Abstract
Background:
Our objective was to determine the effect of therapeutic concentrations of N-acetylcysteine, following intravenous infusion, on the measurement of blood glucose using a Roche Diagnostics glucose dehydrogenase-linked glucose meter compared to hospital laboratory methods.
Methods:
N-acetylcysteine was added to aliquots of blood, with glucose promptly measured by the glucose meter, blood gas analyzer (glucose oxidase comparative method) and following centrifugation, plasma glucose measured with a hexokinase spectrophotometric comparative method. Glucose results were evaluated with linear regression and Bland Altman plots.
Results:
In the presence of NAC, at concentrations greater than 5 mg/dL (0.31 mmol/L), positively biased glucose meter results were compared to the clinical laboratory results. Multivariate linear regression revealed that NAC-mediated meter results are influenced by NAC and glucose concentrations.
Conclusions:
The addition of therapeutic concentrations of NAC to blood produces statistically significant positive biases when measured with the glucose dehydrogenase linked glucose meter device.
Introduction
Accurate glucose measurement is particularly susceptible to pre-analytic and analytic interference conferred by the specificity of enzymes and coupled assay designs that link to spectrophotometric or potentiometric detectors. 1 Risks of misinterpretation of glucose test results for patients have been reported for icodextrin, maltose, ascorbic acid, oxygen, hydrogen peroxide, acetaminophen, and hematocrit.1-3 It is noteworthy that many of these interfering compounds are oxidizing and reducing agents.
On September 28, 2020, Roche Canada released on an urgent medical device correction indicating that N-acetylcysteine (NAC) is a newly identified substance capable of interfering with a glucose dehydrogenase linked glucose meter resulting in erroneously elevated glucose results. 4 Specifically, it was noted “Intravenous administration of N-acetylcysteine which results in blood concentrations >5 mg/dL (307 µmol/L) will cause overestimation of blood glucose results. Do not use during intravenous infusion of N-acetylcysteine.” 4
NAC is a frequently used antidote for acetaminophen poisoning. Acetaminophen is an analgesic drug used to treat pain and fever and is found in more than 200 over-the-counter and prescription medicines. 5 According to Health Canada, approximately 4500 hospitalizations occur each year due to acetaminophen overdose. 6 Clinical symptoms of acetaminophen induced hepatoxicity, including hypoglycemia, are dependent upon the time of ingestion as well as the quantity and formulation of acetaminophen ingested. 5 In clinical pharmacies, NAC is mixed with either a 5% glucose solution or a 0.9% sodium chloride solution, to prepare 3 separate intravenous infusions for acetaminophen overdose. 7 The first infusion comprises an initial loading dose of 150 mg/kg body weight, infused over 1 hour. The second infusion of 50 mg/kg is administered over the next 4 hours and the final infusion of 100 mg/kg over the following 16 hours. This triple intravenous treatment regimen has been reported to result in peak plasma NAC concentrations of 30 to 90 mg/dL (1.84 to 5.50 mmol/L) shortly after the initiation of therapy.7,8 NAC concentrations are reported to rapidly decline and be maintained between 1.1 and 9.0 mg/dL (0.07-0.55 mmol/L) approximately 12 hours after initiation of therapy.7,8 Based on the pharmacokinetics of intravenous NAC therapy, NAC concentrations used to evaluate interference with glucose measurements in this study were chosen to mimic therapeutically attainable plasma concentrations (2.54 to 40.7 mg/dL; 0.16 to 2.5 mmol/L). It is also interesting to note that recent reports have described repetitive errors in NAC solution preparation and variation of >50% between measured and anticipated concentrations of NAC infusions. 9 The objective of this study was to determine the effect of NAC concentrations, achieved following IV infusion for the treatment of acetaminophen overdose, on the measurement of glucose using a Roche Diagnostics glucose dehydrogenase linked glucose meter in comparison to comparative methods in a hospital laboratory.
