Abstract
A novel, multi-dimensional protocol named GENIE has been in use for intensive insulin therapy (IIT, target glucose <140 mg/dL) in the surgical intensive care unit (SICU) after open heart surgery (OHS) at VA Pittsburgh since 2005. Despite concerns over increased mortality from IIT after the publication of the NICE-SUGAR Trial, it remains in use, with ongoing monitoring under the MAGIC GENIE Project showing that GENIE performance over 12 years (2005-2016) aligns with the current consensus that IIT with target blood glucose (BG) <140 mg/dL is advisable only if it does not provoke severe hypoglycemia (SH). Two studies have been conducted to monitor glucometrics and outcomes during GENIE use in the SICU. One compares GENIE (n = 382) with a traditional IIT protocol (FORMULA, n = 289) during four years of contemporaneous use (2005-2008). The other compares GENIE’s impact overall (n = 1404) with a cohort of patients who maintained euglycemia after OHS (euglycemic no-insulin [ENo-I], n = 111) extending across 12 years (2005-2016). GENIE performed significantly better than FORMULA during contemporaneous use, maintaining lower time-averaged glucose, provoking less frequent, severe, prolonged, or repetitive hypoglycemia, and achieving 50% lower one-year mortality, with no deaths from mediastinitis (0 of 8 cases vs 4 of 9 on FORMULA). Those benefits were sustained over the subsequent eight years of exclusive use in OHS patients, with an overall one-year mortality rate (4.2%) equivalent to the ENo-I cohort (4.5%). The results of the MAGIC GENIE Project show that GENIE can maintain tight glycemic control without provoking SH in patients undergoing OHS, and may be associated with a durable survival benefit. The results, however, await confirmation in a randomized control trial.
Keywords
Opinion regarding tight glycemic control (TGC) in critical care has shifted significantly more than once in the past two decades. Until 2001, mild-to-moderate hyperglycemia during critical illness was considered either irrelevant or a potentially beneficial survival adaptation. 1 After the Leuven trial was published, however, the purported mortality benefit of maintaining TGC with intensive insulin therapy (IIT) in critical care 2 was universally accepted. With endorsement by the American Diabetes Association (ADA)3,4 and American College of Endocrinology (AACE), 5 IIT protocols proliferated in intensive care units (ICUs) everywhere.6-10
The surgical ICU (SICU) at VA Pittsburgh Healthcare System (VAPHS) was no exception. An IIT protocol based on the Davidson equation (“FORMULA”; target blood glucose [BG] 90-140 mg/dL)11,12 was implemented in the SICU in 2002 for patients undergoing open heart surgery (OHS). Three years later, a protocol developed in-house named “GENIE” (
We report here the results of the MAGIC GENIE project (an acronym for
Methods
Study Protocols
FORMULA
FORMULA employs the Davidson equation to hand-calculate insulin infusion rates (IR = [BG-60] × an insulin sensitivity factor [ISF, adjusted between 0.02 and 0.08 “as needed” by the supervising physician, per SICU policy at the time]). 12 In 2003, however, the occurrence of six cases of fatal mediastinitis after OHS prompted an inquiry that found all six had uncontrolled hyperglycemia, a known risk factor with an almost eightfold higher risk for deep sternal wound infections (DSWI) after OHS.15,16 A root cause analysis revealed a startling dichotomy in FORMULA performance. Eighty percent of patients achieved BG < 140 mg/dL with relatively little adjustment in ISF, but the other 20% (including the six who died) had persistent hyperglycemia with breakthrough periods of “paradoxical” hypoglycemia. The Committee attributed the poor performance to therapeutic inertia (failure to escalate ISF) or therapeutic delay (delayed ISF escalation) arising from a lack of commitment to TGC among SICU staff. The Committee recommended changing SICU policy in the short term to permit ISF adjustment by nurses without physician input, and changing the IIT protocol in the longer term to one that could (a) circumvent therapeutic inertia, (b) counteract therapeutic delay, and (c) overcome insulin resistance, without increasing the risk of SH. After an extensive literature search failed to find one meeting all four criteria,6-10 the Committee assigned the task of building one to an in-house team comprising a skilled critical care nurse (CAC), an experienced diabetologist (RHR), and a diabetes-savvy programmer (PLP).
GENIE
Table 1 summarizes how clinical strategies based on accepted scientific concepts were translated into practical solutions, and incorporated in a multi-dimensional, continuous decision surface with an intuitive, user-friendly interface (Figure 1a and b) named GENIE, a registered trademark owned by the Department of Veteran’s Affairs. After extensive beta testing under multiple simulated scenarios, it debuted in the SICU in January 2005, initially as an optional alternative to FORMULA. Four years later, GENIE became the exclusive SICU IIT protocol, based on superior glycemic performance.
Concepts, Strategies, and Solutions Incorporated into GENIE’s Multi-Dimensional Platform.

Abbreviations: BG, blood glucose; DAF, dose adjustment factor; NPH, Neutral Protamine Hagedorn; PFD t , proportional fractionation of Dt; SICU, surgical intensive care unit.
