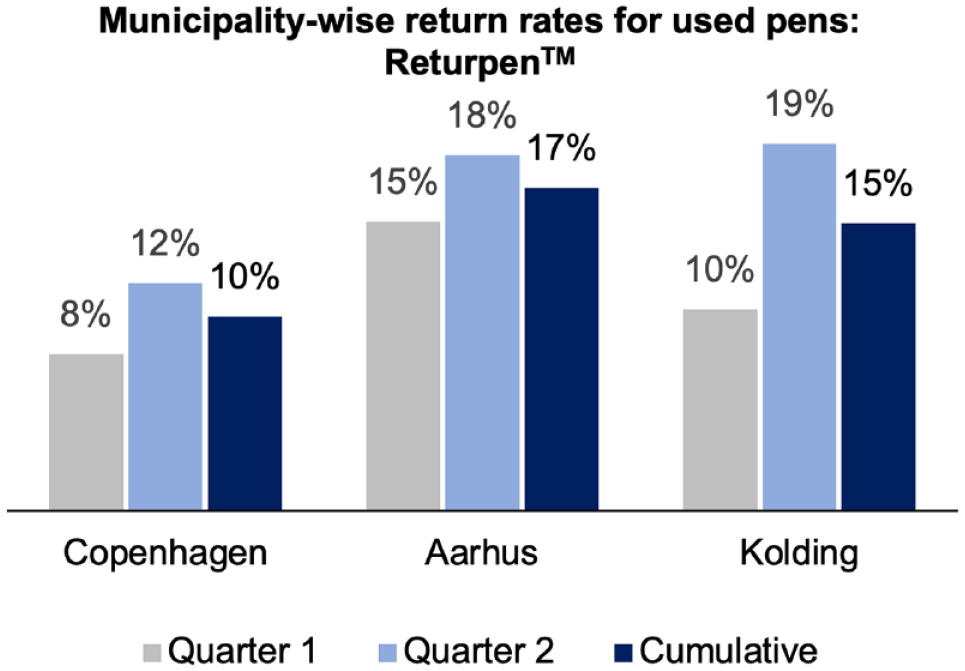
In the first 6 months that the ReturpenTM pilot ran, 66 of the existing 73 pharmacies in the three municipalities participated—37 in Copenhagen, 21 in Aarhus, and 8 in Kolding. Together they helped to collect about 13% of the insulin pens. The impact of the Covid-19 pandemic, marked by lockdown and consequent restrictions in movement ought to be considered, when reflecting on the level of success of this pilot. As seen in Figures 1 and 2, the return rates increased in Q2, indicating that behavior change takes time.
Used pens collected during the pilot phase for ReturpenTM.
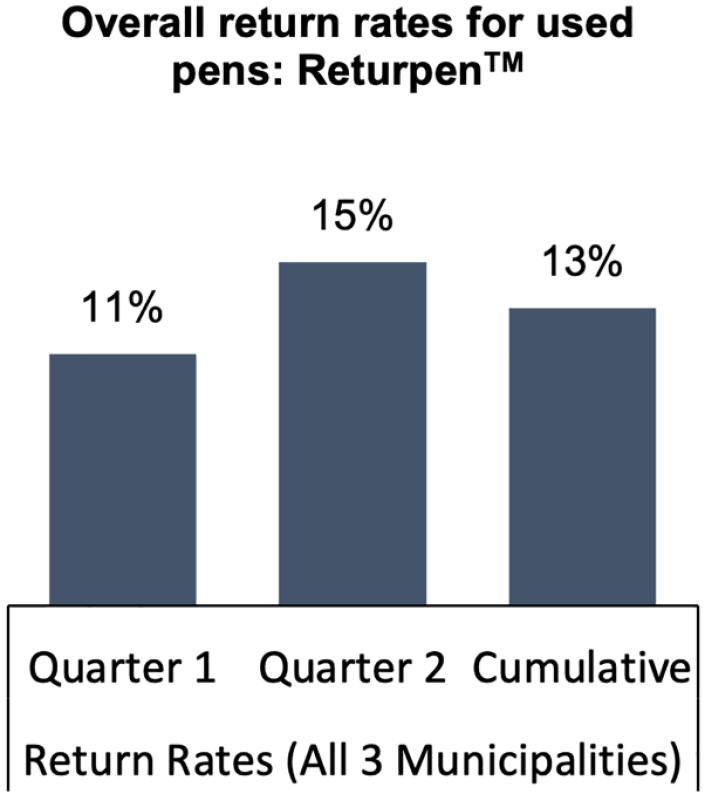
Overall return rates from all municipalities: ReturpenTM.