Methods
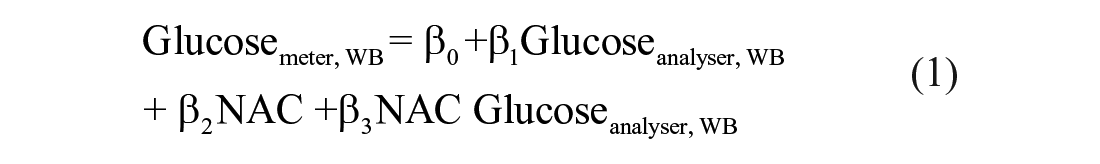
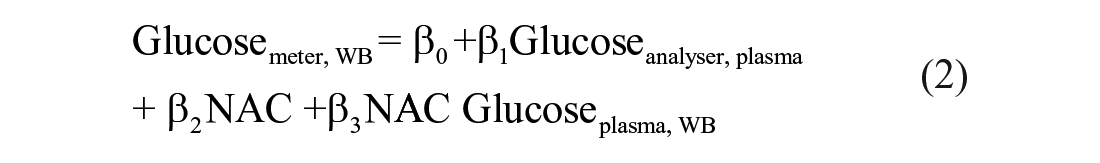
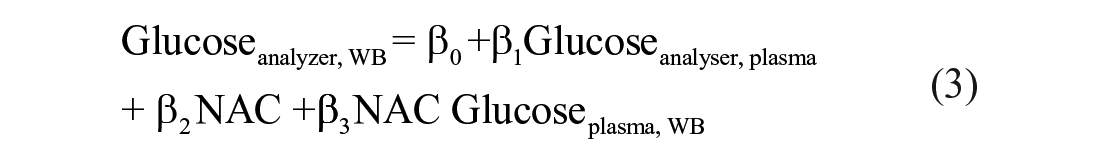
The regression coefficients with 95% confidence intervals and statistical significance for the coefficients are listed in Table 1.
Determination of Partial Regression Coefficients by Linear Regression Models by Ordinary Least Squares for Equations (1) to (3).
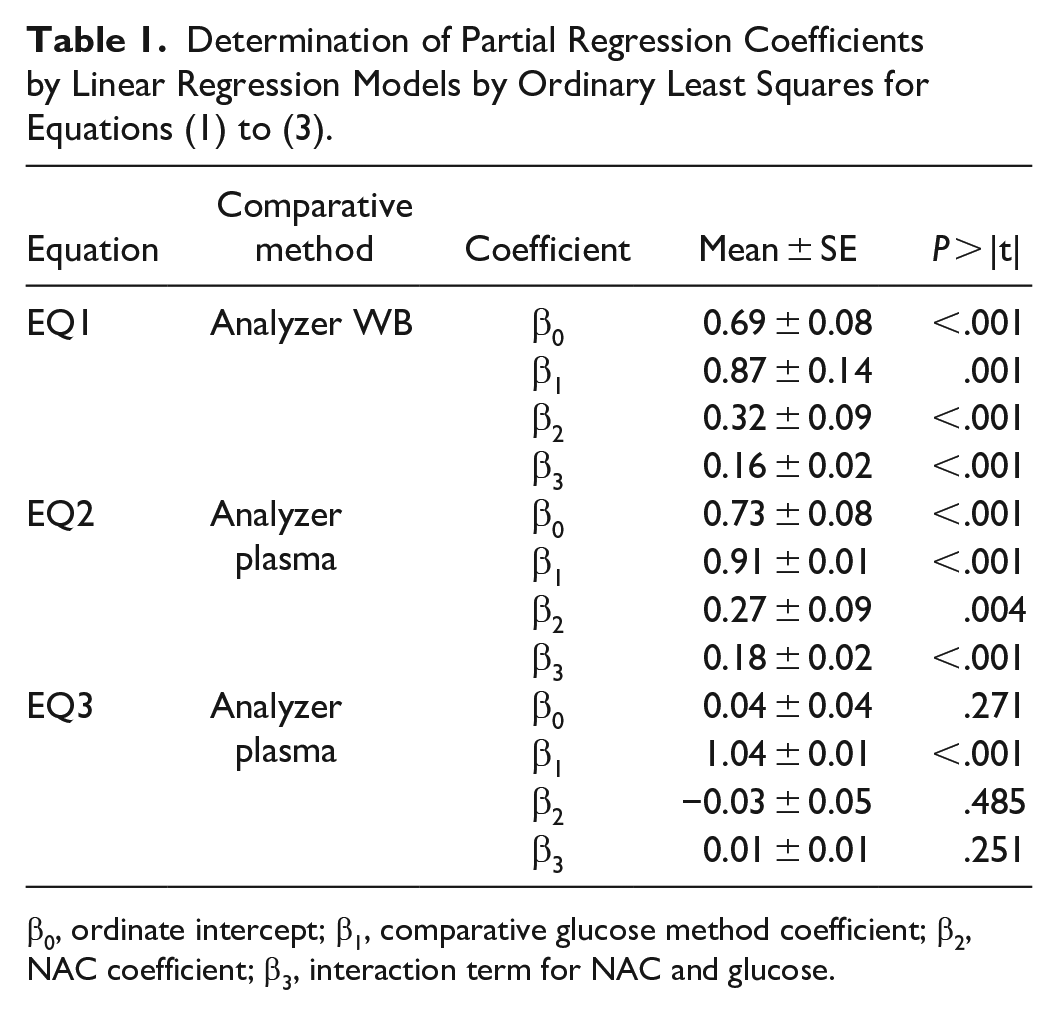
β0, ordinate intercept; β1, comparative glucose method coefficient; β2, NAC coefficient; β3, interaction term for NAC and glucose.
Results