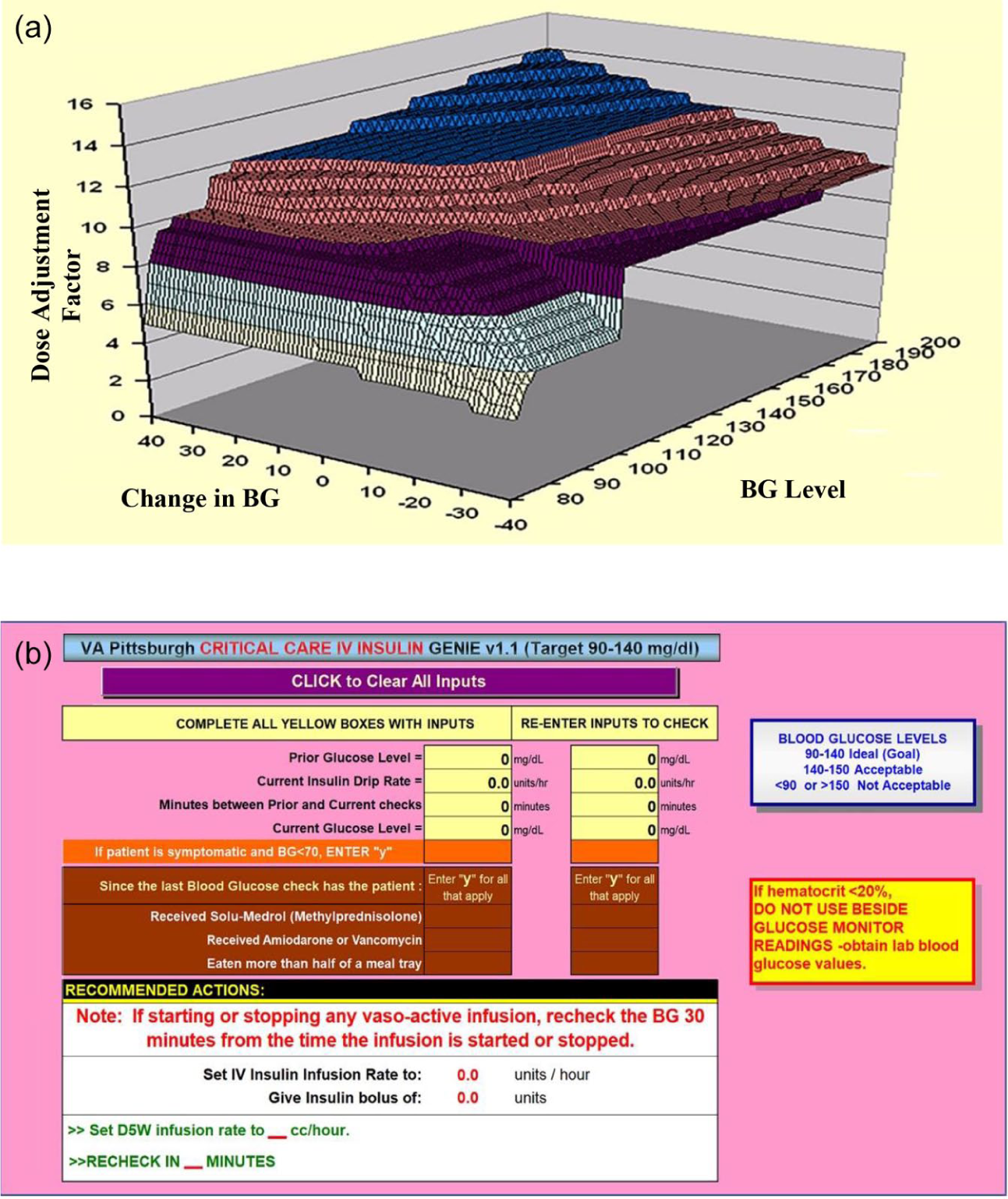
Unique features of GENIE. (a) A multi-dimensional algorithm with a continuous decision surface. (b) An intuitive, user-friendly interface.
Study Population
All patients undergoing OHS between 2005 and 2017 were included if they satisfied four criteria for IIT:
(i) Pre-existing diabetes OR stress hyperglycemia (2 successive BGs > 140 mg/dL)
(ii) An order for IIT (FORMULA or GENIE)
(iii) An insulin infusion dispensed by pharmacy and administered for ≥12 h
(iv) Repetitive BG monitoring for ≥12 h
Study Cohorts
Contemporaneous Use Study (2005-2008) of GENIE vs FORMULA after OHS.
Contemporaneous FORMULA OHS cohort: n = 289
Contemporaneous GENIE OHS cohort: n = 382
Overall Use Study (2005-2016) of GENIE vs a Euglycemic No-Insulin (ENo-I) OHS Cohort
Overall GENIE OHS cohort: 1404 OHS patients on GENIE
Overall ENo-I OHS cohort: 111 OHS patients who maintained BG < 140 mg/dL without requiring any insulin (IIT or subcutaneous) in the SICU (All BGs < 140 mg/dL).
Data Extraction
The following were extracted through a combination of manual chart review and database queries:
a. Clinical factors: Age, gender, diabetes history, components of the Cleveland Clinic Severity Score (CCSS), 16 length of stay in the SICU (SICU-LOS), glucometric data during IIT and in-hospital subcutaneous insulin therapy (ScIT), and cardioprotective drug therapy and HbA1c.
b. Glucometric performance 17 was assessed from:
i. Glycemic control: Mean ± standard error (SE) and median (interquartile range [IQR]) of time-averaged glucose (TAG); duration and number of BGs checks on IIT.
ii. BG variability: Mean ± standard deviation (SD) of mean BGs.
iii. Hyperglycemic exposure: Distribution of TAG < 140 mg/dL vs ≥140 mg/dL, and cumulative time (hours) with BG > 140 mg/dL (mean dwell time > 140 ± SE).
iv. Hypoglycemic exposure: Incidence (number of patients with ≥1 BG episode < 70 mg/dL); severity (episode nadir < 50 mg/dL, or 50-69 mg/dL); Repetitive Frequency (number of episodes per patient with BG < 70 mg/dL); and dwell time<70 (cumulative time [hours] with BG < 70 mg/dL).
v. Time-weighted exposure to glucose <70 mg/dL and ≥140 mg/dL (TWEG<70;TWEG≥140), a composite of severity and duration, calculated by integrating the area under the curve over time.
Outcomes
a. Fatal mediastinitis and DSWI were diagnosed by Infectious Disease consultants, based on CDC criteria. 18
b. One-year mortality was verified by two independent but mutually complementary methods: EITHER a date of death listed in VA records, OR a date of last contact (clinic/laboratory) ≥365 days after OHS recorded in the patient’s chart. Patients with last face-to-face contact <365 days after OHS and no recorded date of death were censured in Kaplan–Meier (KM) analysis as “lost to follow-up” after last contact.
Statistical Analysis
KM analysis was used to derive relative likelihood estimates of mortality/survival based on Cox’s Proportional Hazards Model (hazard ratio [HR], 95% confidence intervals [CI]). 19 Cohort differences were substantiated by t tests (for continuous variables), Mann–Whitney U tests (for ordinal data), χ 2 tests (for categorical data), and log-rank tests (for HR).
Results
Contemporaneous Use Study (2005-2008) of GENIE (n = 382) vs FORMULA (n = 289) after OHS
Glucometric performance
Glucometric performance (Table 2) was significantly better on GENIE than FORMULA, despite equivalent frequency of BG monitoring and IIT duration. The relatively small difference in TAG (GENIE: 117 ± 15 mg/dL; FORMULA: 121 ± 15 mg/dL) was attributable to the glycemic dichotomy mentioned earlier, whereby the majority who did well on FORMULA obscured a problematic minority. The magnitude of difference became clear only when performance was assessed by the four criteria used to build GENIE.
(i) Circumventing therapeutic inertia: Only 23 patients failed to achieve TAG < 140 mg/dL on GENIE (23/382 [6%]) compared to 36 on FORMULA (36/289 [12%]; P = .0004).
(ii) Counteracting therapeutic delay: TAG dwell time>140 (Mean ± SE) on GENIE (46 ± 4 minutes) was less than half that on FORMULA (92 ± 14 minutes; P < .0001).
(iii) Overcoming insulin resistance: The 23 patients who failed to achieve TAG >140 mg/dL on GENIE spent substantially less time above target (dwell time≥140 73 ± 23 min) than the 36 on FORMULA (284 ± 45 minutes; P = .014), with fewer maintaining TAG > 150 mg/dL (GENIE: 10/382 [3.2%]; FORMULA: 22/289 [7.6%]; P = .003).