ReturpenTM also reinforced the hypothesis that the presence of infectious waste is a barrier for recycling SUDs (hygienic obsolescence).
Towards a Take-Back Framework
The ReturpenTM pilot initiative, has fulfilled the overarching objective to establish a reverse value chain for used insulin pens. The value chain comprises of various activities starting with collection through pharmacy drop-off, transportation and storage (reverse logistics by the distributors), and finally the recovery process—involving pre-treatment, shredding, sterilization and recycling of the major material fractions, ready for use as materials for new (non-medical) products. The take-back model for ReturpenTM is summarized in Image 3.
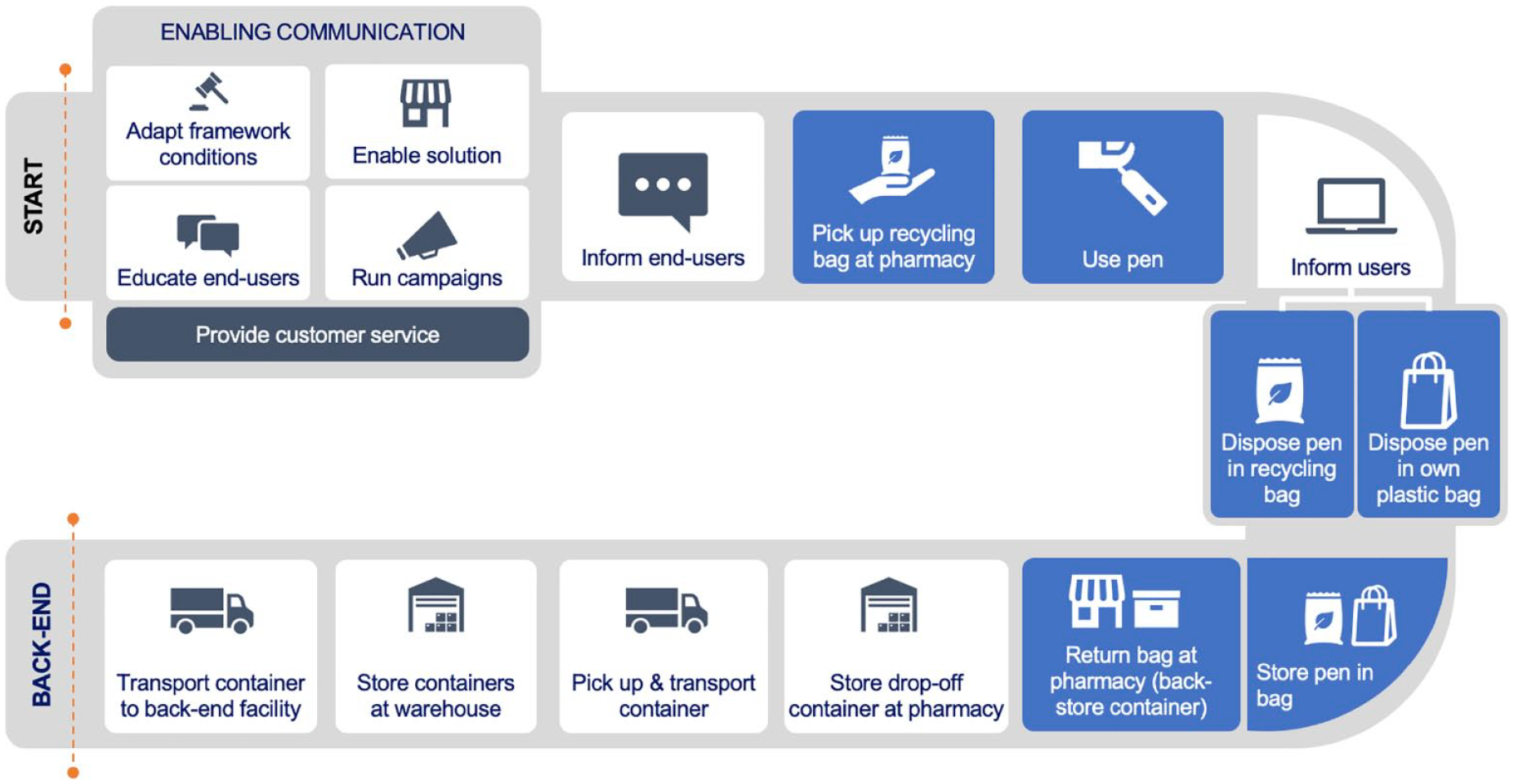
ReturpenTM take-back model.