The accuracy of whole blood glucose measurement with the glucose meter in the presence and absence of NAC was compared with the Radiometer ABL 837 whole blood electrode glucose oxidase and Roche Cobas 502 plasma hexokinase glucose methods. Figure 1 illustrates the positive bias influence of NAC on glucose meter performance relative to both the blood gas analyzer glucose method (panel A) and the plasma glucose method (panel B). The open circles indicate glucose results obtained in the presence of different concentrations of NAC (
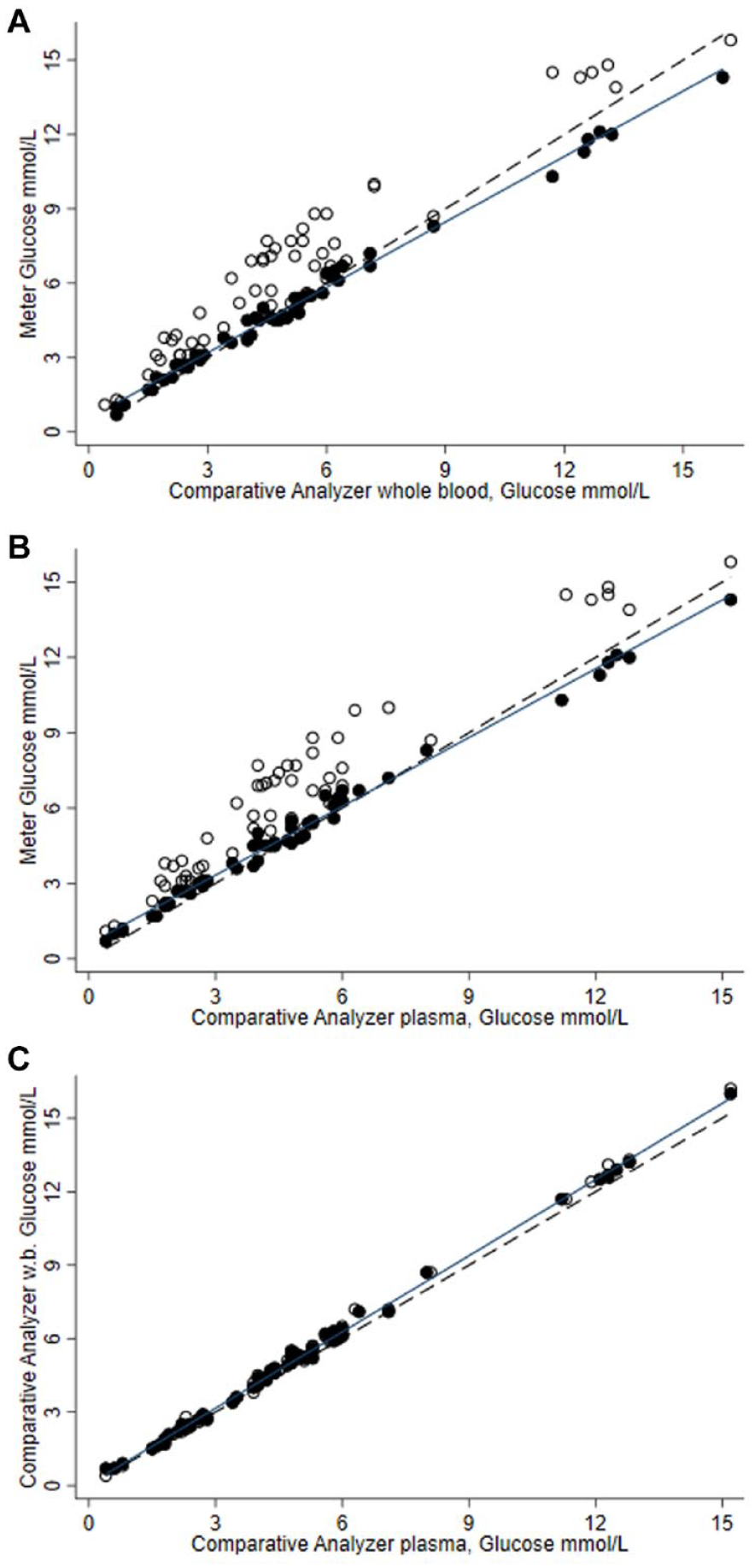
Comparison of glucose method results in the presence of NAC (
The mean bias and 95% confidence intervals determined in the absence of NAC and over the range of glucose concentrations evaluated, are depicted in the Bland Altman graphs in Figure 2. NAC dependent biases (open circles) were overlaid on the Bland Altman graphs to illustrate the extent of bias and variation observed. In the presence of NAC, positive biases for glucose results were obtained with the glucose meter that clearly exceeded the 95% confidence intervals of the control samples (panels A and B). In contrast, NAC-related bias was not observed with glucose results obtained when the blood gas analyzer and chemistry analyzer were compared (panel C).