(iv) Minimizing hypoglycemia risk: Hypoglycemia (BG < 70 mg/dL) was less frequent on GENIE compared with FORMULA (GENIE:65/382 [17%]; FORMULA: 87/289 [30.1%]; P < .001), less severe (BG <50 mg/dL, GENIE: 4/382 [1.1%]; FORMULA: 21/289 [7.3%]; P < .001), less repetitive (GENIE:1.4 ± 0.1 episodes/patient; FORMULA: 1.99 ± 0.15; P < .001), and less prolonged (dwell time<70, GENIE: 1.03 ± 0.12 hours; FORMULA: 1.79 ± 0.25; P < .001).
(v) Minimizing adverse glycemic exposure:
• TWEG≥140 (Hyperglycemic exposure) was ~85% lower on GENIE (12.3 ± 3.2 mg/dL/h) than on FORMULA (80.3 ± 21.3; P = .014).
• TWEG<70 (Hypoglycemic exposure) was reduced ~60% on GENIE (5.9 ± 1.2 mg/dL/h) compared with FORMULA (14.5 ± 2.7; P < .001).
GENIE Performance vs FORMULA During Contemporaneous Use (2005-2008).
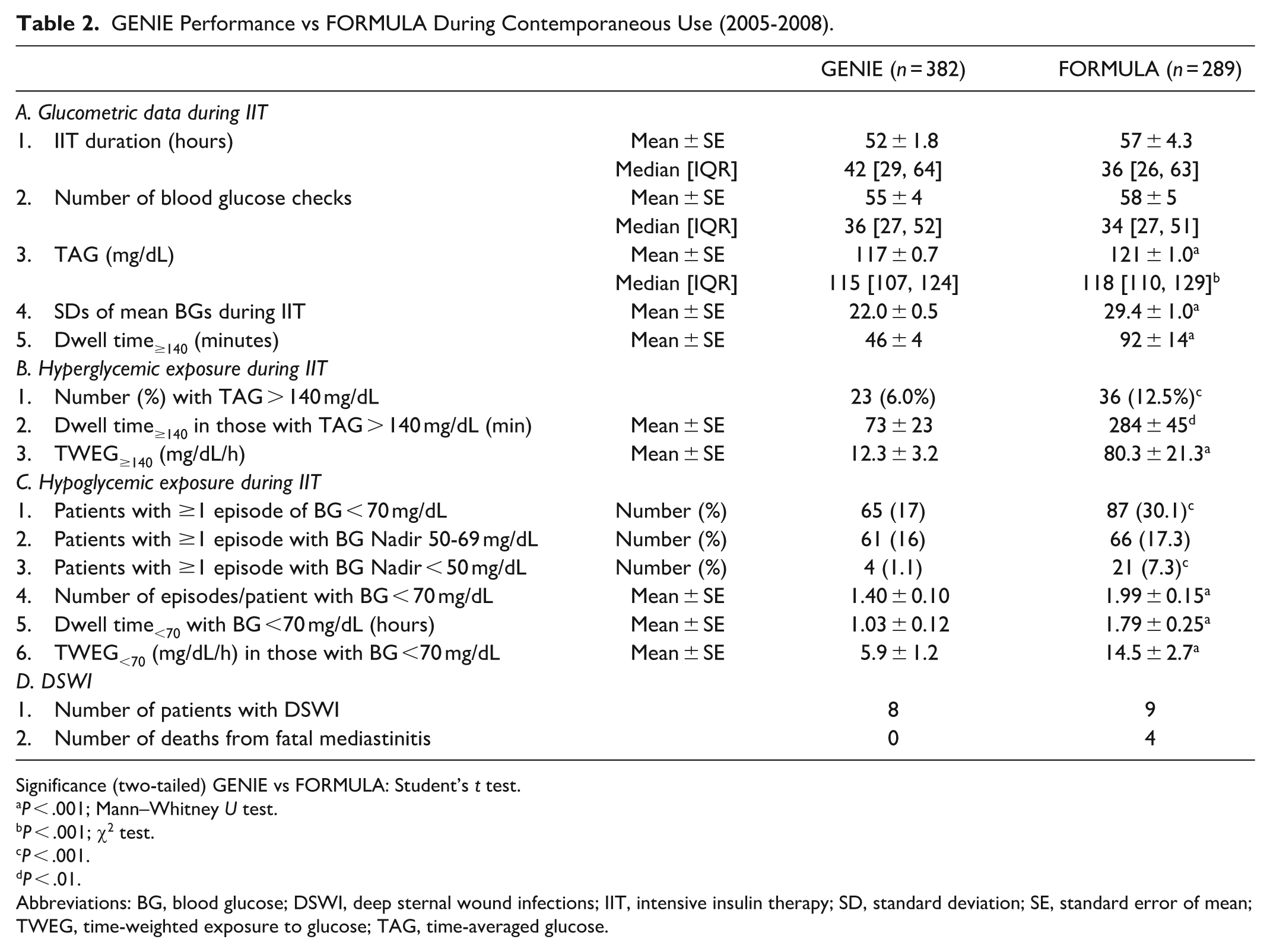
Significance (two-tailed) GENIE vs FORMULA: Student’s t test.
P < .001; Mann–Whitney U test.
P < .001; χ2 test.
P < .001.
P < .01.
Abbreviations: BG, blood glucose; DSWI, deep sternal wound infections; IIT, intensive insulin therapy; SD, standard deviation; SE, standard error of mean; TWEG, time-weighted exposure to glucose; TAG, time-averaged glucose.
Outcomes after OHS
(i) Fatal mediastinitis: Of the nine patients who developed DSWI on FORMULA, four succumbed to fatal mediastinitis, whereas none of the eight GENIE-treated DSWI patients died (GENIE: 0/8 [0%]); FORMULA: 4/9 [44.4%]; Table 2).
(ii) Mortality: One-year mortality was significantly different in the two cohorts (GENIE: 15/382 [3.8%]; FORMULA: 23/289 [8%]; P = .025), with KM analysis showing that GENIE-treated patients had a >50% lower likelihood of dying in the year after OHS than FORMULA-treated patients (HR = 0.48; 95% Confidence Interval [CI] = 0.25, 0.93; P = .026; Figure 2a).
