Abstract

Patient portal utilization has become common in healthcare. A patient portal is a secure Web-based interface paired to an electronic medical record (EMR). Portals provide individuals with an accessible avenue to communicate with providers, access their medical record, and schedule appointments. 1 Secure messaging is a component of patient portals that may promote self-management, shared decision-making, and patient satisfaction by providing opportunities to correspond with providers. 2 In studies exploring the role of patient portals in diabetes self-management, portal use and secure messaging have been associated with positive clinical outcomes, including lower hemoglobin A1c (HbA1c) values.1,3 However, these relationships have not been studied in obstetrics. Pregnancy is a period of increased intensity and frequency of healthcare interactions, and for individuals with diabetes, optimal glycemic control requires significant participation.4,5 Thus, it was of interest to determine if portal use, in particular secure messaging, was associated with glycemic outcomes during pregnancy. The objective of this study was to determine if enrollment in and use of an EMR-linked electronic patient portal were associated with glycemic control during pregnancy.
This was a retrospective cohort study of patients who received prenatal care at a large academic medical center (2014-2016). All patients who had at least one HbA1c value or a postpartum 2-hour oral glucose tolerance test (OGTT) value in the EMR were eligible for inclusion. At the time of study, routine practice at this institution was to conduct HbA1c surveillance only among patients with diabetes or diabetes risk factors. Patients were considered enrolled in the portal if they had an account at the time of delivery. Glycemic control outcomes for enrollees were compared to non-enrollees. Enrollees were further categorized by the number of secure messages sent during pregnancy as active (≥1) vs inactive (0) users. Multivariable Poisson regression was used to assess the associations of (1) portal enrollment and (2) secure messaging activity with initial HbA1c value during pregnancy, postpartum HbA1c value, and postpartum 2-hour OGTT value.
Of the 3450 patients eligible for inclusion, 728 (21%) had either initial HbA1c (N=670), postpartum HbA1c (N=120), or 2-hour postpartum OGTT (N=89) values available. The majority of patients (59.1%, N=430) were enrolled in the portal. Of portal enrollees, 303 (70.5%) met criteria for active use. Glycemic control did not differ significantly by portal enrollment. However, patients who utilized secure messaging had lower odds of having initial HbA1c value ≤7% or postpartum 2-hour OGTT value ≤140 mg/dL (Table 1).
Glycemic Control-Related Outcomes During Pregnancy by Patient Portal Enrollment and Secure Messaging Use.
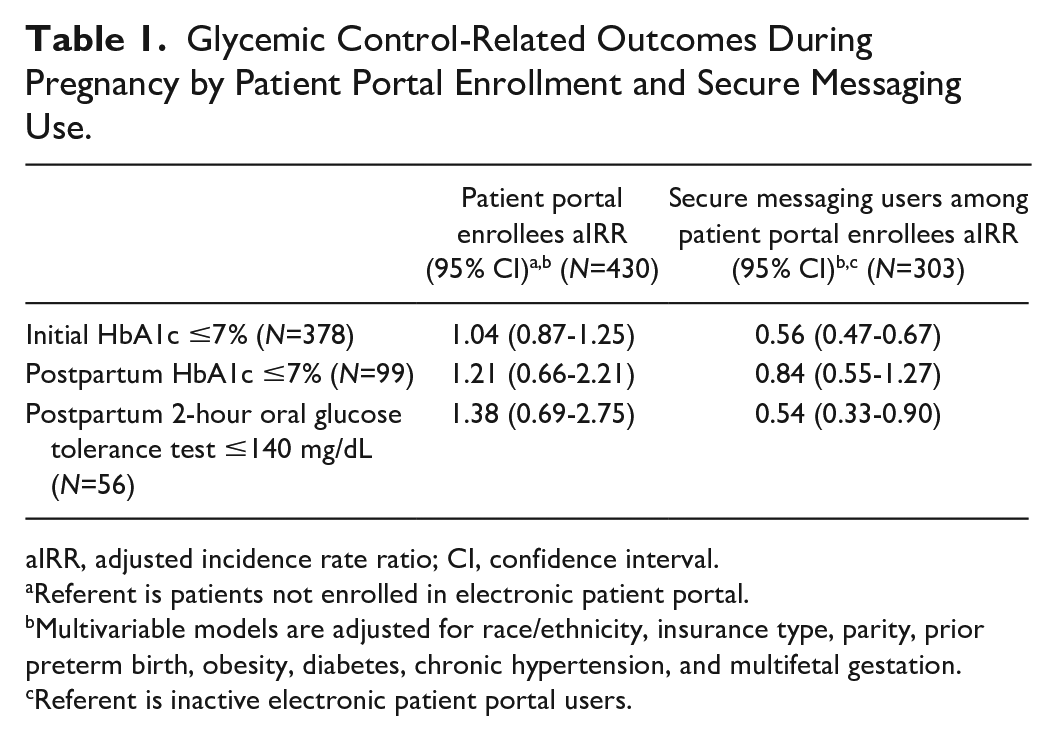
aIRR, adjusted incidence rate ratio; CI, confidence interval.
Referent is patients not enrolled in electronic patient portal.
Multivariable models are adjusted for race/ethnicity, insurance type, parity, prior preterm birth, obesity, diabetes, chronic hypertension, and multifetal gestation.
Referent is inactive electronic patient portal users.
In contrast to studies in primary care, our study showed that patient portal use was not associated with significant differences in glycemic control during pregnancy. Rather, patients who were active users were less likely to have within-goal glycemic control. This finding suggests that pregnant patients with suboptimal glycemic control more readily engaged in secure messaging with providers. Further prospective investigation is necessary to understand patient utilization of portals and how portals may be harnessed as a tool for health promotion and education, particularly for conditions such as diabetes in which frequent provider contact is central to disease management.
Footnotes
Abbreviations
EMR, electronic medical record; HbA1c, hemoglobin A1c; OGTT, oral glucose tolerance test.
Authors’ Note
Presented as a poster presentation at the 2018 Annual meeting of the Society for Reproductive Investigations in San Diego, California.
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported, in part, by the Northwestern University Clinical and Translational Sciences Institute Enterprise Data Warehouse Pilot Program (UL1TR001422). In addition, LMY was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (K12 HD05012) at the time of the study.