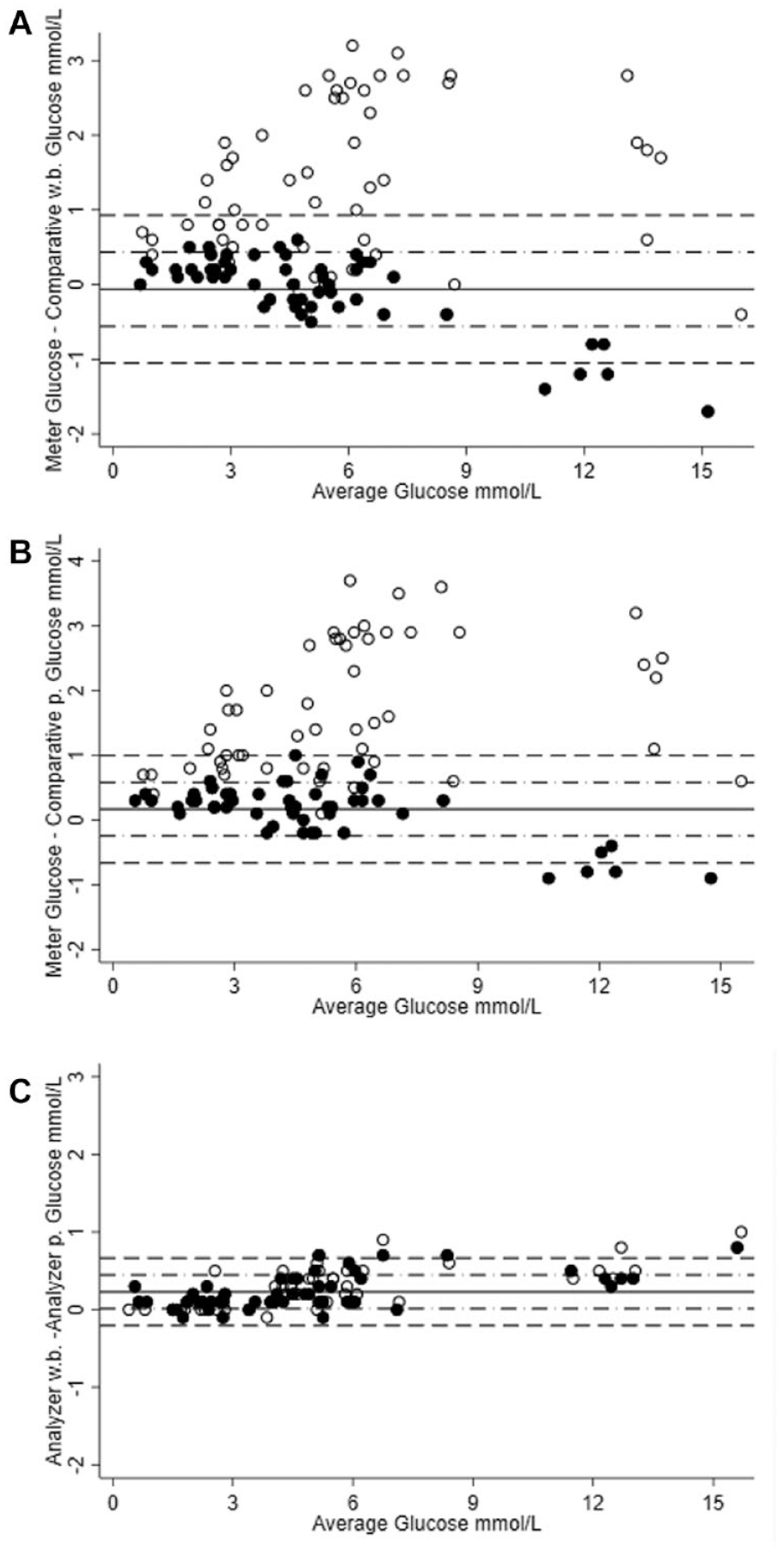
Bland-Altman comparison of glucose method results in the control samples without NAC added (
Multiple linear regression analyses of the models represented by equations (1) to (3) were performed with the data to assess the influence of NAC concentration on glucose meter