(iii) Risk factors known to influence mortality/survival: There was no difference in prevalence of risk factors in the two cohorts, such as demographics (predictors of risk), preoperative CCSS (a predictor of mortality after OHS), SICU-LOS (a measure of complexity/complications of critical care), post-SICU BGs (a measure of in-hospital glycemic management), one-year average A1c in patients with diabetes (a measure of outpatient glycemic management), and cardioprotective drug therapy (a determinant of outcomes after cardiac surgery) (Table 3).20,21
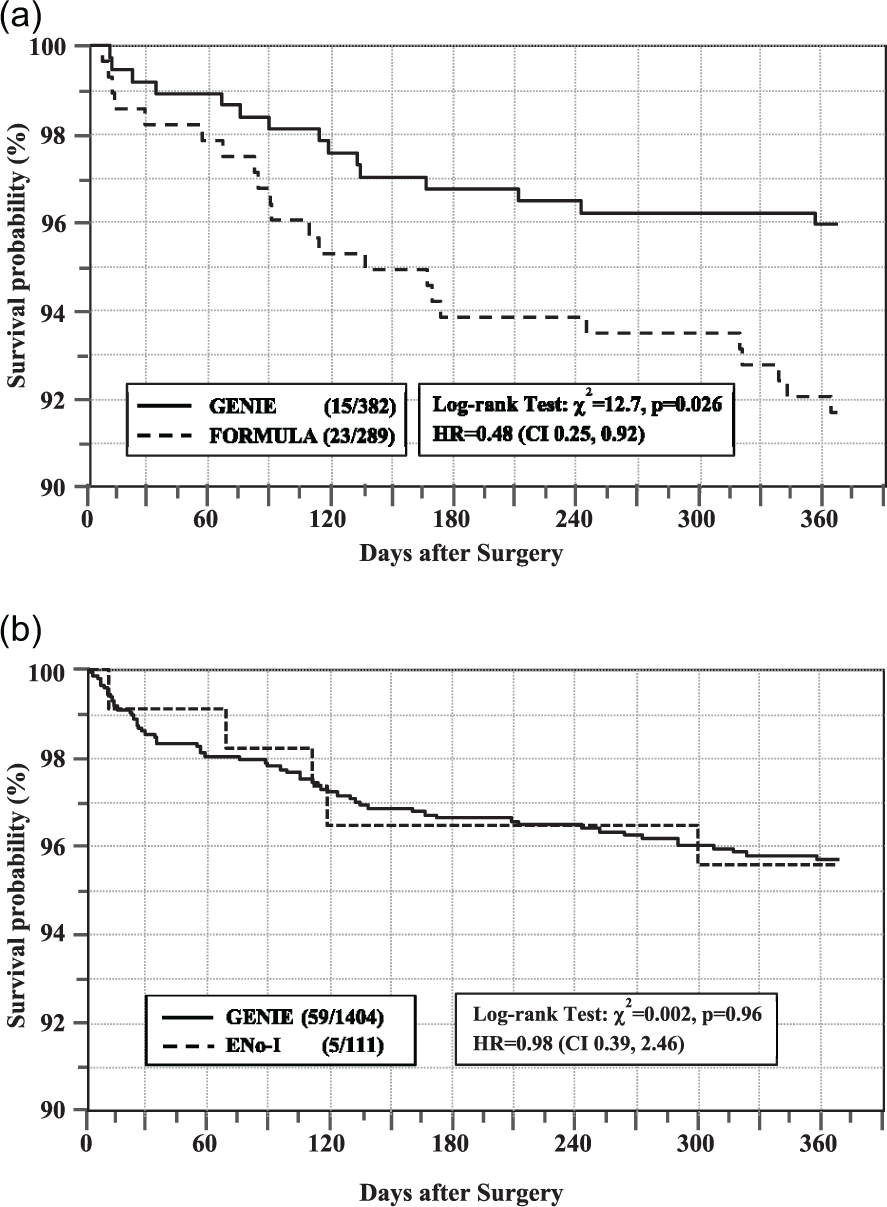
Kaplan–Meier survival analysis. (a) GENIE vs FORMULA during contemporaneous use (2005-2008). (b) GENIE vs euglycemic no-insulin (Eno-I) OHS cohort (2005-2017).
Factors Influencing Mortality in the GENIE and FORMULA Cohorts.
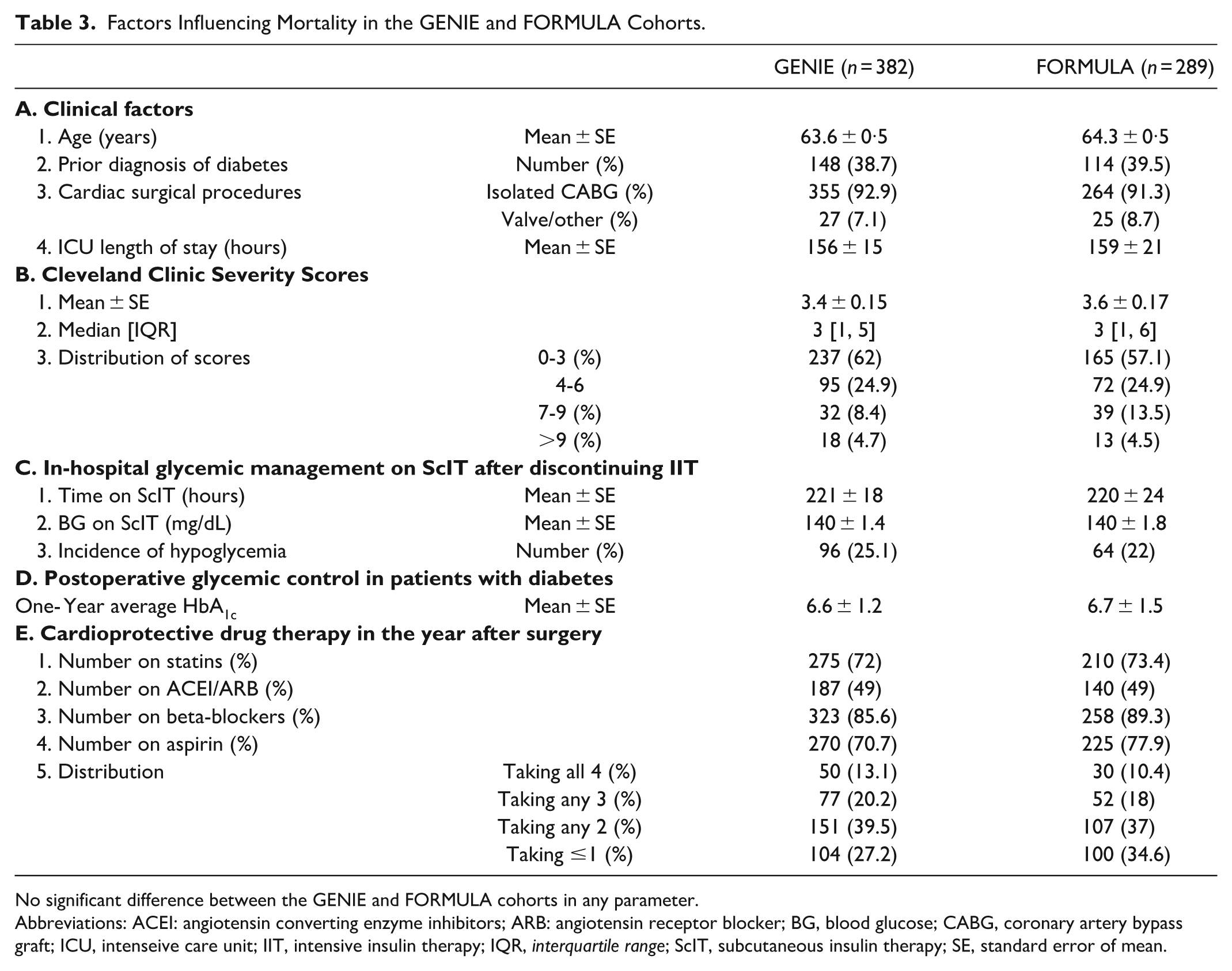
No significant difference between the GENIE and FORMULA cohorts in any parameter.