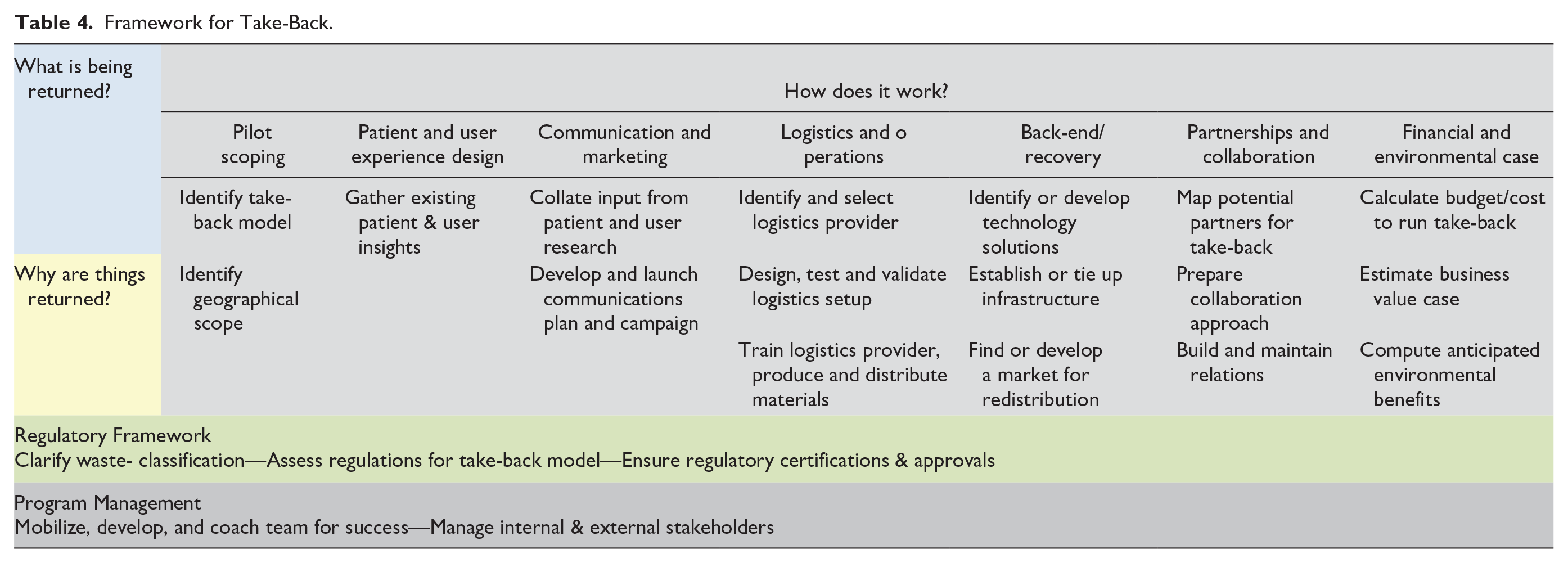
ReturpenTM provides insights that help further elaborate the De Brito and Dekker 11 model for Reverse Logistics. Especially when the “How” element is examined, the initiative demonstrated how multiple competencies are needed to design and implement a take-back system. A workstream-based approach may be valuable in covering various aspects of take-back design and implementation. In Table 4, an initial framework is proposed for designing and implementing a take-back program based on the insights from ReturpenTM. This framework will be further elaborated over coming research iterations and on the inclusion of other case studies and theoretical insights.
Framework for Take-Back.
Conclusions and Perspectives
This paper aimed to explore how businesses can design and implement take-back for SUDs. ReturpenTM demonstrates how a workstream-based approach can provide a practical framework for take-back. It highlights the need to understand the complexities and burdens of patients living with diabetes and design user-centric solutions. The initiative identifies critical design principles for take-back and reinforces the importance of collaboration based on a shared environmental vision. An initial framework for take-back design, is proposed based on DeBrito’s framework, as applied to the ReturpenTM pilot case.
This study did not cover certain aspects of take-back, such as performance indicators, especially environmental and financial considerations, or user feedback. There could also be possibilities of using other collection models and end-of-life options such as reprocessing of SUDs, which will be further explored in pending research iterations.
Lead players in the pharmaceutical industry are taking proactive measures to contribute to a more circular economy for disposable medical devices. ReturpenTM is a lighthouse for practitioners aiming to implement take-back for SUDs. Following this pilot, a nationwide introduction is currently being investigated along with several pilot programs in other countries.
Footnotes
Abbreviations
ADR, International Carriage of Dangerous Goods by Road; API, Active Pharmaceutical Ingredient; EPR, Extended Producer Responsibility; LCA, Life Cycle Assessment; ReturpenTM, Program name for the return of used insulin pens in Denmark; SUD, Single-Use Medical Device.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