bias and if this bias was statistically significant, Table 1. The glucose meter demonstrated evidence that glucose bias was statistically significant and predicted by NAC concentration (β2 = 0.32;
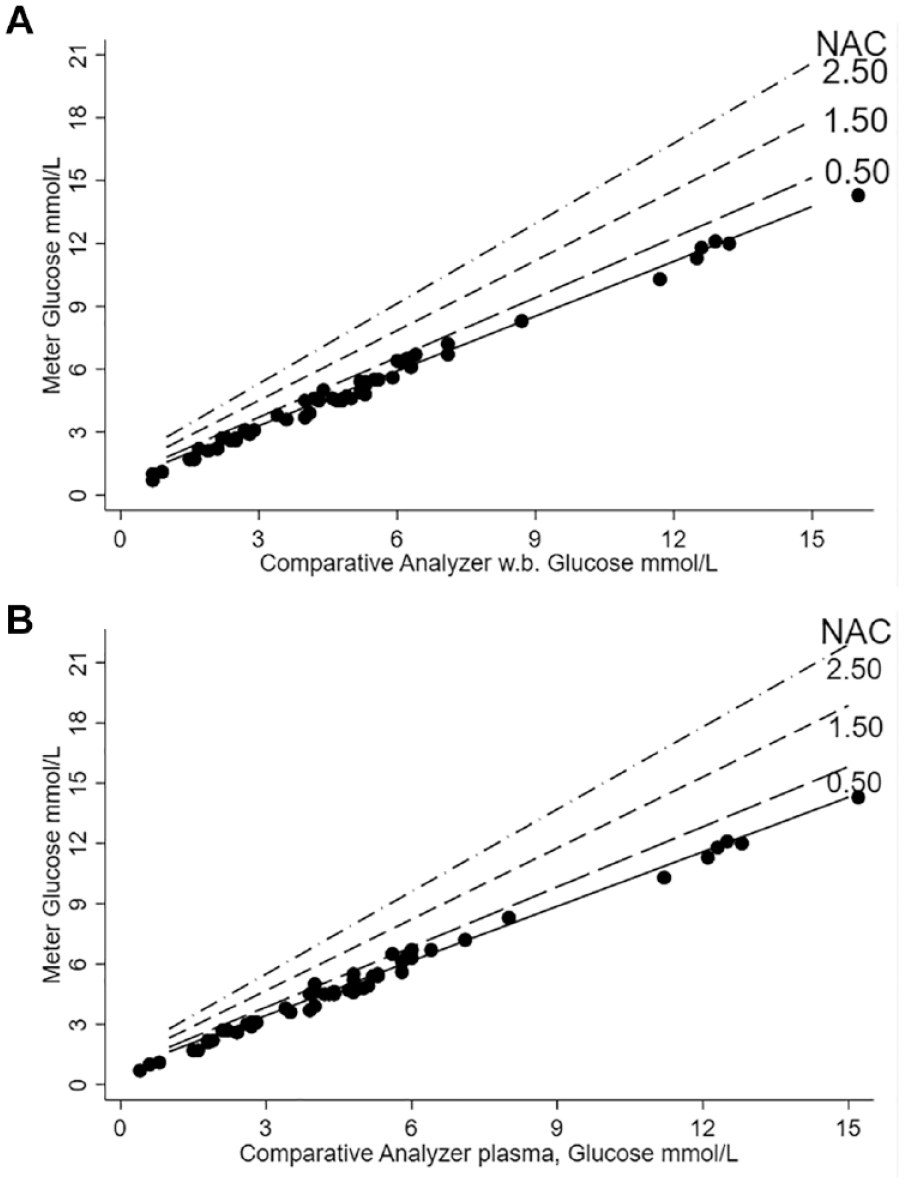
Predicted influence of NAC on glucose measurement with the glucose meter.