Abbreviations: ACEI: angiotensin converting enzyme inhibitors; ARB: angiotensin receptor blocker; BG, blood glucose; CABG, coronary artery bypass graft; ICU, intenseive care unit; IIT, intensive insulin therapy; IQR, interquartile range; ScIT, subcutaneous insulin therapy; SE, standard error of mean.
Overall Use Study (2005-2016) of Outcomes after OHS in the GENIE and ENo-I Cohorts
Glucometrics
TAG < 140 mg/dL was achieved in 1290/1404 (92%) GENIE-treated subjects, compared with 100% (by definition) of the 113 Eno-I subjects, with no significant difference in achieved TAG (mean ± SE; GENIE: 119 ± 0.4 mg/dL, Eno-I: 123 ± 1.1 mg/dL). However, hypoglycemia in the ENo-I cohort was less frequent (BG < 70 mg/dL; GENIE: 296/1404 [21.1%]); ENo-I: 10/111 [9%]), with no subjects experiencing BG < 40 mg/dL, compared with 7/1404 [0.5%] on GENIE. Notably, significant protocol violations (delayed BG rechecks) were found in all 7 patients with SH in the GENIE cohort (required in 30 min, rechecked at 120-180 minutes in 4; required in 60 minutes, rechecked at 180-210 minutes in 3).
Mortality
The number of deaths in the two cohorts were similar at 90 days (GENIE: 31/1404 [2.2%]; ENo-I: 2/111 [1.8%]) and one year (GENIE: 59/1404 [4.2%]; ENo-I: 5/111 [4.5%]), with KM survival analysis (Figure 2b) revealing overlapping survival curves (HR = 0.98 [CI: 0.39, 2.46, P = .96).
Accounting for biases related to time and selection
Figure 3 shows that rates of mortality (Figure 3a), SH (Figure 3b), and fatal mediastinitis (Figure 3c) in the GENIE cohort remained constant (equivalent to the ENo-I cohort), across the initial 4 years of contemporaneous use, the next 4 years of exclusive use, and the last 4 years of exclusive use, ruling out time-related biases. Similarly, higher rates of mortality, SH, and fatal mediastinitis also remained constant in the 4 years before implementing GENIE (2001-2004: 57/695 [8.2%]) and the 4 years of contemporaneous use in the FORMULA cohort (2005-2008: 23/289 [8%]), ruling out any selection bias during contemporaneous use.
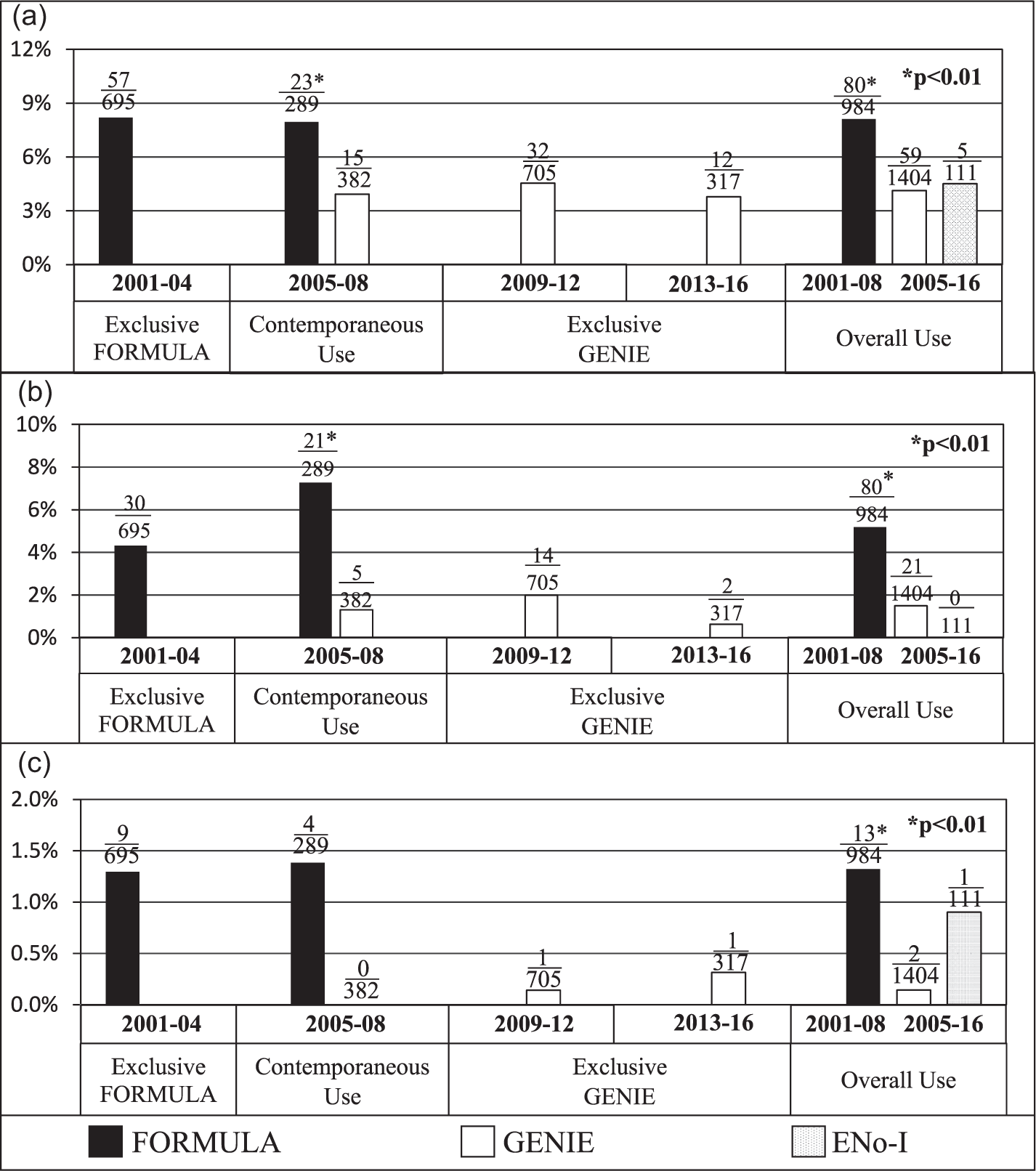
Outcomes over time in different OHS cohorts. (a) One-year mortality after OHS. (b) Severe hypoglycemia (BG < 50 mg/dL). (c) Fatal mediastinitis.
We also periodically verified that there were no material changes in protocols, procedures, and policies in the SICU. In 2013, however, the Critical Care Chief retired, and the Chief of Cardiothoracic Surgery, who performed >75% of OHS at VAPHS, was recruited elsewhere. There was a sharp drop in OHS procedures while his replacement was being recruited, from 695, 671, and 705 in each of the three preceding 4-year periods to 317 in 2013-2016 (Figure 3). However, the mortality rate remained constant, suggesting that the change did not impact outcomes.
Discussion
The results of the MAGIC GENIE project show that maintaining near-euglycemia with GENIE after OHS without provoking SH (target BG 90-140 mg/dL, achieved in >90% of patients) provides a significant and durable mortality benefit, equivalent to maintaining near-euglycemia without requiring insulin. Even if protocol violations are accepted as an inevitable accompaniment of IIT, achieving an SH rate of 0.5% in routine clinical care, without the specialized nurse training, day-to-day physician oversight or dedicated resources of randomized controlled trials (RCTs) is striking. Table 4 shows that, despite those resources, pooled SH rates in two meta-analyses of RCTs of IIT in surgical critical care, one before NICE-SUGAR (Wiener et al 22 : 5%), one after (Griesdale et al 23 : 10.7%), were much higher than on GENIE, reflecting an acceptance of SH as the unavoidable price of the purported mortality benefit of TGC with IIT. 24 That perception was overturned by the NICE-SUGAR trial, laying the foundation of the current consensus that TGC (target BG < 140 mg/dL) in critical illness is inadvisable unless SH can be prevented.24-26
Incidence of Hypoglycemia with Various CDS Platforms and in RCTs of IIT during Critical Care.
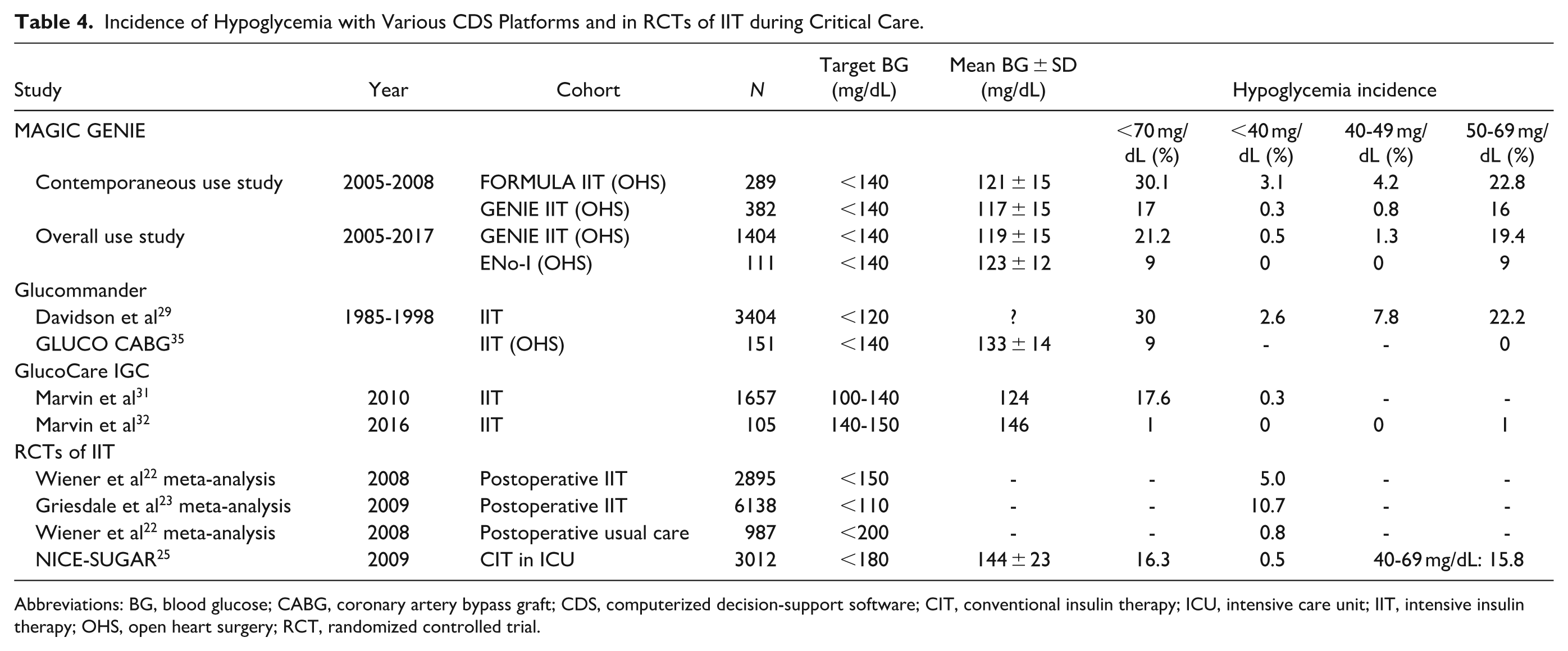
Abbreviations: BG, blood glucose; CABG, coronary artery bypass graft; CDS, computerized decision-support software; CIT, conventional insulin therapy; ICU, intensive care unit; IIT, intensive insulin therapy; OHS, open heart surgery; RCT, randomized controlled trial.
Avoiding SH during IIT, however, is easier said than done with current technology. Subcutaneous CGM sensors are not approved for use in the ICU in the United States, 27 and there are no signs of an imminent breakthrough in real-time blood glucose sensor technology. 28 As a result, the focus has shifted to open-loop computerized decision-support software (CDS) programs for IIT, like Glucommander and GlucoCare, the two best-known commercially available CDS protocols.29,30 Both platforms employ two-dimensional equations of varying slopes to model the relationship between blood glucose and insulin infusion rate, and neither incorporates bolus insulin dosing (although an updated version of GlucoCare does).31,32 GENIE, by contrast, uses a continuous multi-dimensional decision surface that incorporates routine bolus insulin dosing, leading to better performance, in theory. Proving it in practice, however, requires head-to-head comparative studies, which do not exist, ruling out a direct assessment of their relative merits. Nevertheless, an objective, albeit limited indirect comparison of performance is still possible using published glucometric and outcomes data from observational studies (Table 4).