Discussion
The aim of this study was to determine the effect of therapeutic concentrations of NAC on the measurement of glucose using a Roche Diagnostics glucose dehydrogenase linked glucose meter in comparison to hospital laboratory comparative methods. This study evaluated and confirmed the medical device warning from Roche Diagnostics and the associated risk of interference with patient glucose results in the presence of therapeutic NAC concentrations, achieved following intravenous administration. 4 NAC has primarily been used to treat acetaminophen poisoning, but, it has also been prescribed as a cryoprotective agent during cancer chemotherapy as well as a preventative treatment for contrast-induced nephropathy.12,13 NAC dosing regimens, used to treat acetaminophen toxicity, have traditionally used 150 mg/ kg IV loading doses which can result in mean maximum plasma NAC concentrations of 55.4 mg/dL (3.4 mmol/L) 8 and, according to current study results, can produce a significant positive bias in the measurement of glucose with the GDH linked glucose meter. Several investigations describing the pharmacokinetics of NAC after single or multiple doses of oral NAC in healthy individuals indicate peak NAC plasma concentration of approximately 3.5 mg/L, which, based on our study results would have little to no effect on the performance of the Roche glucose meter, independent of the endogenous glucose concentration.14,15
The addition of NAC (>5 mg/dL or 0.31 mmol/L) to whole blood resulted in a statistically significant, concentration dependent positive bias in glucose measurement with the GDH linked glucose meter under investigation. Two NIST traceable comparative methods (plasma hexokinase and whole blood glucose oxidase) were used in this study to confirm the glucose meter measurements were not influenced by NAC. Genzen et al., 16 previously reported that supra-therapeutic NAC concentrations (up to 1000 mg/dL) inhibited several Trinder based assays on the Roche Cobas 8000 chemistry analyzer. However, we report NAC concentrations, achievable with IV administration of NAC, have no effect on the performance of the Radiometer ABL 837 blood gas electrode-based glucose oxidase method or the Roche plasma hexokinase method.
In the current study, we demonstrate that a whole blood NAC concentration of 407 mg/L (2.5 mmol/L) can produce a significant positive bias in glucose measurement using the Roche glucose meter. Multivariate analysis revealed that this interference is dependent upon the concentration of both NAC and the endogenous glucose level. For example, a 77% positive bias is predicted with a mixture of 54 mg/dL (3 mmol/L) glucose and NAC at a 40.7 mg/dL (2.5 mmol/L). Whereas, at a higher glucose concentration 216 mg/dL (12 mmol/L) in combination with the same NAC concentration (40.7 mg/dL), the expected positive bias is attenuated to 39%. To our knowledge, this is the first description of the multivariate nature of NAC susceptibility of a GDH-based glucose meter to positive bias. The mechanism of NAC-mediated positive bias with the Inform II glucose meter has not been described. The Inform II glucose meter utilizes membrane-linked GDH and pyrroloquinoline quinone (PQQ) to detect glucose and NAC is known to directly react with membrane-linked PQQ. 16 The direct reaction of NAC with membrane-bound PQQ is a potential mechanism for positive bias that can be assessed in future studies. NAC is known to react with hydrogen peroxide and mediate negative bias in glucose oxidase based glucose meters via the Trinder reaction 17 and Trinder reaction mediated-negative bias is not consistent with the observed positive bias on the Inform II meter. NAC-mediated positive bias is an important observation that should be considered in the design of future interference studies and interpretations of NAC-mediated interference with glucose measurements on clinical management of patients. Future clinical studies include an assessment of risk associated with the use of glucose dehydrogenase linked glucose meters in acetaminophen poisoning cases. Furthermore, additional studies could also assess if glucose dehydrogenase-PQQ linked glucose meters are susceptible to interference with endogenous thiols such as cysteine and homocysteine.
Conclusion
The experiments and analyses in this investigation demonstrated statistically significant positive biases for blood glucose measured with a Roche Diagnostics GDH linked glucose meter, caused by the addition of NAC within the therapeutic ranges (40.7, 20.35, 10.18, 5.09, and 2.54 mg/dl; 2.50, 1.25, 0.63, 0.32, and 0.16 mmol/L) attainable following IV administration.
Footnotes
Abbreviations
NAC, N-acetyl cysteine; PQQ, pyrroloquinoline quinone.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs M. E. Lyon and A. W. Lyon received financial support from Nova Biomedical as a third party consultant to conduct statistical analyses of clinical trial data for regulatory submission. Dr M. E. Lyon and Dr A.W. Lyon received speaking honoraria from Roche Diagnostics, Nova Biomedical, Radiometer, and Draeger Biomedical.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Role of Sponsor
No Sponsor