Glucometric data in 5802 patients on Glucommander show that BG in a significant proportion remains >150 mg/dL for several hours.29,33 That compares with a mean time to target of 1.7 hours on GENIE, and an average dwell time>140 of 46 ± 4 minutes, despite which GENIE generated less hypoglycemia than Glucommander (<70 mg/dL: 21.2% vs 30%; <50 mg/dL: 1.8% vs 7.8%; <40 mg/dL: 0.5% vs 2.6%). However, the GLUCO coronary artery bypass graft (CABG) surgery study, a relatively small RCT of Glucommander (n = 151), reported lower hypoglycemia rates, albeit at the expense of a higher mean BG (133 ± 14 mg/dL).29,33 A large observational study of the GlucoCare IGC System also reported hypoglycemia rates similar to those with GENIE, but hypoglycemia was virtually eliminated on a newer version with a higher target (140-150 mg/dL).31,32
Although the glucometric benefit of GENIE over Glucommander and GlucoCare in practice might be debatable, its outcome benefit is not (Table 5). There are no outcomes studies of GlucoCare, and the only one of Glucommander (GLUCO CABG) showed no mortality benefit from maintaining BG 100-140 mg/dL after OHS vs a more conservative target (140-180 mg/dL). 34 However, the mean BG on GlucoCare (133 ± 14 mg/dL) was higher than even with FORMULA (121 ± 15 mg/dL), not just GENIE (117 ± 15 mg/dL), as was 90-day mortality (~5%, approximated from Figure 2A; FORMULA: 3.8%; GENIE: 0.8%). 34 Any connection between the higher mean BG and lack of mortality benefit is purely speculative.
Mortality After Open Heart Surgery With and Without IIT.
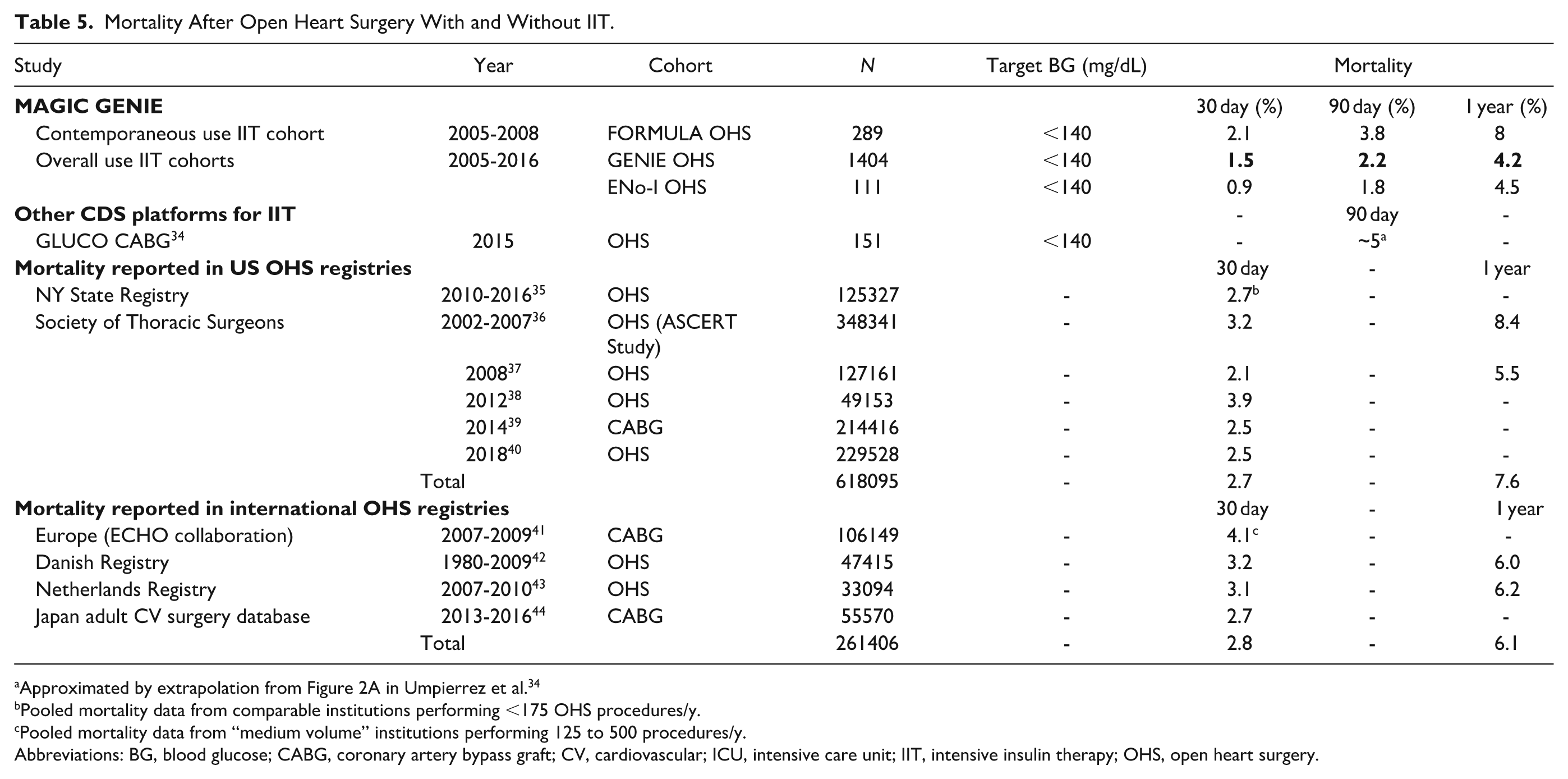
Pooled mortality data from comparable institutions performing <175 OHS procedures/y.
Pooled mortality data from “medium volume” institutions performing 125 to 500 procedures/y.
Abbreviations: BG, blood glucose; CABG, coronary artery bypass graft; CV, cardiovascular; ICU, intensive care unit; IIT, intensive insulin therapy; OHS, open heart surgery.
GENIE’s impact on mortality, on the other hand, was not only superior to FORMULA (which employed the same two-dimensional equation as Glucommander), it was equivalent to not requiring insulin to maintain near-euglycemia, and was durable up to a year after OHS. Table 5 shows, furthermore, that GENIE’s mortality benefit was at least equivalent to, if not somewhat better than pooled mortality rates in several large US and international OHS registries.35-44 In other words, there was no “excess mortality” above the “expected” after OHS from using GENIE to maintain near-euglycemia without provoking SH.
From a clinical perspective, avoiding SH during TGC may be paramount. Hypoglycemia in critical illness correlates with mortality, although the association may not be causal, as the NICE-SUGAR investigators were careful to point out. 25 The association holds true for both iatrogenic and spontaneous hypoglycemia, leading some to argue that SH in critical illness may be a pre-terminal predictor of imminent death,45 -47 and that hyperglycemia may be a beneficial survival adaptation in critical illness.48,49
Our results suggest to the contrary that hyperglycemia is detrimental, not beneficial. The most compelling evidence comes from the virtual elimination of fatal mediastinitis during 13 years of GENIE use. Uncontrolled hyperglycemia, long known to predispose to surgical site infections, 50 is a major risk factor for DSWI mortality after OHS.51-54 High-quality evidence, predating GENIE, and unequivocally affirmed across the years, shows that controlling hyperglycemia after OHS significantly reduces that risk.15,55-57
Unlike the relationship between hyperglycemia and infections, the pathophysiology of the relationship between SH and death is uncertain. Two putative mechanisms are neuronal injury from fuel starvation, 58 and distributive shock 25 from cardiac ischemia, 59 dysrhythmias, 60 or autonomic dysregulation. 61 Both, however, have an immediacy that does not explain the delayed and durable benefit associated with GENIE, which is more consistent with protection against long-lasting damage, such as endothelial injury induced by oxidative stress/cytokine activation during hypoglycemia.62,63 In the early postoperative period, such effects may be particularly deleterious to newly implanted vein grafts undergoing extensive intimal re-modeling in the face of intense hemodynamic shear stress and wall tension.64,65 Those effects, which suggest enhanced endothelial vulnerability in the early postoperative period, have been implicated in subsequent vein graft failure.66,67 The early reduction in both hypoglycemic and hyperglycemic exposure on GENIE in the SICU (but not subsequently during ScIT in the hospital) is consistent with a durable mortality benefit from early protection against endothelial damage.
One potential weakness of the project is its non-randomized retrospective observational design, which falls short of the standards of comparative effectiveness research. 67 Some of that weakness is offset by the fact that the benefit was realized in day-to-day clinical care of all participants, free of both the selection bias of most non-randomized observational trials and the exclusionary bias of idealized (homogeneous) RCT populations. 68 The fact that the benefit was achieved without dedicated resources, such as specialized nurse training and direct physician oversight, enhances its practical value. Nevertheless, the inherent weakness of the design means that an RCT is needed to confirm the benefit, and whether a reduction in hyperglycemic or hypoglycemic exposure is responsible.
Another potential weakness of the study design is its vulnerablity to time-related biases, such as changes in care policies, personnel, or surgical techniques leading to progressive improvement in mortality across its 12-year duration. However, there was no decline in mortality in the GENIE cohort, remaining stable across the 12-year study duration (Figure 3). This parallels data from the STS database (Table 5) showing that mortality in institutions with comparable OHS output have remained stable since at least 2008.
In the final analysis, the results of the MAGIC GENIE Project are not undermined by the study’s observational design or prolonged duration. They show that incorporating well-recognized practical strategies, based on widely accepted scientific concepts, into a unique, multi-dimensional platform (GENIE) can maintain TGC without provoking SH during routine critical care in patients undergoing OHS. This may be associated with a durable survival benefit, although that awaits confirmation in a randomized controlled trial.
Footnotes
Acknowledgements
The study was conducted as part of clinical operations at VAPHS exclusively with intramural funding, resources, and support.
Harsha Rao, Candace Cunningham, and Peter Perreiah had full access to the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
The authors wish to thank Rajiv Jain, MD, former Chief of Staff, VAPHS, for his advocacy, encouragement, and support during the development and implementation of GENIE; Robert Muder, MD, former Chief of Infectious Diseases, VAPHS and Chair of the Mediastinitis Inquiry; Paul Rodgers, MD, former Chief of Critical Care, VAPHS; Joyce Ewing, RN, Program Leader, Critical Care Nursing Service Line, VAPHS; Marco Zenati, MD, former Chief of Cardio-thoracic Surgery, VAPHS; Mark Wilson, MD, Chief of Surgery, VAPHS; and Frederick DeRubertis, MD, former Vice President, Medicine Service Line, VAPHS, for their active support and encouragement of the MAGIC GENIE project; and Mary Korytkowski, MD, Professor of Medicine, Division of Endocrinology, University of Pittsburgh School of Medicine, MD, for reviewing the manuscript and making suggestions to improve it.
Last, but not the least, this complex and long-running effort would not have been possible without the active engagement and cooperation of SICU nurses over the past 15+ years. Their dedication and ongoing feedback are singularly responsible for GENIE’s success in improving patient outcomes.
Abbreviations
ACEI, Angiotensin Converting Enzyme Inhibitors, ARB, Angiotensin Receptor Blocker, BG, Blood glucose, CCSS, Cleveland Clinic Severity Score, CDS, Computerized Decision-support Software, CI, 95% Confidence Interva, CIT, Conventional Insulin Therapy, DSWI, Deep Sternal Wound Infections, HR, Hazard Ratio, IIT, Intensive Insulin Therapy, IQR, Interquartile Range, IR, Infusion Rate, KM, Kaplan-Meier, LOS, Length of Stay, OHS, Open Heart Surgery, RCT, Randomized Controlled Trial, ScIT, Subcutaneous Insulin Therapy, SD, Standard Deviation, SE, Standard Error, SH, Severe Hypoglycemia, SICU, Surgical Intensive Care Unit, STS, Society of Thoracic Surgeons, TAG, Time-Averaged Glucose, TGC, Tight Glycemic Control, TWEG, Time-Weighted Exposure to Glucose, VAPHS, VA Pittsburgh Healthcare System.
Author Contributions
1. Contributions to the work:
a) Conception and Design: Rao, Perreiah, Cunningham
b) Acquisition and analysis of data: Rao, Perreiah,
c) Interpretation: Rao, Perreiah, Cunningham
2. Contributions to the manuscript:
a) Drafting the work: Rao
b) Reviewing/revising intellectual content: Rao, Perreiah, Cunningham
3. Final approval: Rao, Perreiah, Cunningham
4. Accountability for accuracy and integrity of the work: Rao, Perreiah, Cunningham.
R. Harsha Rao, MD, has a joint appointment as Professor of Medicine, Division of Endocrinology, University of Pittsburgh, School of Medicine, Pittsburgh, PA.
GENIE is a registered trademark for “Glycemic Expert System for Nurse Implemented Euglycemia” owned by the VA Technology Transfer Program, Department of Veterans Affairs, 810 Vermont Avenue, NW (12TT) Washington DC 20420, and may not be used for any purpose without their express permission. © 2013 All Rights Reserved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The contents of this paper do not reflect the views of the Department of Veterans Affairs or the US Government.
